-
Name
4-Methoxycinnamic acid
- EINECS 212-594-0
- CAS No. 830-09-1
- Article Data190
- CAS DataBase
- Density 1.195 g/cm3
- Solubility
- Melting Point 173-175 °C
- Formula C10H10O3
- Boiling Point 342.6 °C at 760 mmHg
- Molecular Weight 178.188
- Flash Point 138.6 °C
- Transport Information
- Appearance white fine crystalline powder
- Safety 26-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Cinnamicacid, p-methoxy- (6CI,8CI);3-(4-Methoxyphenyl)-2-propenoic acid;3-(4-Methoxyphenyl)acrylic acid;3-(4-Methoxyphenyl)propenoic acid;4-Methoxycinnamic acid;4'-Methoxycinnamic acid;Bernel hydro;NSC 5303;NSC623437;O-Methyl-p-coumaric acid;p-Methoxycinnamic acid;3-(4-methoxyphenyl)prop-2-enoic acid;2-propenoic acid, 3-(4-methoxyphenyl)-;3-(4-methoxyphenyl)acrylic acid;
- PSA 46.53000
- LogP 1.79300
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium carbonate; palladium diacetate; tris-(o-tolyl)phosphine In N,N,N,N,N,N-hexamethylphosphoric triamide; water at 100℃; for 1.5h; | 98% |
| With tetrabutylammomium bromide; sodium carbonate In water at 90℃; for 22h; Heck Reaction; Green chemistry; | 90% |
| Stage #1: acrylic acid With sodium carbonate In water Stage #2: With copper (II) carbonate hydroxide In water for 0.166667h; Autoclave; Stage #3: 1-bromo-4-methoxy-benzene Reagent/catalyst; Further stages; | 88.1% |
-
-
832-01-9, 3901-07-3, 19310-29-3
methyl p-methoxycinnamate

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With iron(III) sulfate; water In toluene at 110℃; for 2h; Ionic liquid; | 98% |
| With sodium hydroxide In tetrahydrofuran; ethanol; water at 20℃; for 12h; |

| Conditions | Yield |
|---|---|
| With piperidine In pyridine at 100 - 120℃; for 2.25h; | 97.4% |
| With piperidine; pyridine at 80℃; for 7h; | 97.5% |
| With piperidine; pyridine at 75℃; for 7h; Perkin Carboxylic Acid Synthesis; | 97.5% |

| Conditions | Yield |
|---|---|
| With tributyl-amine In N,N-dimethyl-formamide at 90℃; for 7h; Heck Reaction; Reflux; | 97% |
| With potassium hydroxide In water at 90℃; for 5h; Mizoroki-Heck reaction; | 96% |
| Stage #1: acrylic acid With potassium tert-butylate In water at 20℃; for 0.166667h; Inert atmosphere; Stage #2: para-iodoanisole With C16H21Br2N3Pd In water at 100℃; for 24h; Mizoroki-Heck reaction; Inert atmosphere; Sealed tube; | 95% |
-
-
2033-24-1
cycl-isopropylidene malonate

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With 2-carbamoylhydrazine-1-sulfonic acid In neat (no solvent) at 110℃; for 0.416667h; Reagent/catalyst; Green chemistry; | 96% |
-
-
104-92-7
1-bromo-4-methoxy-benzene

-
-
292638-85-8
acrylic acid methyl ester

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium carbonate; palladium In water at 170℃; for 0.25h; Heck Reaction; Microwave irradiation; | 93% |
| With tetrabutylammomium bromide; potassium carbonate In water at 170℃; for 0.333333h; Heck Reaction; Microwave irradiation; | 93% |
-
-
108-24-7
acetic anhydride

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With zinc diacetate; potassium acetate In 5,5-dimethyl-1,3-cyclohexadiene at 145℃; Catalytic behavior; Temperature; Reagent/catalyst; Reflux; Large scale; | 92.5% |
| With sodium tetramethoxyborate; lithium chloride In 1-methyl-pyrrolidin-2-one for 12h; Perkin reaction; Heating; | 59% |
| With sodium acetate at 175 - 180℃; im geschlossenen Rohr; | |
| With sodium acetate at 170℃; im geschlossenen Rohr; | |
| With sodium acetate |
-
-
110621-98-2
Methyl 2-iodo-3-(4-methoxyphenyl)propionate

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol for 3h; Heating; | 89% |
-
-
905966-49-6
(C6H4OCH3)CHCH(BO2C5H10)

-
-
124-38-9
carbon dioxide

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; copper(l) chloride; 1,3-bis[2,6-diisopropylphenyl]imidazolium chloride In tetrahydrofuran at 70℃; under 760.051 Torr; for 24h; | 89% |
-
-
24393-56-4, 51507-22-3, 1929-30-2
ethyl p-methoxycinnamate

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 20℃; for 24h; | 75% |
| Stage #1: ethyl p-methoxycinnamate With sodium hydroxide In ethanol Stage #2: With hydrogenchloride In water | 64% |
-
-
696-62-8
para-iodoanisole

-
-
140-88-5
ethyl acrylate

-
A
-
24393-56-4, 51507-22-3, 1929-30-2
ethyl p-methoxycinnamate

-
B
-
100-66-3
methoxybenzene

-
C
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; dimethyl sulfoxide at 90℃; for 5h; Solvent; Sealed tube; | A 66% B 16% C 19% |
| With potassium carbonate In water; N,N-dimethyl-formamide at 90℃; for 5h; Solvent; Reagent/catalyst; Sealed tube; | A 26% B 22% C 31% |


| Conditions | Yield |
|---|---|
| Stage #1: acetic acid With sodium tetrahydroborate at 20 - 100℃; for 2h; Stage #2: 4-methoxy-benzaldehyde In 1-methyl-pyrrolidin-2-one at 185 - 190℃; for 12h; | 59% |
| Stage #1: acetic acid With sodium tetrahydroborate at 10 - 100℃; for 1.5h; Stage #2: 4-methoxy-benzaldehyde In 1-methyl-pyrrolidin-2-one at 185 - 190℃; for 10h; Knoevenagel Condensation; Reflux; | 55% |

| Conditions | Yield |
|---|---|
| With acetic acid In 1-methyl-pyrrolidin-2-one at 185 - 195℃; | 58% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate In water at 100℃; for 3h; | 50% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate In water at 100℃; for 3h; | 50% |
-
-
696-62-8
para-iodoanisole

-
-
140-88-5
ethyl acrylate

-
A
-
24393-56-4, 51507-22-3, 1929-30-2
ethyl p-methoxycinnamate

-
B
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1,2-dimethoxyethane; water at 90℃; for 5h; Reagent/catalyst; Sealed tube; regioselective reaction; | A 35% B 28% |


| Conditions | Yield |
|---|---|
| With sodium acetate In water at 90℃; for 5h; Sealed tube; regioselective reaction; | 16% |
-
-
36650-51-8, 18166-63-7
4-methoxycinnamic acid amide

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In tetrahydrofuran; methanol; water for 4h; Reflux; | 12% |

| Conditions | Yield |
|---|---|
| at 25℃; under 0.750075 - 75.0075 Torr; for 4h; Schlenk technique; | 6% |

| Conditions | Yield |
|---|---|
| With silver(l) oxide |
-
-
141-82-2
malonic acid

-
-
123-11-5
4-methoxy-benzaldehyde

-
A
-
5678-45-5
3-amino-3-(4-methoxyphenyl)propionic acid

-
B
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With ammonia |


| Conditions | Yield |
|---|---|
| With methanol; potassium carbonate |
-
-
141-82-2
malonic acid

-
-
836-41-9
p-methoxybenzylidene-phenylamine

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With ethanol |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite |

| Conditions | Yield |
|---|---|
| With methanol; potassium hydroxide Verseifen des entstandenen Methylesters mit Alkali; | |
| With methanol; potassium hydroxide at 140℃; im geschlossenen Rohr; Verseifen des entstandenen Methylesters mit Alkali; |
-
-
31448-61-0, 77820-32-7
(2RS,3SR)-2,3-dibromo-3-(4-methoxyphenyl)propanoic acid

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With sodium iodide In 2-methoxy-ethanol at 49.9℃; for 168h; Kinetics; Mechanism; Thermodynamic data; in dark; ΔH(excit.), ΔS(excit.), ΔG(excit.); other temperatures; |

| Conditions | Yield |
|---|---|
| With water; toluene-4-sulfonic acid; acetic acid; copper(l) chloride 1.) reflux, 3.5 h, 2.) reflux, 1.5 h; Yield given. Multistep reaction; |
-
-
52913-16-3
2-(1,3-dioxo-1,3-dihydro-isoindol-2-yl)-3-(4-methoxyphenyl)-propionic acid

-
A
-
136918-14-4
phthalimide

-
B
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 24h; Irradiation; Yield given. Yields of byproduct given; |

4-Methoxycinnamic acid Specification
The p-Methoxycinnamic acid, with the CAS registry number 830-09-1 and EINECS registry number 213-405-4, has the systematic name of 3-(4-methoxyphenyl)prop-2-enoic acid. And the molecular formula of this chemical is C10H10O3. It is a kind of white fine crystalline powder, and belongs to the classification code of Drug/Therapeutic Agent. What's more, it is often used in the cosmetics, and it is also used as medicine intermediate to produce esmolol.
The physical properties of p-Methoxycinnamic acid are as following: (1)ACD/LogP: 2.36; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.89; (4)ACD/LogD (pH 7.4): -0.85; (5)ACD/BCF (pH 5.5): 1.23; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 15.45; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 3; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 35.53 Å2; (13)Index of Refraction: 1.591; (14)Molar Refractivity: 50.38 cm3; (15)Molar Volume: 149 cm3; (16)Polarizability: 19.97×10-24cm3; (17)Surface Tension: 46.2 dyne/cm; (18)Density: 1.195 g/cm3; (19)Flash Point: 138.6 °C; (20)Enthalpy of Vaporization: 61.88 kJ/mol; (21)Boiling Point: 342.6 °C at 760 mmHg; (22)Vapour Pressure: 2.86E-05 mmHg at 25°C.
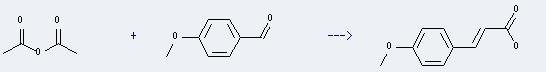
Preparation of p-Methoxycinnamic acid: This chemical can be prepared by acetic acid anhydride and 4-methoxy-benzaldehyde. The reaction will need reagent sodium tetramethoxyborate and LiCl, and the solvent 1-methyl-pyrrolidin-2-one. The reaction time is 12 hours with heating, and the yield is about 59%.
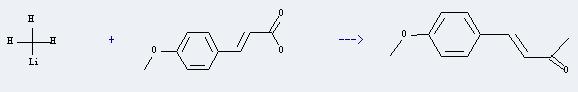
Uses of p-Methoxycinnamic acid: It can react with methyllithium to produce 4t-(4-methoxy-phenyl)-but-3-en-2-one. This reaction will need the solvent diethyl ether. The reaction temperature is 5-20 hours, and the yield is about 62.5%.
You should be cautious while dealing with this chemical. It irritates eyes, respiratory system and skin. Therefore, you had better take the following instructions: Wear suitable, gloves and eye/face protection, and in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)C=Cc1ccc(OC)cc1
(2)InChI: InChI=1/C10H10O3/c1-13-9-5-2-8(3-6-9)4-7-10(11)12/h2-7H,1H3,(H,11,12)
(3)InChIKey: AFDXODALSZRGIH-UHFFFAOYAN
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD | intraperitoneal | > 1300mg/kg (1300mg/kg) | BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) BEHAVIORAL: ATAXIA BEHAVIORAL: REGIDITY | Indian Journal of Pharmaceutical Sciences. Vol. 49, Pg. 77, 1987. |
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View