-
Name
4,4'-Oxydianiline
- EINECS 202-977-0
- CAS No. 101-80-4
- Article Data60
- CAS DataBase
- Density 1.216 g/cm3
- Solubility Insoluble in water
- Melting Point 188-192 °C(lit.)
- Formula C12H12N2O
- Boiling Point 389.4 °C at 760 mmHg
- Molecular Weight 200.24
- Flash Point 218.9 °C
- Transport Information UN 3077 9/PG 3
- Appearance odorless colorless crystals or an odorless fine, beige powder
- Safety 53-45-61-36/37-26-20
- Risk Codes 45-46-23/24/25-51/53-62-36/37/38-22
-
Molecular Structure
-
Hazard Symbols
 T,
T, N
N
- Synonyms 4,4'-Diaminobiphenyl oxide;4,4'-Diaminodiphenyl ether;4,4'-Diaminodiphenyl oxide;4,4'-Diaminophenylether;4,4'-Oxybis(aniline);4,4'-Oxybis[benzenamine];Aniline,4,4'-oxydi- (6CI,8CI);4,4'-Diaminobiphenyl ether;
- PSA 61.27000
- LogP 3.80570
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen; triethylamine In ethanol; water at 110℃; under 30003 Torr; for 48h; Autoclave; | 99% |
| Stage #1: bis(4-nitrophenyl)ether With pyrographite In N,N-dimethyl-formamide at 140℃; for 2h; Stage #2: With 5%-palladium/activated carbon; hydrogen In N,N-dimethyl-formamide at 140℃; under 7500.75 Torr; for 5h; Temperature; | 99.5% |
| With palladium 10% on activated carbon; hydrogen In methanol at 20℃; for 2h; | 98% |
-
-
28896-49-3
bis(4-iodophenyl) ether

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; caesium carbonate | 98% |

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| With hydrogen In ethanol at 75 - 80℃; under 22502.3 Torr; Autoclave; | 90.8% |
-
-
76263-55-3
2-Iodo-4-nitro-1-(4-nitro-phenoxy)-benzene

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| With copper(I) chloride; potassium borohydride In methanol for 0.5h; Ambient temperature; | 82% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride; caesium carbonate; 1,2-bis-(diphenylphosphino)ethane; bis(pinacol)diborane In N,N-dimethyl-formamide at 152℃; for 6h; | 80% |
| Multi-step reaction with 2 steps 1.1: sodium hydride / dimethyl sulfoxide / 0.17 h / 20 °C 1.2: 80 °C 2.1: palladium 10% on activated carbon; hydrogen / methanol / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium hydroxide / dimethyl sulfoxide / 12 h / 80 °C 2: palladium 10% on activated carbon; hydrogen / methanol / 2 h / 20 °C View Scheme |
-
-
98-95-3
nitrobenzene

-
A
-
123-30-8
4-amino-phenol

-
B
-
101-80-4
4,4'-oxydiphenylene diamine

-
C
-
62-53-3
aniline

-
D
-
95-55-6
2-amino-phenol

| Conditions | Yield |
|---|---|
| With hypophosphorous acid; palladium on activated charcoal In water at 80℃; for 4h; Product distribution; var. of temp., ratio, degree of conversion, solvent, catalyst; | A 47% B n/a C n/a D n/a |

-
-
101-84-8
diphenylether

-
A
-
101-80-4
4,4'-oxydiphenylene diamine

-
B
-
139-59-3
4-phenoxyanilin

-
C
-
2688-84-8
2-phenoxyaniline

| Conditions | Yield |
|---|---|
| With O-(4-nitrobenzoyl)hydroxylammonium trifluoromethanesulfonate; iron(II) bromide; silver(I) triflimide In 2,2,2-trifluoroethanol; water at 30℃; for 2h; | A 19.8% B 19.8% C 39.5% |


| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonia; water at 170℃; |

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonia; water at 170℃; | |
| With ammonium hydroxide; caesium carbonate In ethanol; water at 130℃; for 24h; Inert atmosphere; Sealed tube; |

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
7664-93-9
sulfuric acid

-
-
100-65-2
N-Phenylhydroxylamine

-
A
-
101-80-4
4,4'-oxydiphenylene diamine

-
B
-
122-37-2
4-anilinophenol

-
C
-
62-53-3
aniline

-
D
-
92-87-5
p,p'-diaminobiphenyl


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 92 percent / K2CO3 / dimethylformamide / 10 h / 145 - 150 °C 2: 95 percent / NH2NH2; FeCl3; activated charcoal / dioxane View Scheme | |
| Multi-step reaction with 2 steps 1: KOH / dimethylformamide 2: hydrazine hydrate / Raney-nickel View Scheme | |
| Multi-step reaction with 2 steps 1: 230 °C 2: tin; hydrochloric acid; alcohol View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / N,N-dimethyl-formamide / 8 h / 145 - 150 °C / Inert atmosphere 2: iron(III) chloride hexahydrate; pyrographite; hydrazine hydrate / ethanol / 6 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1.1: sodium hydride / dimethyl sulfoxide / 0.17 h / 20 °C 1.2: 80 °C 2.1: palladium 10% on activated carbon; hydrogen / methanol / 2 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 92 percent / K2CO3 / dimethylformamide / 10 h / 145 - 150 °C 2: 95 percent / NH2NH2; FeCl3; activated charcoal / dioxane View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: KOH / dimethylformamide 2: hydrazine hydrate / Raney-nickel View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / N,N-dimethyl-formamide / 8 h / 145 - 150 °C / Inert atmosphere 2: iron(III) chloride hexahydrate; pyrographite; hydrazine hydrate / ethanol / 6 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: fuming nitric acid 2: tin; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 230 °C 2: tin; hydrochloric acid; alcohol View Scheme |

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 2: fuming nitric acid 3: tin; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 2: fuming nitric acid 3: tin; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sodium hydride / dimethyl sulfoxide / 0.17 h / 20 °C 1.2: 80 °C 2.1: palladium 10% on activated carbon; hydrogen / methanol / 2 h / 20 °C View Scheme |
-
-
72606-99-6
1,4,7,10,17,20,23,26-octaoxa<10.10>m-cyclophane-13,29-dicarboxylic acid

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| With pyridine; triphenyl phosphite; lithium chloride In 1-methyl-pyrrolidin-2-one at 100℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| With pyridine; triphenyl phosphite; lithium chloride In 1-methyl-pyrrolidin-2-one at 100℃; for 3h; | 100% |
-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| With pyridine; triphenyl phosphite; lithium chloride In 1-methyl-pyrrolidin-2-one at 100℃; for 3h; | 100% |
-
-
50-00-0
formaldehyd

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
1575814-59-3
6,6'-(4,4'-oxybis(4,1-phenylene))bis(8-nitro-3,5,6,7-tetrahydro-2H-thiazolo[3,2-c]pyrimidine)

| Conditions | Yield |
|---|---|
| In water at 90℃; for 0.05h; Mannich Aminomethylation; Microwave irradiation; Green chemistry; | 100% |

-
-
5431-44-7
2,6-Pyridinedicarboxaldehyde

-
-
21797-13-7
tetrakis(acetonitrile)palladium(II) tetrafluoroborate

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
6016-04-2
N,N’-di(3-pyridyl)-1,4,5,8-naphthalenetetracarboxydiimide

| Conditions | Yield |
|---|---|
| In chloroform; acetonitrile at 60℃; for 20h; | 100% |


| Conditions | Yield |
|---|---|
| With dichloromethane; triethylamine for 24h; Inert atmosphere; | 99.9% |
-
-
100-52-7
benzaldehyde

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
7203-56-7
4,4′-bis(benzylideneamino)diphenyl ether

| Conditions | Yield |
|---|---|
| With acetic acid In toluene for 2h; Reflux; Dean-Stark; | 99% |
| In ethanol Heating; | 69% |
| In ethanol Heating; |
-
-
90-02-8
salicylaldehyde

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
7107-96-2
2,2'-[oxybis(4,1-phenylenenitrilomethylylidene)]diphenol

| Conditions | Yield |
|---|---|
| With acetic acid In toluene for 2h; Dean-Stark; Reflux; | 99% |
| In ethanol at 78℃; for 3h; | 96% |
| In ethanol for 6h; Reflux; | 92% |
| With sodium acetate In methanol at 70℃; for 1h; | 90% |
| In ethanol for 1h; Heating; |

| Conditions | Yield |
|---|---|
| 99% |

| Conditions | Yield |
|---|---|
| With hydrogen sulfide In chloroform at 20 - 40℃; for 3.5h; | 99% |
-
-
5431-44-7
2,6-Pyridinedicarboxaldehyde

-
-
21797-13-7
tetrakis(acetonitrile)palladium(II) tetrafluoroborate

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-Pyridinedicarboxaldehyde; tetrakis(acetonitrile)palladium(II) tetrafluoroborate; 4,4'-oxydiphenylene diamine In acetonitrile at 60℃; for 20h; Stage #2: tris(3-pyridyl)-1,3,5-benzene In acetonitrile at 60℃; for 2h; | 99% |

-
-
82645-24-7
1,5-bis(benzaldehydeoxy)-3-oxopentane

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 264h; | 99% |
-
-
108-24-7
acetic anhydride

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
3070-86-8
N-[4-(4-acetylaminophenoxy)phenyl]acetamide

| Conditions | Yield |
|---|---|
| In acetone at 0℃; | 98% |
| With triethylamine In acetone at 0℃; for 2h; | 98% |
| In chlorobenzene at 90 - 95℃; for 2h; | 93% |
| In acetic acid for 2h; Ambient temperature; | 77.4% |
-
-
101-80-4
4,4'-oxydiphenylene diamine

- polymer, ordered poly(amide-thioether), Mn 19000, Mw 36000; monomer(s): 3,3-[4,4-thiobis(phenylenethio)]bis(2,4-dichlorophenyl propionate); 4,4-oxydianiline
-
polymer, ordered poly(amide-thioether), Mn 19000, Mw 36000; monomer(s): 3,3-[4,4-thiobis(phenylenethio)]bis(2,4-dichlorophenyl propionate); 4,4-oxydianiline

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol In 1-methyl-pyrrolidin-2-one at 80℃; for 48h; | 98% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; Ni(0)*2IPr In 1,4-dioxane at 100℃; | 98% |
-
-
36978-41-3
2,3',3,4'-biphenyltetracarboxylic acid dianhydride

-
-
3711-04-4
2,3,2',3'-biphenyltetracarboxylic dianhydride

-
-
2420-87-3
3,3',4,4'-biphenyltetracarboxylic anhydride

-
-
101-80-4
4,4'-oxydiphenylene diamine

- polymer; monomer(s): 2,3,3\,,4\-biphenyltetracarboxylic dianhydride; 2,3,2\,,3\-biphenyltetracarboxylic dianhydride; 3,4,3\,,4\-biphenyltetracarboxylic dianhydride; 4,4\-oxydianiline
-
polymer; monomer(s): 2,3,3\,,4\-biphenyltetracarboxylic dianhydride; 2,3,2\,,3\-biphenyltetracarboxylic dianhydride; 3,4,3\,,4\-biphenyltetracarboxylic dianhydride; 4,4\-oxydianiline

| Conditions | Yield |
|---|---|
| 98% |

-
-
36978-41-3
2,3',3,4'-biphenyltetracarboxylic acid dianhydride

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| 98% |
-
-
916066-54-1
3,3'-bis(N-aminophthalimide)

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
2421-28-5
dianhydride of benzophenone-3,4,3',4'-tetracarboxy acid

- polymer, Mn/Mw = 1.6, Mn = 51210 g/mol, Mw = 82240 g/mol, inherent viscosity = 0.50 dL/g; monomer(s): 3,3\-bis(N-aminophthalimide); 4,4\-oxydianiline; 3,3\44,4\-biphenyltetracarboxylic acid dianhydride
-
polymer, Mn/Mw = 1.6, Mn = 51210 g/mol, Mw = 82240 g/mol, inherent viscosity = 0.50 dL/g; monomer(s): 3,3\-bis(N-aminophthalimide); 4,4\-oxydianiline; 3,3\44,4\-biphenyltetracarboxylic acid dianhydride

| Conditions | Yield |
|---|---|
| With 4-chloro-phenol; benzoic acid at 100 - 190℃; | 98% |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 12h; Heating; | 98% |
-
-
66-77-3
1-naphthaldehyde

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
387850-01-3
4,4'-bis(1-naphthylmethylideneamino)diphenyl ether

| Conditions | Yield |
|---|---|
| With acetic acid In toluene for 2h; Inert atmosphere; Reflux; Dean-Stark; | 98% |

| Conditions | Yield |
|---|---|
| In ethanol for 3.5h; Reflux; Schlenk technique; Inert atmosphere; | 98% |
| In methanol at 40℃; for 6h; | 80% |
| In methanol at 40℃; for 6h; | 80% |
-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| With pyridine; triphenyl phosphite; lithium chloride; 1-methyl-pyrrolidin-2-one at 115℃; for 96h; | 97% |
-
-
19362-77-7
4,4'-dimercaptodiphenyl sulfide

-
-
4513-42-2
2,4-dichlorophenoxy acrylate

-
-
101-80-4
4,4'-oxydiphenylene diamine

| Conditions | Yield |
|---|---|
| With TEA; benzotriazol-1-ol In 1-methyl-pyrrolidin-2-one at 20 - 80℃; for 50h; | 97% |

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
198328-20-0
bis(5-carboxy-1,3-phenylene)-20-crown-6

| Conditions | Yield |
|---|---|
| With pyridine; triphenyl phosphite; lithium chloride In 1-methyl-pyrrolidin-2-one at 100℃; for 3h; | 97% |

| Conditions | Yield |
|---|---|
| 97% |

| Conditions | Yield |
|---|---|
| With 5,15,10,20-tetraphenylporphyrin; calcium chloride In pyridine; 1-methyl-pyrrolidin-2-one at 60 - 130℃; for 10.5h; Inert atmosphere; | 97% |
-
-
102-09-0
bis(phenyl) carbonate

-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
21649-32-1
4,4'-oxydiphenylene bis-phenylcarbamate

| Conditions | Yield |
|---|---|
| With isobutyric Acid; 1,3,5-tris(N,N-dimethylaminopropyl)-hexahydro-s-triazine In toluene at 55℃; for 48h; | 97% |
-
-
101-80-4
4,4'-oxydiphenylene diamine

-
-
48180-65-0
1-azido-4-(4-azidophenoxy)benzene

| Conditions | Yield |
|---|---|
| Stage #1: 4,4'-oxydiphenylene diamine With toluene-4-sulfonic acid In water for 0.0166667h; Stage #2: With sodium nitrite In water Stage #3: With sodium azide In water | 96% |
| Stage #1: 4,4'-oxydiphenylene diamine With 1-methyl-2-oxopyrrolidinium hydrogen sulfate In water at 20℃; for 0.0166667h; Grinding; Stage #2: With sodium nitrite In water at 20℃; Grinding; Stage #3: With sodium azide In water at 20℃; Grinding; | 94% |
| Stage #1: 4,4'-oxydiphenylene diamine With hydrogenchloride; sodium nitrite In water at 0 - 5℃; for 0.25h; Stage #2: With sodium azide In water at 0℃; | 90% |
4,4'-Oxydianiline Chemical Properties
Molecular structure of 4,4'-Oxydianiline (CAS NO.101-80-4) is:
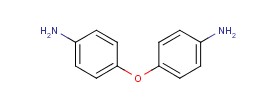
Product Name: 4,4'-Oxydianiline
CAS Registry Number: 101-80-4
IUPAC Name: 4-(4-Aminophenoxy)aniline
Molecular Weight: 200.23648 [g/mol]
Molecular Formula: C12H12N2O
XLogP3: 1.4
H-Bond Donor: 2
H-Bond Acceptor: 3
EINECS: 202-977-0
Melting Point: 188-192 °C(lit.)
Surface Tension: 55.9 dyne/cm
Density: 1.216 g/cm3
Flash Point: 218.9 °C
Enthalpy of Vaporization: 63.87 kJ/mol
Boiling Point: 389.4 °C at 760 mmHg
Vapour Pressure: 2.87E-06 mmHg at 25 °C
Water Solubility: Insoluble. <0.01 g/100 mL at 15 °C
Stability: Stable. Combustible. Incompatible with strong oxidizing agents. Hygroscopic.
Product Categories: Intermediates of Dyes and Pigments; Biphenyl & Diphenyl ether; Diphenyl Ethers (for High-Performance Polymer Research); Functional Materials; Reagent for High-Performance Polymer Research; Monomer for Polyimide film
4,4'-Oxydianiline Uses
4,4'-Oxydianiline (CAS NO.101-80-4) is used as the monomer of polyimide,which is high temperature resistant polymer material. It can be used in the production of a wide variety of polymer resins. It is also used in the production of a wide variety of polymer resins and plastic industry.
4,4'-Oxydianiline Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | 650mg/kg (650mg/kg) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 43, 1982. | |
| mouse | LD50 | intraperitoneal | 300mg/kg (300mg/kg) | BEHAVIORAL: MUSCLE WEAKNESS LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Hygiene and Sanitation Vol. 33(10-12), Pg. 137, 1968. |
| mouse | LD50 | oral | 685mg/kg (685mg/kg) | Hygiene and Sanitation Vol. 33(10-12), Pg. 137, 1968. | |
| rabbit | LD50 | intraperitoneal | 650mg/kg (650mg/kg) | BEHAVIORAL: MUSCLE WEAKNESS LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Hygiene and Sanitation Vol. 33(10-12), Pg. 137, 1968. |
| rabbit | LD50 | oral | 700mg/kg (700mg/kg) | Hygiene and Sanitation Vol. 33(10-12), Pg. 137, 1968. | |
| rat | LD50 | intraperitoneal | 365mg/kg (365mg/kg) | BEHAVIORAL: MUSCLE WEAKNESS LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Hygiene and Sanitation Vol. 33(10-12), Pg. 137, 1968. |
| rat | LD50 | oral | 725mg/kg (725mg/kg) | Hygiene and Sanitation Vol. 33(10-12), Pg. 137, 1968. |
4,4'-Oxydianiline Consensus Reports
NTP 10th Report on Carcinogens. IARC Cancer Review: Group 2B IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 7 ,1987,p. 56.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Sufficient Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 29 ,1982,p. 203.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Inadequate Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 16 ,1978,p. 301.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) . NCI Carcinogenesis Bioassay (feed); Clear Evidence: mouse, rat NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NCI-CG-TR-205 ,1980. . Reported in EPA TSCA Inventory.
4,4'-Oxydianiline Safety Profile
Confirmed carcinogen with experimental carcinogenic, neoplastigenic, and tumorigenic data. Poison by intraperitoneal route. Moderately toxic by ingestion. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx.
Hazard Codes:  T,
T,  N
N
Risk Statements: 45-46-23/24/25-51/53-62-36/37/38-22
R45:May cause cancer.
R46:May cause heritable genetic damage.
R23/24/25:Toxic by inhalation, in contact with skin and if swallowed.
R51/53:Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment.
R62:Risk of impaired fertility.
R36/37/38:Irritating to eyes, respiratory system and skin.
R22:Harmful if swallowed.
Safety Statements: 53-45-61-36/37-26-20
S53:Avoid exposure - obtain special instructions before use.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S61:Avoid release to the environment. Refer to special instructions / safety data sheets.
S36/37:Wear suitable protective clothing and gloves.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S20:When using, do not eat or drink.
RIDADR: UN 3077 9/PG 3
WGK Germany: 3
RTECS: BY7900000
HazardClass: 6.1
PackingGroup: III
HS Code: 29222900
4,4'-Oxydianiline Standards and Recommendations
DFG MAK: Animal Carcinogen, Suspected Human Carcinogen
4,4'-Oxydianiline Specification
4,4'-Oxydianiline , its cas register number is 101-80-4. It also can be called Bis(4-aminophenyl) ether ; Di-(4-aminophenyl)ether ; 4,4'-Diaminobiphenyl oxide ; 4,4'-Diaminodiphenyl ether ; 4,4'-Diaminodiphenyl oxide ; 4,4'-Diaminophenyl ether ; 4,4'-Diaminophenyl oxide ; 4,4'-Oxybisaniline ; 4,4'-Oxybisbenzenamine ; 4,4'-Oxydiphenylamine ; 4,4-Diaminodiphenyl ether ; 4-13-00-01038 (Beilstein Handbook Reference) ; 4-Aminophenyl ether ; AI3-18375 ; Aniline, 4,4'-oxydi- ; BRN 0475735 ; Bis(4-aminophenyl) ether ; Bis(4-aminophenyl)ether ; Bis(p-aminophenyl) ether ; Bis(p-aminophenyl)ether ; CCRIS 491 ; Dadpe ; Diaminodiphenyl ether ; Ether, 4,4'-diaminodiphenyl ; HSDB 1316 ; NCI-C50146 ; NSC 37075 ; Oxybis(4-aminobenzene) ; Oxydi-p-phenylenediamine ; Oxydianiline ; p,p'-Diaminodiphenyl ether ; p,p'-Oxybis(aniline) ; p,p'-Oxydianiline ; p-Aminophenyl ether ; Aniline, 4,4'-oxydi- ; Benzenamine, 4,4'-oxybis- . It is a odorless colorless crystals or an odorless fine, beige powder. It is an ether derivative of aniline. also it is a useful monomer and cross-linking agent for polymers.
Related Products
- 4-06-00-02342 (Beilstein Handbook Reference)
- 4,10-Ace-1,2-benzanthracene
- 4,10-Dioxatricyclo[5.2.1.0(2,6)]decan-8-en-3-one
- 4-(1,1,2,2-Tetrafluoroethoxy)benzoicacid
- 4-(1,1,2,2-Tetrafluoroethoxy)chlorobenzene
- 4-(1,1,2,2-Tetrafluoroethoxy)nitrobenzene
- 4-(1,1,2,2-Tetrafluoroethoxy)toluene
- 4-(1,1-Difluoropropan-2-yl)benzene-1-sulfonyl chloride
- 4-(1,1-Dioxothiazolidin-2-yl)benzoate
- 4′-(1,2,3,4-TETRAHYDRO-4-(4-HYDROXY-2-OXO-2H-1-BENZOPYRAN-3-YL)-2-NAPHTHALENYL)(1,1′-BIPHENYL)-4-CARBONITRILE, cis-
- 101804-01-7
- 1018068-81-9
- 1018069-81-2
- 1018125-39-7
- 1018135-23-3
- 101-81-5
- 10181-56-3
- 101819-99-2
- 101820-58-0
- 10182-48-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View