Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Afine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:92-87-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:92-87-5
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:off-white solid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:Potentially muta
Cas:92-87-5
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung, LG, Merck, Thermo Fisher Scientific and so o
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Wuhan Fortuna Chemical Co.,Ltd
1.High quality : the purity is 99% min . through multiple producing procedures. 2.Competitive price : low price because of our skilled production technolpgy ,save the production cost at most , and give big profit room to our customers224.Safe and fa
Hangzhou Dingyan Chem Co., Ltd
Hangzhou Dingyan Chem is a leading manufacturer and supplier of chemicals in China.We develop,produce and distribute high quality pharmaceuticals, intermediates, special chemicals and other fine chemicals. We could give you: 1.Best quality in your re
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:92-87-5
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:92-87-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
HWRK Chem
in storeAppearance:White or light red crystalline powder Package:25g Application:Organic Chemicals
Bide Pharmatech Ltd
Our mission is to provide high-quality and innovative products to our customers. By offering a broad range of products, custom synthesis and personalized services, Bide can help scientists speeding up their research in the chemical and pharmaceutical
Amadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:92-87-5
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryHENAN SUNLAKE ENTERPRISE CORPORATION
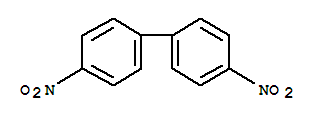
Benzidine Basic information Product Name: Benzidine Synonyms: (1,1’-Biphenyl)-4,4’-diamine;[1,1’-biphenyl]-4,4’-diamine;[1,1'-Biphenyl]-
Cas:92-87-5
Min.Order:500 Gram
Negotiable
Type:Trading Company
inquiryHuBei Ipure Biotech CO.,ltd
HuBei ipure is a diversified product production and operation enterprise, with API, pharma intermediates, and other fine chemicals as well as R&D and pigments development and sales as one of the large enterprises, with more than 130 acres of pl
Cas:92-87-5
Min.Order:1 Kilogram
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryCerametek Materials(ShenZhen)Co., Ltd.
sub/micron to mesh powders (near/spheres, whiskers, flake, fiber, porous) and grain/ball, thin/ thick metal / alloys pieces / parts, thin and thick (particle) film materials /target, and custom-made crystals, substrates/parts. In the industries and a
Hebei Fulong Import & Export Co., Ltd.
Product name:Benzidine Synonyms:(1,1’-Biphenyl)-4,4’-diamine;[1,1’-biphenyl]-4,4’-diamine;[1,1'-Biphenyl]-4,4'-diamine;4,4’-bianiline;4,4’-biphenylenediamine;4,4’-diamino-bipheny;4,4’-diaminodiphenyl;Appearance:crystalline powder Storage:Store at RT
Nanjing Norris Pharm Technology Co., Ltd.
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
Nanjing Chemlin Chemical Co., Ltd.
please contact us to confirm the required quantity and quote, we will provide you with COA,NMR,HPLC and other relevant information Storage:Store in a cool place. Keep container tightly closed in a dry and well-ventilated place Package:as your needs A
Shanghai AngewChem Co., Ltd.
Shanghai AngewChemCo., Ltd. is an innovative enterprise on fine chemicals and pharmaceuticals. Based on Shanghai R&D center and Hunan chemical manufacturing plant, we offer chemical research, process development, and large-scale production. Complete
Cas:92-87-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryPure Chemistry Scientific Inc.
Benzidine Application:822674
Nanjing Raymon Biotech Co., Ltd.
[1,1'-Biphenyl]-4,4'-diamine Storage:keep in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/pharma intermediate Transportation:By Sea,by Air,By courier like DHL or Fedx. Port:Shanghai/Shenzhen
Jiangxi shengwei chemical technology co. LTD
Jiangxi Shengwei Chemical Technology Co., Ltd.supplies scientific research products as below:1.High-purity organic chemical reagents;2.Analytical reagents; 3.Biochemical reagents;4.Chemical raw materials;5.Experimental instruments;6.Experimental cons
Synthetic route

| Conditions | Yield |
|---|---|
| With C24H12Cu2F9N4O7; tetrabutylammomium bromide; ammonia; caesium carbonate In water; ethylene glycol at 110 - 140℃; for 16h; | 100% |
| With bis(tri-ortho-tolylphosphine)palladium(0); (R)-(-)-1-[(S)-2-(dicyclohexylphosphino)ferrocenyl]ethyl-di-tert-butylphosphine; ammonia; sodium t-butanolate In 1,4-dioxane at 100℃; for 12h; Inert atmosphere; | 79% |

| Conditions | Yield |
|---|---|
| With magnesium In water at 100℃; for 2h; Sealed tube; | 96% |
| With tetrabutylammomium bromide; palladium diacetate; potassium carbonate In water; N,N-dimethyl-formamide Buchwald-Hartwig Coupling; | 75% |
| With tetrabutylammonium tetrafluoroborate In acetonitrile at 20℃; for 8h; Electrochemical reaction; | 38% |

| Conditions | Yield |
|---|---|
| With hydrazine In methanol at 50℃; | 95% |
| With sodium tetrahydroborate In ethanol at 24.84℃; for 2h; | 95% |
| With hydrazine hydrate In ethanol at 70℃; for 4h; chemoselective reaction; | 93% |

| Conditions | Yield |
|---|---|
| With carbon dioxide; aluminium; 1-butyl-3-methylimidazolium trifluoromethanesulfonimide at 65℃; under 116262 Torr; for 16h; Ullmann reaction; | 95% |
| With 4-(3'-butyl-1'-imidazolio)-1-butanesulfonic acid hydrogen sulfate; aluminium In carbon dioxide at 45℃; under 116262 Torr; for 16h; Ullmann reaction; Supercritical conditions; | 94% |
| With sulphonic acid functionalized porphyrin meso-substituted triazolium anchored ionic liquid at 20℃; for 16h; Catalytic behavior; Time; Ullmann Condensation; Irradiation; | 91% |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; potassium acetate; bis(pinacol)diborane In ethanol at 60℃; for 8h; Suzuki-Miyaura Coupling; Inert atmosphere; | 94.3% |
| With potassium carbonate In N,N-dimethyl-formamide for 0.75h; | 90% |
| With magnesium In water at 100℃; for 3.5h; Sealed tube; | 85% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetra(n-butyl)ammonium hydroxide; ammonia In water at 25℃; for 24h; Inert atmosphere; Sealed tube; chemoselective reaction; | 91% |
-
-
1221066-38-1
N4,N4'-bis(diphenylmethylene)biphenyl-4,4'-diamine

-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran; water at 20℃; for 2h; | 82% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol at 70℃; for 4h; chemoselective reaction; | A 82% B 7% |
-
-
89415-43-0
(4-aminophenyl)boronic acid

-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; potassium carbonate In water; acetone for 3h; | 80% |

| Conditions | Yield |
|---|---|
| In ethanol; water | A 30% B 70% |
| With hydrogenchloride; lithium chloride In ethanol at 0℃; for 0.5h; Product distribution; kinetic isotope effects; reaction of var. labeled hydrazobenzenes, var. time; | A 91.5 mg B 15.8% |
| With hydrogenchloride; lithium chloride In ethanol at 0℃; for 0.5h; | A 91.5 mg B 15.8% |

| Conditions | Yield |
|---|---|
| With L-Cysteine; potassium hydroxide In water; dimethyl sulfoxide at 100℃; for 0.333333h; Green chemistry; | 70% |
| Multi-step reaction with 2 steps 1: copper / N,N-dimethyl-formamide / Heating 2: hydrazine; 3% Pd/C / ethanol / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: copper / N,N-dimethyl-formamide / 12 h / 160 °C 2: hydrazine; 3% Pd/C / ethanol / 12 h / 20 - 100 °C View Scheme |
-
-
635-46-1
1,2,3,4-tetrahydroisoquinoline

-
-
1211-40-1
4-amino-4'-nitrobiphenyl

-
A
-
91-22-5
quinoline

-
B
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With nickel-nitrogen-doped carbon framework In water at 145℃; for 18h; Inert atmosphere; Sealed tube; Green chemistry; | A 68% B 70% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride In water for 3h; Reflux; | 67% |
| With hydrogenchloride In ethanol at 0℃; for 2.5h; Rearrangement; | 51% |
| Thermodynamic data; |

| Conditions | Yield |
|---|---|
| With LiCrH4*2LiCl*2THF In tetrahydrofuran at 25℃; for 12h; | A 64% B 31% |
| With hydrogenchloride; N-benzylthiazolium bromide; benzaldehyde; triethylamine 1) MeOH, r.t., 20 h, 2) MeOH, HCl; Yield given. Multistep reaction. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol at 70℃; for 4h; chemoselective reaction; | A 27% B 55% |

| Conditions | Yield |
|---|---|
| With L-Cysteine; potassium hydroxide In water; dimethyl sulfoxide at 100℃; for 0.666667h; Green chemistry; | 55% |
-
-
106-40-1
4-bromo-aniline

-
-
7440-66-6
zinc

-
A
-
301300-48-1
NH2C6H4ZnBr

-
B
-
62-53-3
aniline

-
C
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid; cobalt(II) bromide; zinc dibromide In acetonitrile the mixt. in CH3CN was stirred at room temp., then arylbromide was added, stirred at room temp.; GC analysis; | A 40% B 52% C 0% |


| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; titanium tetrachloride In diethyl ether at 20℃; for 15h; Solvent; Inert atmosphere; | A 48% B 25% C 5% |


| Conditions | Yield |
|---|---|
| With 9,10-Dicyanoanthracene; ammonia In water; acetonitrile for 20h; Irradiation; | A 13% B 43% |
-
-
367-24-8
4-bromo-2-fluoroaniline

-
B
-
448-97-5
3,3'-difluorobiphenyl-4,4'-diamine

-
C
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium formate; cetyltrimethylammonim bromide; palladium on activated charcoal In water for 4h; Heating; | A 15% B 33% C 10% |
| Stage #1: 4-bromo-2-fluoroaniline With palladium on activated charcoal; sodium formate; cetyltrimethylammonim bromide; sodium hydroxide In water for 4h; Reflux; Stage #2: With sodium acetate for 20h; Reflux; |

-
-
106-39-8
bromochlorobenzene

-
-
52234-21-6
(adamantane-1,3-diyl)dimethanamine

-
A
-
1287287-13-1
3-(4-chlorophenylaminomethyl)adamantane-1-carbaldehyde

-
B
-
1287287-09-5
4-chloro-N-[3-(4-chlorophenylaminomethyl)adamantan-1-ylmethyl]aniline

-
C
-
106-47-8
4-chloro-aniline

-
D
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; bis(dibenzylideneacetone)-palladium(0); sodium t-butanolate In 1,4-dioxane for 7h; Inert atmosphere; Reflux; | A 25% B 31% C 18% D 7% |

-
-
2515-46-0
3-methyl-1,5-diphenyl-4,5-dihydro-1H-pyrazole

-
A
-
29475-88-5
trans-2-styrylindole

-
B
-
1896-62-4
(E)-benzalacetone

-
C
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With PPA; Polyphosphoric acid (PPA) at 170℃; for 0.666667h; | A 30% B n/a C 0.35 g |

-
-
2515-46-0
3-methyl-1,5-diphenyl-4,5-dihydro-1H-pyrazole

-
A
-
29475-88-5
trans-2-styrylindole

-
B
-
122-57-6
1-Phenylbut-1-en-3-one

-
C
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With PPA at 130℃; Product distribution; Mechanism; studies of the Fischer indole synthesis; | A 25% B 21% C 18% |
| With PPA at 130℃; | A 25% B 21% C 18% |

-
-
106-37-6
1.4-dibromobenzene

-
-
51545-05-2
1,3-bis(aminoethyl)adamantane

-
C
-
1287287-20-0
4-bromo-N-(2-{3-[2-(4-bromophenylamino)ethyl]adamantan-1-yl}ethyl)aniline

-
D
-
106-40-1
4-bromo-aniline

-
E
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; bis(dibenzylideneacetone)-palladium(0); sodium t-butanolate In 1,4-dioxane for 7h; Inert atmosphere; Reflux; | A n/a B n/a C 11% D 7% E 9% |

-
-
7073-69-0
2-(2-bromophenyl)propanol

-
-
214360-73-3
4-(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)aniline

-
B
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate Suzuki Coupling; Inert atmosphere; | A 10% B n/a |

-
-
109-99-9
tetrahydrofuran

-
-
24488-76-4
phenylcalcium iodide

-
A
-
92-52-4
biphenyl

-
B
-
1126-79-0
n-butyl phenyl ether

-
C
-
1227476-15-4
Azobenzene

-
D
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With dinitrogen monoxide Ambient temperature; Yield given; | A n/a B n/a C 6% D n/a |
| With dinitrogen monoxide Product distribution; Ambient temperature; | A n/a B n/a C 6% D n/a |

-
-
110-86-1
pyridine

-
-
855256-72-3
4,4'-Bis-(N'-phenyl-hydrazino)-biphenyl

-
-
64-19-7
acetic acid

-
A
-
120623-26-9
4,4'-Bis-(phenyl-trans-azo)-biphenyl

-
B
-
62-53-3
aniline

-
C
-
1797-23-5
4'-phenylazo-biphenyl-4-ylamine

-
D
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| unter Kohlendioxid; |


| Conditions | Yield |
|---|---|
| With potassium hydroxide; hydrazine hydrate; palladium |

| Conditions | Yield |
|---|---|
| Acetylation; | 100% |
| In acetone for 3h; | 100% |
| for 0.00277778h; Green chemistry; | 98% |
-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With ammonium vanadate; sulfuric acid | 100% |
| With ammonium cerium(IV) nitrate; sulfuric acid In water |

| Conditions | Yield |
|---|---|
| for 120h; Ambient temperature; | 100% |

-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 48h; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; PEG-600 In dichloromethane; water at 20℃; for 1h; Acylation; | 99% |
-
-
37784-17-1
N-(tert-butoxycarbonyl)-D-proline

-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| With N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane at 20℃; for 2h; | 99% |
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| With formic acid In methanol at 20℃; for 25h; Reflux; | 99% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 0.166667h; Milling; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| In melt for 0.333333h; | 99% |
-
-
98-09-9
benzenesulfonyl chloride

-
-
92-87-5
p,p'-diaminobiphenyl

-
-
52945-06-9
N,N'-(biphenyl-4,4'-diyl)dibenzenesulfonamide

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 20℃; for 4h; pH=Ca. 8; | 98% |
| With pyridine |
-
-
591-17-3
meta-bromotoluene

-
-
108-90-7
chlorobenzene

-
-
92-87-5
p,p'-diaminobiphenyl

-
-
65181-78-4
4,4'-bis(m-tolylphenylamino)biphenyl

| Conditions | Yield |
|---|---|
| With chloro[2-(dicyclohexylphosphino)-3 ,6-dimethoxy-2’,4’, 6’-triisopropyl- 1,1’-biphenyl] [2-(2-aminoethyl)phenyl]palladium(II); sodium t-butanolate; ruphos In 1,4-dioxane at 110℃; for 24h; Inert atmosphere; | 98% |

-
-
5396-30-5
9-chloro-1,2,3,4-tetrahydroacridine

-
-
76-05-1
trifluoroacetic acid

-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| Stage #1: 9-chloro-1,2,3,4-tetrahydroacridine; p,p'-diaminobiphenyl With trichlorophosphate In pentan-1-ol for 40h; Reflux; Stage #2: trifluoroacetic acid In water | 98% |

-
-
24677-78-9
2,3-dihydroxybenzaldehyde

-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| In methanol | 97% |
| With ethanol |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
92-87-5
p,p'-diaminobiphenyl

-
-
206182-66-3
tert-butyl (4'-amino-[1,1'-biphenyl]-4-yl)carbamate

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; water; N,N-dimethyl-formamide at 20℃; for 5h; | 97% |
| With sodium carbonate In 1,4-dioxane Ambient temperature; | 90% |
| In 1,4-dioxane at 20℃; for 24h; Inert atmosphere; | 85% |
| In 1,4-dioxane for 24.5h; Inert atmosphere; | 85% |
| In 1,4-dioxane at 20℃; for 25h; Inert atmosphere; | 85% |
-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| Stage #1: p,p'-diaminobiphenyl; naphthalen-2-ylmethyl pyridine-2-carbimidothioate hydrobromide In ethanol; acetonitrile at 20℃; Cooling with ice; Stage #2: With sodium hydroxide In ethanol at 0℃; pH=Ca. 10; Stage #3: With hydrogenchloride In ethanol at 0 - 20℃; | 97% |
-
-
1613522-41-0
1-(benzyloxy)-3-((2,4-difluorobenzyl)carbamoyl)-2-oxo-1,2-dihydro-1,8-naphthyridin-4-yl 4-methylbenzenesulfonate

-
-
92-87-5
p,p'-diaminobiphenyl

-
-
1613522-45-4
4-((4'-amino-[1,1'-biphenyl]-4-yl)amino)-1-(benzyloxy)-N-(2,4-difluorobenzyl)-2-oxo-1,2-dihydro-1,8-naphthyridine-3-carboxamide

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 50℃; for 1h; | 97% |
-
-
58125-40-9
2-(2-chloroacetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carbonitrile

-
-
92-87-5
p,p'-diaminobiphenyl

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 4h; Reflux; | 97% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 20 - 25℃; for 0.75h; Sonication; | 96% |

| Conditions | Yield |
|---|---|
| With 1,10-((ethane-1,2-diylbis(oxy))bis(ethane-2,1-diyl))bis(3-sulfo-1H benzimidazol-3-ium)chloride In neat (no solvent) at 50℃; for 1h; Green chemistry; | 96% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; potassium iodide In water; acetonitrile at 80℃; for 15h; | 95% |
| With tert.-butylhydroperoxide; 3 A molecular sieve; zirconium(IV) tert-butoxide In dichloromethane for 5h; Ambient temperature; | 90% |
| With sulfuric acid Diazotization.Behandlung der Diazoniumsalz-Loesung mit NaNO2 und Kupfer(I)-kupfer(II)-sulfit; |
-
-
123-08-0
4-hydroxy-benzaldehyde

-
-
92-87-5
p,p'-diaminobiphenyl

-
-
132556-90-2
4-amino-4'-(p-hydroxy)benzylideneimino-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In N,N-dimethyl-formamide at 80℃; for 4h; | 95% |
| With piperidine In ethanol Heating; |
-
-
5100-23-2
2-isocyanatobenzoyl chloride

-
-
92-87-5
p,p'-diaminobiphenyl

-
-
131526-24-4
3,3'-(4,4'-Biphenylene)bis(2,4-dioxo-1,2,3,4-tetrahydroquinazoline)

| Conditions | Yield |
|---|---|
| With triethylamine In toluene 1.) 1 h, 0 deg C, 2.) 1 h, room temperature, 3.) 36 h, reflux; | 95% |

| Conditions | Yield |
|---|---|
| In di-isopropyl ether Heating; | 95% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; Ni(0)*2IPr In 1,4-dioxane at 100℃; | 95% |
| With palladium diacetate; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium hydroxide In tetrahydrofuran; ethanol; water; toluene; tert-butyl alcohol at 100℃; for 16h; Glovebox; Inert atmosphere; Schlenk technique; | 116.5 mg |
-
-
90-02-8
salicylaldehyde

-
-
92-87-5
p,p'-diaminobiphenyl

-
-
70952-41-9
4,4'-bis{5-(1-formyl-2-hydroxyphenyl)azo}diphenyl

| Conditions | Yield |
|---|---|
| Stage #1: p,p'-diaminobiphenyl With hydrogenchloride; sodium nitrite In water Cooling with ice; Stage #2: salicylaldehyde With sodium hydroxide In water at 0 - 5℃; | 95% |
| Multistep reaction.; | 76% |
-
-
90-46-0
9-hydroxyxanthene

-
-
92-87-5
p,p'-diaminobiphenyl

-
-
1408244-78-9
N4,N4'-di(9H-xanthen-9-yl)-[1,1'-biphenyl]-4,4’-diamine

| Conditions | Yield |
|---|---|
| With ferric hydrogen sulphate In ethanol for 0.5h; Reflux; regioselective reaction; | 95% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View














 T,
T, N,
N, F,
F, Xn
Xn