Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:637-69-4
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:637-69-4
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:637-69-4
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 6 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:637-69-4
Min.Order:1 Kilogram
FOB Price: $1000.0
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:637-69-4
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:637-69-4
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
4-Methoxystyrene CAS:637-69-4 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermedia
Cas:637-69-4
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:637-69-4
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:637-69-4
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:637-69-4
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
4-Methoxystyrene Chemical Properties Melting point 2°C Boiling point 41-42 °C/0.5 mmHg (lit.) density 1.009 g/mL at 25 °C (lit.) refractive index n20/D 1.562 Fp 170 °F storage temp. 2-8°C solubility Sli
Cas:637-69-4
Min.Order:1 Gram
FOB Price: $66.0
Type:Lab/Research institutions
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:637-69-4
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Xiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & CommercialHisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, special chemicals and OLED
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white powder Storage:Sealed in a cool ,dry and microtherm place , avoid light . Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:It is an important raw mat
Win-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:637-69-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,Pharmaceutical intermediates Transportation:air,sea,courier
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:637-69-4
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Cas:637-69-4
Min.Order:0
Negotiable
Type:Manufacturers
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:637-69-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHANGZHOU TIANYE CHEMICALS CO., LTD.
We product this chemical more than 10 years . We are very experience to export it to many countries, Our superior & stable quality , competitive price gain warm reception from our customers. Application:Chemical intermediate
Guangdong Juda Chemical Industrial Co.,Limited
Factory supply high purity low priceAppearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Synthetic route

| Conditions | Yield |
|---|---|
| (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In tetrahydrofuran for 0.0833333h; | 100% |
| (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In tetrahydrofuran for 5h; further catalysts; | 73% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; zinc(II) chloride 1.) THF, RT, 20 min, 2.) RT, 1 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With cesium fluoride; palladium diacetate In 1,4-dioxane at 100℃; for 3h; Stille cross-coupling; | 99% |
| With tris(dibenzylideneacetone)dipalladium(0) chloroform complex; cesium fluoride; 3-tert-butyl-5-methyl-1-(2-(diphenylphosphino)phenyl)-1H-pyrazole In toluene at 60℃; for 10h; Stille coupling; Inert atmosphere; | 88% |
| With cesium fluoride In 1,4-dioxane at 100 - 110℃; for 22h; Stille Cross Coupling; | 80% |

| Conditions | Yield |
|---|---|
| With hydrogen; 3-azapentane-1,5-diamine In methanol at 25℃; for 6h; | 99% |
| With pyridine; hydrogen at 25℃; under 760.051 Torr; for 6h; Reagent/catalyst; Solvent; Green chemistry; chemoselective reaction; | 96% |
| With hydrogen In methanol at 20℃; for 40h; chemoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With [CuCl(ClIPr)]; sodium t-butanolate; tert-butyl alcohol In tetrahydrofuran; hexane at 40℃; for 20h; Inert atmosphere; chemoselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With 1-butyl-3-methyl-3H-imidazol-1-ium fluoride In acetonitrile at 70℃; Hiyama Coupling; Inert atmosphere; | 98% |
| With sodium hydroxide; 4-hydroxyacetophenone oxime-derived palladacycle In water at 120℃; for 0.166667h; Hiyama reaction; microwave irradiation; | 82% |
| With trans-[1,3-bis(2,6-diisopropylphenyl)imidazolin-2-ylidene]PdCl2(NC5H5); sodium hydroxide In 1,4-dioxane; water at 80℃; for 7h; Hiyama cross-coupling; | 41 %Chromat. |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tributylmethylammonium bis(trifluoromethanesulfonyl)imide salt at 160℃; for 5h; | 97.5% |
| With 1,10-Phenanthroline; copper hydroxide In 1-methyl-pyrrolidin-2-one at 210℃; for 1h; Inert atmosphere; Green chemistry; | 68% |
| With copper(I) sulfate; copper(II) sulfate; hydroquinone In quinoline for 0.5h; Heating; | 29.1% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 100℃; for 5h; Reagent/catalyst; Solvent; Temperature; Hiyama Coupling; Sealed tube; | 97% |
| With sodium hydroxide; 4-hydroxyacetophenone oxime-derived palladacycle In water at 120℃; for 0.166667h; Hiyama reaction; microwave irradiation; | 84% |
| With tetrabutyl ammonium fluoride; palladium dichloride In tetrahydrofuran at 65℃; for 9h; Hiyama coupling; | 78% |

| Conditions | Yield |
|---|---|
| With potassium hydrogensulfate at 190℃; under 22 Torr; Pressure; Temperature; Large scale; | 96.3% |
| With phosphotungstic acid In dimethyl sulfoxide at 100℃; for 1h; Inert atmosphere; | 91% |
| With 1,1,1-trichloro-3,3,3-trifluoro-propan-2-one; toluene-4-sulfonic acid; hydroquinone In benzene for 10h; Heating; | 88% |

| Conditions | Yield |
|---|---|
| With hydrogen In hexane at 50℃; under 760.051 Torr; for 10h; | A 96% B 4% |
| With hydrogen In methanol at 20℃; under 760.051 Torr; for 24h; | |
| With hydrogen In methanol at 25℃; for 2h; | A 89 %Chromat. B 11 %Chromat. |

| Conditions | Yield |
|---|---|
| Stage #1: 4-Methoxybenzyl alcohol With triethylphosphine hydrobromide at 100℃; for 0.166667h; Microwave irradiation; Stage #2: formaldehyd With potassium carbonate In water at 100℃; for 0.0833333h; Microwave irradiation; | 96% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetone at 20℃; for 124h; | 96% |
-
-
1227062-94-3
tert-butyl (3R,αR)-3-[N-benzyl-N-(α-methyl-p-methoxybenzyl)amino]-7-iodoheptanoate

-
A
-
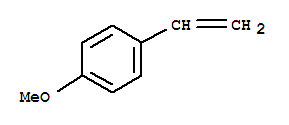
637-69-4
4-Methoxystyrene

-
B
-
1227062-91-0
tert-butyl (R)-2-[N(1)-benzylpiperidin-2-yl]acetate

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 16h; | A 65% B 94% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether 1) -70 deg C, 1-2 h, 2) -70 - 20 deg C, 15 h; | 93% |
| In tetrahydrofuran | 91% |
| In tetrahydrofuran; ethanol at -70℃; for 12h; Product distribution; use of var. solvents (H2O and H2O/EtOH), var. temp. and other reagent (Cl3Mo=CH2); | 92 % Chromat. |
| In tetrahydrofuran; ethanol for 12h; -70 degC to room temp.; | 92 % Chromat. |

| Conditions | Yield |
|---|---|
| With palladium(II) acetylacetonate; potassium fluoride In N,N-dimethyl-formamide at 130℃; under 1500.15 Torr; for 3h; Reagent/catalyst; Time; Hiyama Coupling; Inert atmosphere; | 93% |
-
-
1779-49-3
Methyltriphenylphosphonium bromide

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
637-69-4
4-Methoxystyrene

| Conditions | Yield |
|---|---|
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran at -78℃; for 0.25h; Inert atmosphere; Stage #2: 4-methoxy-benzaldehyde In tetrahydrofuran Inert atmosphere; | 91% |
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran at -78℃; for 2h; Stage #2: 4-methoxy-benzaldehyde In tetrahydrofuran at 20℃; | 88.9% |
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran for 0.25h; Stage #2: 4-methoxy-benzaldehyde In tetrahydrofuran Wittig olefination; Further stages.; | 82% |
-
-
24680-50-0, 71277-11-7, 1963-36-6
4-methoxy-trans-cinnamaldehyde

-
-
637-69-4
4-Methoxystyrene

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; oxygen; sodium carbonate In isopropyl alcohol at 120℃; for 24h; regioselective reaction; | 91% |
| With pyridine; palladium diacetate; triphenylphosphine In water at 100℃; for 24h; Inert atmosphere; | 72% |
| With palladium 10% on activated carbon; sodium carbonate In isopropyl alcohol at 120℃; for 6h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With methoxy(cyclooctadiene)rhodium(I) dimer; N,N-Dimethylacrylamide; 3-Methoxybenzoic acid; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In toluene at 90℃; for 24h; Inert atmosphere; Glovebox; Sealed tube; chemoselective reaction; | 91% |

| Conditions | Yield |
|---|---|
| With Methyltriphenylphosphonium bromide; potassium carbonate In 1,4-dioxane at 100℃; for 16h; Microwave irradiation; Sealed tube; Inert atmosphere; | 90% |
| With aluminium silicate; acetic anhydride at 400℃; | |
| Multi-step reaction with 3 steps 1: 85 percent / tetrahydrofuran 2: 96 percent / H2O2, (NH4)6MoO7*4H2O / ethanol / 65 h / Ambient temperature 3: 63 percent / LDA / tetrahydrofuran / 1) -78 deg C, 1 h, 2) r.t., 1 h View Scheme |

| Conditions | Yield |
|---|---|
| With palladium diacetate; C22H27P; triethylamine In 1,4-dioxane; Hexadecane at 120℃; for 20h; Reagent/catalyst; Heck Reaction; Schlenk technique; Inert atmosphere; Autoclave; | 90% |
| With tributyl-amine; potassium carbonate; palladium In N,N-dimethyl-formamide at 150℃; under 7757.43 Torr; for 1h; Heck reaction; microwave irradiation; | 67% |
| With dichloro bis(acetonitrile) palladium(II); C9H13N3O; triethylamine In N,N-dimethyl-formamide at 160℃; under 4500.45 Torr; for 2h; Catalytic behavior; Heck Reaction; regioselective reaction; | 26% |
| With lithium perchlorate; triethylamine; triphenylphosphine In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide electrolysis; | 70 % Chromat. |
| With dichlorobis(tri-O-tolylphosphine)palladium; triethylamine In acetonitrile at 70℃; under 3345.86 Torr; Temperature; Reagent/catalyst; Heck Reaction; |

| Conditions | Yield |
|---|---|
| With Wilkinson's catalyst; isopropyl alcohol; 4-diphenylphosphanyl-benzoic acid 2-trimethylsilanyl-ethyl ester In tetrahydrofuran at 25℃; for 0.5h; | 90% |
| With (IMes)CuCl; triphenylphosphine; isopropyl alcohol In 1,4-dioxane; diethyl ether at 60℃; for 16h; | 82% |
| With tris(triphenylphosphine)rhodium(l) chloride; triphenylphosphine; isopropyl alcohol In tetrahydrofuran at 25℃; for 2h; Reagent/catalyst; chemoselective reaction; | 73% |
| With RhCl(PPh3)3; triphenylphosphine In 1,4-dioxane; isopropyl alcohol; toluene at 50℃; | 40 mg |
-
-
696-62-8
para-iodoanisole

-
-
4627-10-5
4,4,6-trimethyl-2-vinyl-1,3,2-dioxaborinane

-
-
637-69-4
4-Methoxystyrene

| Conditions | Yield |
|---|---|
| With silver(l) oxide; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 67℃; for 24h; Suzuki-Miyaura reaction; | 90% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 66℃; for 1.5h; | 89% |

| Conditions | Yield |
|---|---|
| With P(i-BuNCH2CH2)2(BnNCH2CH2)N; dimethyl-N-fluoroamine; tris(dibenzylideneacetone)dipalladium (0) In 1,4-dioxane at 100 - 110℃; Stille reaction; | 89% |
| With Pd((CH3)2C6H3NC(CH3)CHC(CH3)NC6H3(CH3)2)(CH3)(P(CH2CH3)3); cesium fluoride In tetrahydrofuran at 50℃; for 6h; Stille Cross Coupling; | 85% |
| With tris(dibenzylideneacetone)dipalladium (0); tri-tert-butyl phosphine; cesium fluoride In 1,4-dioxane at 100℃; for 48h; Stille cross-coupling; | 83% |

| Conditions | Yield |
|---|---|
| With Hoveyda-Grubbs catalyst second generation; di-μ-bromobis-(tritert-butylphosphine)dipalladium(I) In tetrahydrofuran at 60℃; under 7500.75 Torr; for 16h; Autoclave; | 89% |

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one; copper(l) iodide; sodium chloride at 90℃; for 6h; other reagent: MnBr2, NaCl, N-methylpyrrolidone; | 88% |
| With palladium; N,N-dimethyl-formamide In 1-methyl-pyrrolidin-2-one at 120℃; for 15h; | 44% |
| With 9-phosphatriptycene deriv; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran at 50℃; for 2h; Stille coupling; | 64 % Spectr. |
-
-
87305-45-1
(R,S)-NN-dimethyl-N-phenacyl-1-p-methoxyphenylethylammonium bromide

-
A
-
637-69-4
4-Methoxystyrene

-
B
-
62224-36-6
4,4'-(butane-2,3-diyl)bis(methoxybenzene)

-
C
-
3319-15-1
rac-1-(4-methoxyphenyl)-ethanol

-
D
-
87258-60-4
2-dimethylamino-3-p-methoxyphenyl-1-phenylbutan-1-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0℃; for 14h; Further byproducts given; | A 0.4% B 4% C 1.1% D 87% |
| With sodium hydroxide In water at 0℃; for 14h; Further byproducts given; | A 0.4% B 4% C 1.1% D 87% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; C21H19N5Pd(2+)*2BF4(1-) In decane; acetonitrile at 45℃; for 4h; Wacker Oxidation; | 100% |
| With water; oxygen In methanol; dimethyl sulfoxide at 80℃; under 1520.1 Torr; for 20h; Wacker Oxidation; Autoclave; | 99% |
| With palladium diacetate; 9-tert-butyl-10-methylanthracene ozonide In acetonitrile for 72h; Ambient temperature; | 93% |

| Conditions | Yield |
|---|---|
| With hydrogen; 1,5-hexadienerhodium(I)-chloride dimer In hexane for 7h; Ambient temperature; pH=7.6; | 100% |
| With C28H18Fe(1-)*K(1+)*C12H24O6*C4H8O; hydrogen In toluene at 60℃; under 1500.15 Torr; for 24h; Reagent/catalyst; Temperature; Pressure; Time; chemoselective reaction; | 100% |
| With hydrogen In d(4)-methanol at 20℃; under 760.051 Torr; for 6h; | 100% |
-
-
637-69-4
4-Methoxystyrene

-
-
117340-79-1
2-bromo-1-(4-methoxyphenyl)ethanol

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; water In acetonitrile at 20℃; | 100% |
| With N,N'-dibromo-N,N'-(1,2-ethanediyl)bis(p-toluenesulfonamide); ammonium acetate; water In acetone at 20℃; for 0.0333333h; | 95% |
| With hydrogen bromide; dimethyl sulfoxide In water at 60℃; for 12h; | 93% |
-
-
637-69-4
4-Methoxystyrene

-
-
51756-01-5
diisopropoxyphosphorylsulfenyl chloride

-
-
139488-92-9
Thiophosphoric acid S-[2-chloro-2-(4-methoxy-phenyl)-ethyl] ester O,O'-diisopropyl ester

| Conditions | Yield |
|---|---|
| In tetrachloromethane at 25℃; for 2h; Rate constant; | 100% |
| In tetrachloromethane at 25℃; for 2h; | 98% |

| Conditions | Yield |
|---|---|
| With 1H-imidazole; [MnIII(TPP)(OAc)]; tetra-n-butylammonium hydrogen monopersulfate In dichloromethane at 20℃; for 0.5h; | 100% |
| With dihydrogen peroxide In ethanol; water at 50℃; for 1h; Catalytic behavior; Green chemistry; | 99% |
| With dihydrogen peroxide; cobalt(II) acetate In water at 20℃; for 3h; Ionic liquid; Green chemistry; | 98% |

| Conditions | Yield |
|---|---|
| With sodium periodate; C31H29Br2N3Ru*CH2Cl2 In water; ethyl acetate; acetonitrile at 25℃; for 0.5h; Inert atmosphere; Schlenk technique; | 100% |
| With ruthenium trichloride; [bis(acetoxy)iodo]benzene In dichloromethane; water at 30℃; for 2.5h; Solvent; | 92.1% |
| With dihydrogen peroxide In acetonitrile at 65℃; for 6h; | 92% |
-
-
637-69-4
4-Methoxystyrene

| Conditions | Yield |
|---|---|
| In benzene-d6 reaction in NMR tube, soln. was kept at room temp. for 2 h; solvent was removed in vac.; NMR; | 100% |
-
-
637-69-4
4-Methoxystyrene

-
-
1146-43-6
N-(4-hydroxyphenyl)-4-methylbenzenesulfonamide

-
-
1133046-88-4
N-(2-(4-methoxyphenyl)-2,3-dihydrobenzofuran-5-yl)-4-methylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| With bis-[(trifluoroacetoxy)iodo]benzene | 100% |
-
-
637-69-4
4-Methoxystyrene

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
31007-06-4
2-(4-methoxyphenyl)propionitrile

| Conditions | Yield |
|---|---|
| With 3-[bis-(4-methoxy-phenyl)-phosphanyl]-2H-isoquinolin-1-one; N-{6-[bis-(4-methoxy-phenyl)-phosphanyl]-pyridin-2-yl}-2,2-dimethyl-propionamide; bis(1,5-cyclooctadiene)nickel (0) In toluene at 35℃; for 25h; Inert atmosphere; regioselective reaction; | 100% |
-
-
637-69-4
4-Methoxystyrene

| Conditions | Yield |
|---|---|
| With 1-chloro-1λ3-benzo[d][1,2]iodaoxol-3(1H)-one; trimethylsilylazide; cesium fluoride In dichloromethane at 0℃; for 12h; Mechanism; Reagent/catalyst; Temperature; Solvent; | 100% |

| Conditions | Yield |
|---|---|
| With C28H24FN2O3PPd; tetrabutylammomium bromide; sodium acetate at 140℃; for 12h; Catalytic behavior; Inert atmosphere; Schlenk technique; regioselective reaction; | 100% |
-
-
67-56-1
methanol

-
-
637-69-4
4-Methoxystyrene

-
-
42866-92-2
2-(4-methoxyphenyl)acetaldehyde dimethyl acetal

| Conditions | Yield |
|---|---|
| With Pyridine-2,6-dicarboxylic acid; iron(II) tetrafluoroborate hexahydrate; [bis(acetoxy)iodo]benzene at 20℃; for 20h; Molecular sieve; Inert atmosphere; regioselective reaction; | 99% |
| With ammonium iodide; Oxone at 30℃; for 3h; Green chemistry; regioselective reaction; | 78% |
| With thallium(III) nitrate | |
| Stage #1: methanol; 4-Methoxystyrene With ammonium iodide for 0.0166667h; Inert atmosphere; Stage #2: With Oxone at 30℃; |
-
-
637-69-4
4-Methoxystyrene

-
-
55962-05-5
[N-(p-tolylsulfonyl)imino]phenyliodinane

-
-
155721-36-1
2-(4-methoxyphenyl)-1-tosylaziridine

| Conditions | Yield |
|---|---|
| With [CuI(tris[(tetramethylguanidino)-phenyl]amine)][PF6] In acetonitrile for 2h; Inert atmosphere; Molecular sieve; | 99% |
| With C12H18CuN6(2+)*2C16AlF36O4(1-) In acetonitrile at 25℃; for 0.0166667h; Inert atmosphere; | 93% |
| With iodine In water at 20℃; for 12h; Green chemistry; | 87% |

| Conditions | Yield |
|---|---|
| With 2-benzyloxymethylphenyldiphenylphosphine; bi(allylnickel bromide); sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate In dichloromethane at 0 - 23℃; under 760.051 Torr; for 72h; Schlenk technique; | 99% |
| With hexakis(acetonitrile)nickel(II) tetrafluoroborate; diethylaluminium chloride; triphenylphosphine In dichloromethane; toluene at 20℃; under 7500.6 Torr; for 1h; hydrovinylation; | 76% |
| With 2; diethylaluminium chloride; triphenylphosphine In dichloromethane; toluene under 7500.6 Torr; for 0.5h; Yield given; |
-
-
637-69-4
4-Methoxystyrene

-
-
32345-64-5, 147159-72-6, 147254-29-3, 13603-63-9
1-(4-methoxyphenyl) ethane-1,2-diol

| Conditions | Yield |
|---|---|
| With dmap; osmium(VIII) oxide; 4-methylmorpholine N-oxide In water; acetone at 20℃; | 99% |
| Stage #1: 4-Methoxystyrene With formic acid; 2,6-dichloropyridine N-oxide; dichlororuthenium(IV) meso-tetrakis(2,6-dichlorophenyl)porphyrin In chloroform at 20℃; Darkness; Stage #2: With potassium carbonate In methanol | 95% |
| With osmium(VIII) oxide; 4-methylmorpholine N-oxide In tetrahydrofuran; water; tert-butyl alcohol at 20℃; for 12h; Sealed tube; | 94% |
-
-
637-69-4
4-Methoxystyrene

-
-
13296-94-1
2-bromo-4-nitroaniline

-
-
178970-21-3
2-amino-5-nitro-4'-methoxystilbene

| Conditions | Yield |
|---|---|
| With palladium diacetate; triethylamine; tris-(o-tolyl)phosphine In N,N-dimethyl-formamide at 120℃; for 72h; | 99% |
-
-
637-69-4
4-Methoxystyrene

-
-
60556-87-8
2-carbonyl-3-butenenitrile

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 22h; hetero-Diels-Alder cycloaddition; | 99% |
-
-
637-69-4
4-Methoxystyrene

-
-
586-78-7
para-nitrophenyl bromide

-
A
-
4648-33-3
(E)-1-methoxy-4-(4-nitrostyryl)benzene

-
B
-
28358-68-1
1-(4-methoxyphenyl)-1-(4-nitrophenyl)ethylene

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium carbonate; bis-<(4-Cl-2-PdCl-Ph)-(4-Cl-Ph)-methanone oxime> In N,N-dimethyl-formamide at 130℃; for 4h; Product distribution; Further Variations:; Reaction partners; Reagents; Temperatures; Heck-Mizoroki-Jeffery reaction; | A 99% B n/a |


| Conditions | Yield |
|---|---|
| With N-iodo-succinimide In dichloromethane at -78 - 0℃; | 99% |
| Stage #1: 4-Methoxystyrene; 3-methyl-2-buten-1-ol In dichloromethane at 20℃; for 0.0833333h; Stage #2: With N-iodo-succinimide In dichloromethane at -78 - 0℃; Further stages.; | 99% |
-
-
637-69-4
4-Methoxystyrene

| Conditions | Yield |
|---|---|
| With bromodichloromethane; sodium iodide In acetonitrile at 90℃; for 12h; Reagent/catalyst; Solvent; Schlenk technique; Inert atmosphere; Sealed tube; | 99% |
| With bis(acetylacetonato)palladium(II); trifluoroacetic acid In dichloromethane at 20℃; for 0.0833333h; stereoselective reaction; | 92% |
| With ethanol; triphenylphosphine; zinc; bis(triphenylphosphine)dichlorocobalt(II) In acetonitrile at 80℃; for 18h; | 82% |
| With ruthenium trichloride In tetrahydrofuran | 81% |
-
-
637-69-4
4-Methoxystyrene

-
-
2759-28-6
1-phenylmethylpiperazine

-
-
266674-64-0
N-benzyl-N'-[2-(4-methoxyphenyl)ethyl]piperazine

| Conditions | Yield |
|---|---|
| Stage #1: 1-phenylmethylpiperazine With n-butyllithium In tetrahydrofuran; hexane at 20℃; for 0.166667h; Stage #2: 4-Methoxystyrene In tetrahydrofuran; hexane at 20℃; for 24h; Further stages.; | 99% |
-
-
637-69-4
4-Methoxystyrene

-
-
35059-50-8
t-butyl diazoacetate

| Conditions | Yield |
|---|---|
| With C67H51IrN2O2 In tetrahydrofuran at -50℃; for 24.1667h; optical yield given as %ee; enantioselective reaction; | 99% |
| chiral phenyliridium(III)-salen complex In tetrahydrofuran at -50 - 20℃; | 99 % Spectr. |

| Conditions | Yield |
|---|---|
| With CO In tetrahydrofuran | 99% |


| Conditions | Yield |
|---|---|
| With bi(allylnickel bromide); C43H34NO2P; sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate In dichloromethane at -78℃; under 760.051 Torr; for 1h; Reagent/catalyst; | 99% |
| With bi(allylnickel bromide); {benzyl[(S)-1-(1-naphthyl)ethyl]amino}[(R)-1,1'-binaphthyl-2,2'-diyldioxy]phosphine; sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate In dichloromethane at -78℃; for 1h; Inert atmosphere; enantioselective reaction; |
-
-
637-69-4
4-Methoxystyrene

-
-
352-34-1
4-fluoro-1-iodobenzene

-
-
39769-24-9
1-(4-methoxystyryl)-4-fluorobenzene

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 2,5-di(2,4,6-trimethylphenyl)-1,2,5-thiadiazolidine-1-oxide; triethylamine In N,N-dimethyl-formamide at 200℃; for 0.166667h; Heck reaction; Microwave irradiation; | 99% |
| With tris-(dibenzylideneacetone)dipalladium(0); 2,5-di(2,4,6-trimethylphenyl)-1,2,5-thiadiazolidine-1-oxide; triethylamine In N,N-dimethyl-formamide at 200℃; for 0.166667h; Mizoroki-Heck reaction; Microwave irradiation; | 99% |
-
-
637-69-4
4-Methoxystyrene

-
-
460-00-4
1-Bromo-4-fluorobenzene

-
-
39769-24-9
1-(4-methoxystyryl)-4-fluorobenzene

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 2,5-di(2,4,6-trimethylphenyl)-1,2,5-thiadiazolidine-1-oxide; tetrabutylammomium bromide; sodium acetate In 1-methyl-pyrrolidin-2-one at 200℃; for 0.5h; Heck reaction; Microwave irradiation; | 99% |
| With 2,5-di(2,4,6-trimethylphenyl)-1,2,5-thiadiazolidine-1-oxide; tetrabutylammomium bromide; sodium acetate; bis(dibenzylideneacetone)-palladium(0) In 1-methyl-pyrrolidin-2-one at 200℃; for 0.5h; Mizoroki-Heck reaction; Microwave irradiation; | 99% |
| With dichloro bis(acetonitrile) palladium(II); 10F6P(1-)*C235H200N20O10(10+); potassium hydroxide In 1-methyl-pyrrolidin-2-one at 160℃; for 5h; Heck Reaction; Inert atmosphere; | 95% |
| With potassium phosphate; C34H46Br4N8Pd2 In N,N-dimethyl-formamide at 110℃; for 4h; Heck Reaction; | 73% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View