-
Name
6,7-Dimethoxy-3,4-dihydroquinazoline-4-one
- EINECS 635-063-9
- CAS No. 13794-72-4
- Article Data137
- CAS DataBase
- Density 1.33 g/cm3
- Solubility
- Melting Point 309-311 °C
- Formula C10H10N2O3
- Boiling Point 374.1 °C at 760 mmHg
- Molecular Weight 206.201
- Flash Point 180.1 °C
- Transport Information
- Appearance White to off-white crystalline powder
- Safety 26-37/39
- Risk Codes 20/21/22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms 4(1H)-Quinazolinone,6,7-dimethoxy- (9CI);6,7-Dimethoxy-4(3H)-quinazolinone;6,7-Dimethoxy-4-quinazolone;6,7-Dimethoxyquinazoline-4(3H)-one;6,7-Dimethoxyquinazoline-4-one;6,7-Dimethoxy-1H-quinazolin-4-one;
- PSA 64.21000
- LogP 0.94030
Synthetic route
-
-
26759-46-6
methyl 2-amino-4,5-dimethoxybenzoate

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With sodium methylate; formamide In methanol; N,N-dimethyl-formamide for 10h; Heating; | 97% |
-
-
3473-63-0
formamidine acetic acid

-
-
5653-40-7
2-Amino-4,5-dimethoxybenzoic acid

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 140 - 170℃; | 95.7% |
| In 2-methoxy-ethanol at 125℃; for 8h; | 89.5% |
| Stage #1: formamidine acetic acid; 2-Amino-4,5-dimethoxybenzoic acid In ethyl methyl ether Reflux; Stage #2: With ammonium hydroxide In water | 89% |
-
-
20323-74-4
2-carboethoxy-4,5-dimethoxyaniline

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| at 165 - 170℃; | 95% |
| at 168 - 170℃; Inert atmosphere; | 89% |
| at 165 - 170℃; for 6h; Inert atmosphere; | 73.2% |
-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
5653-40-7
2-Amino-4,5-dimethoxybenzoic acid

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| at 110 - 115℃; for 3h; | 94.45% |
| at 140 - 145℃; for 4h; | 93% |
| at 150℃; | 89% |
-
-
26759-46-6
methyl 2-amino-4,5-dimethoxybenzoate

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With ammonia; acetic acid at 150℃; for 2h; Product distribution / selectivity; | 93% |
| With ammonium acetate at 150℃; for 2h; Product distribution / selectivity; | 92.1% |
| With formic acid at 145℃; for 18h; | 91% |
-
-
16712-16-6, 64165-14-6, 65387-22-6, 70179-79-2, 540-69-2
ammonium formate

-
-
5653-40-7
2-Amino-4,5-dimethoxybenzoic acid

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With trimethyl orthoformate In methanol at 70℃; for 4h; Reflux; | 93% |
| With trimethyl orthoformate In methanol at 70℃; for 4h; | 93% |
-
-
13790-39-1
6,7-dimethoxy-4-chloroquinazoline

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With potassium hydroxide In tetrahydrofuran; water at 75℃; for 24h; | 91% |
-
-
5653-40-7
2-Amino-4,5-dimethoxybenzoic acid

-
-
463-52-5
formamidine

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| In 2-methoxy-ethanol at 100℃; | 88% |
-
-
4101-33-1
6,7-dimethoxy-N,N-quinazoline

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With ammonium cerium (IV) nitrate; acetic acid In water | 86% |
| With peracetic acid; sulfuric acid In ethanol at 60℃; for 4h; Solvent; Reagent/catalyst; | 83% |
| With ammonium cerium (IV) nitrate; acetic acid In water at 20℃; for 0.0833333h; |

| Conditions | Yield |
|---|---|
| With [Cp*Ir(2,2′-bpyO)(H2O)]; caesium carbonate at 130℃; for 2h; Microwave irradiation; Green chemistry; | 86% |
| With [Cp*Ir(2,2'-bpyO)(H2O)]; caesium carbonate at 130℃; for 2h; Inert atmosphere; Microwave irradiation; | 86% |
| With oxygen; copper(II) acetate monohydrate; caesium carbonate at 110℃; for 6h; | 49% |
-
-
128823-83-6
methyl 2-amino-5,6-dimethoxybenzoate

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With ammonium formate at 170℃; for 12h; | 86% |
-
-
64-18-6
formic acid

-
-
102714-71-6
2-nitro-4,5-dimethoxybenzonitrile

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| Stage #1: 4,5-dimethoxy-2-nitrobenzonitrile With iron(III) chloride; hydrazine hydrate In methanol; water for 3h; Heating; Stage #2: formic acid With hydrogenchloride In water at 130℃; for 3h; Further stages.; | 85% |
-
-
5653-40-7
2-Amino-4,5-dimethoxybenzoic acid

-
-
149-73-5
trimethyl orthoformate

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With ammonium acetate In methanol at 120℃; for 3h; | 85% |
-
-
26759-46-6
methyl 2-amino-4,5-dimethoxybenzoate

-
-
16712-16-6, 64165-14-6, 65387-22-6, 70179-79-2, 540-69-2
ammonium formate

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| In formamide at 190 - 200℃; for 2h; | 84.7% |
| In formamide at 140℃; for 24h; |
-
-
64-18-6
formic acid

-
-
26759-46-6
methyl 2-amino-4,5-dimethoxybenzoate

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| at 160℃; for 10h; | 84.22% |

-
-
67-56-1
methanol

-
-
26961-27-3
2-amino-4,5-dimethoxybenzonitrile

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With cobalt(II) nitrate hexahydrate; tris(2-diphenylphosphinoethyl)phosphine; water; caesium carbonate at 150℃; for 26h; Inert atmosphere; Sealed tube; | 84% |
-
-
6313-33-3
formamidine hydrochloride

-
-
5653-40-7
2-Amino-4,5-dimethoxybenzoic acid

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| at 210℃; for 0.25h; | 79% |
| at 80 - 200℃; | 65% |
| Stage #1: formamidine hydrochloride; 2-Amino-4,5-dimethoxybenzoic acid at 210℃; for 0.25h; Heating / reflux; Stage #2: With sodium hydroxide In water at 80℃; | 64% |
| Stage #1: formamidine hydrochloride; 2-Amino-4,5-dimethoxybenzoic acid at 210℃; for 0.5h; Stage #2: With sodium hydroxide In water | 64% |
| at 210℃; for 0.5h; | 64% |
-
-
5004-88-6
4,5-dimethoxyanthranilamide

-
-
67-68-5
dimethyl sulfoxide

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; dipotassium peroxodisulfate at 160℃; for 2h; Microwave irradiation; | 62% |
-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
A
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| at 186℃; | A 12.5% B 50% |

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
A
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| at 180 - 185℃; for 2h; | A 11.4% B 46.5% |

-
-
5653-40-7
2-Amino-4,5-dimethoxybenzoic acid

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With formamide In water | 18% |
| Multi-step reaction with 2 steps 1: 81.6 percent / Et3N / dioxane / 2 h / 20 °C 2: 12.5 percent / 186 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 76.8 percent / Et3N / dioxane / 3 h / 20 °C 2: 11.4 percent / 2 h / 180 - 185 °C View Scheme |
-
-
290-87-9
1,3,5-Triazine

-
-
5653-40-7
2-Amino-4,5-dimethoxybenzoic acid

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With piperidine In ethanol Cyclization; |
-
-
5004-88-6
4,5-dimethoxyanthranilamide

-
-
149-73-5
trimethyl orthoformate

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
64-18-6
formic acid

-
-
5004-88-6
4,5-dimethoxyanthranilamide

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| for 5h; Heating; | 1.18 g |
| Heating; |
-
-
2024-83-1
veratronitrile

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: 94 percent / aq. nitric acid / 1 h / 35 °C 2.1: FeCl3; hydrazine hydrate / H2O; methanol / 3 h / Heating 2.2: 85 percent / HCl / H2O / 3 h / 130 °C View Scheme |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: SOCl2 2: NH4OH 3: NaBH4 / CuSO4 4: Heating View Scheme | |
| Multi-step reaction with 4 steps 1: SOCl2 2: aq. NH3 3: CuSO4; NaBH4 4: Heating View Scheme | |
| Multi-step reaction with 3 steps 1.1: thionyl chloride / 0.83 h / Heating 1.2: 88.5 percent / NH4OH 2.1: NaBH4; CuSO4 / methanol / 4 h 3.1: 1.18 g / 5 h / Heating View Scheme |
-
-
4959-60-8
4,5-dimethoxy-2-nitrobenzamide

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaBH4 / CuSO4 2: Heating View Scheme | |
| Multi-step reaction with 2 steps 1: CuSO4; NaBH4 2: Heating View Scheme | |
| Multi-step reaction with 2 steps 1: NaBH4; CuSO4 / methanol / 4 h 2: 1.18 g / 5 h / Heating View Scheme |
-
-
29568-78-3
4,5-dimethoxy-2-nitrobenzoyl chloride

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: NH4OH 2: NaBH4 / CuSO4 3: Heating View Scheme | |
| Multi-step reaction with 3 steps 1: aq. NH3 2: CuSO4; NaBH4 3: Heating View Scheme |
-
-
5004-88-6
4,5-dimethoxyanthranilamide

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With formic acid Heating / reflux; |
-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

-
-
179688-52-9
6-hydroxy-7-methoxyquinazolin-4(3H)-one

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; DL-methionine at 120℃; for 30h; | 100% |
| With methanesulfonic acid; L-methionine at 100℃; for 22h; | 94% |
| With methanesulfonic acid; L-methionine at 100℃; for 22h; | 94% |
-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

-
-
16064-15-6
6,7-dihydroxy-3H-quinazolin-4-one

| Conditions | Yield |
|---|---|
| With hydrogen bromide In water at 135℃; for 18.5h; | 100% |
| Stage #1: 3H-6,7-dimethoxyquinazolin-4-one With hydrogen bromide In water at 110℃; Reflux; Stage #2: With ammonia In water pH=7.0 - 7.5; | 98.62% |
| With hydrogen bromide at 120℃; | 97% |
-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

-
-
13790-39-1
6,7-dimethoxy-4-chloroquinazoline

| Conditions | Yield |
|---|---|
| With thionyl chloride In N,N-dimethyl-formamide for 6h; Heating / reflux; | 98% |
| With thionyl chloride; N,N-dimethyl-formamide for 6h; Heating / reflux; | 98% |
| In N-methyl-acetamide; thionyl chloride; dichloromethane | 98% |

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

-
-
179688-52-9
6-hydroxy-7-methoxyquinazolin-4(3H)-one

| Conditions | Yield |
|---|---|
| In methanesulfonic acid; water | 97% |
-
-
624-70-4
1-chloro-2-iodoethane

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

-
-
1609073-07-5
3-(2-chloroethyl)-6,7-dimethoxyquinazolin-4(3H)-one

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 50℃; Inert atmosphere; | 96% |
| In N,N-dimethyl-formamide at 50℃; | 96% |
-
-
160879-62-9
methyl 1-phenylprop-2-enyl carbonate

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With chloro(1,5-cyclooctadiene)rhodium(I) dimer; (R)-(+)-(6,6'-dimethoxybiphenyl-2,2'-diyl)bis[bis(3,5-di-tert-butylphenyl)phosphine] In 1,2-dichloro-ethane at 80℃; for 24h; Schlenk technique; Inert atmosphere; enantioselective reaction; | 92% |
-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

-
-
1260640-98-9
4-bromo-6,7-dimethoxyquinazoline

| Conditions | Yield |
|---|---|
| With carbon tetrabromide; triphenylphosphine In dichloromethane at 0 - 20℃; for 2h; | 90.35% |
-
-
108-24-7
acetic anhydride

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

-
-
179688-53-0
6-acetoxy-7-methoxy-3,4-dihydroquinazolin-4-one

| Conditions | Yield |
|---|---|
| Stage #1: 3H-6,7-dimethoxyquinazolin-4-one With methanesulfonic acid; DL-methionine at 120℃; for 24h; Stage #2: acetic anhydride With pyridine at 100℃; for 22h; | 87% |
| With methanesulfonic acid; DL-methionine 1.) 100 deg C, 3 h, 2.) Py; Multistep reaction; |
-
-
2969-81-5
4-bromoethylbutanoate

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 10h; Alkylation; | 87% |
-
-
539-74-2
Ethyl 3-bromopropionate

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 10h; Alkylation; | 82% |

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate In water; dimethyl sulfoxide at 50℃; for 24h; Minisci Aromatic Substitution; Sealed tube; | 82% |
-
-
821-06-7
(E)-1,4-dibromobutene

-
-
13794-72-4
3H-6,7-dimethoxyquinazolin-4-one

| Conditions | Yield |
|---|---|
| Stage #1: 3H-6,7-dimethoxyquinazolin-4-one With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 0.5h; Stage #2: (E)-1,4-dibromobutene In N,N-dimethyl-formamide at 20℃; for 2h; | 81% |
6,7-Dimethoxy-3,4-dihydroquinazoline-4-one Chemical Properties
IUPAC Name: 6,7-Dimethoxy-1H-quinazolin-4-one
Synonyms of 6,7-Dimethoxy-3,4-dihydroquinazoline-4-one (CAS NO.13794-72-4): 6,7-Dimethoxy-1H-quinazolin-4-one ; 6,7-Dimethoxy-4(3H)-quinazolinone ; 4(1H)-Quinazolinone,6,7-dimethoxy- (9CI)
CAS NO: 13794-72-4
Molecular Formula: C10H10N2O3
Molecular Weight: 206.20
Molecular Structure: 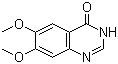
H bond acceptors: 5
H bond donors: 1
Freely Rotating Bonds: 2
Polar Surface Area: 51.13 Å2
Index of Refraction: 1.6
Molar Refractivity: 52.89 cm3
Molar Volume: 154.5 cm3
Surface Tension: 44.6 dyne/cm
Density: 1.33 g/cm3
Flash Point: 180.1°C
Melting point: 309-311°C
Enthalpy of Vaporization: 62.15 kJ/mol
Boiling Point: 374.1 °C at 760 mmHg
Vapour Pressure: 8.53E-06 mmHg at 25°C
SMILES: O=C2\N=C/Nc1cc(OC)c(OC)cc12
InChI: InChI=1/C10H10N2O3/c1-14-8-3-6-7(4-9(8)15-2)11-5-12-10(6)13/h3-5H,1-2H3,(H,11,12,13)
InChIKey: DMSRMHGCZUXCMJ-UHFFFAOYAF
Std. InChI: InChI=1S/C10H10N2O3/c1-14-8-3-6-7(4-9(8)15-2)11-5-12-10(6)13/h3-5H,1-2H3,(H,11,12,13)
Std. InChIKey: DMSRMHGCZUXCMJ-UHFFFAOYSA-N
Product Categories of 6,7-Dimethoxy-3,4-dihydroquinazoline-4-one (CAS NO.13794-72-4): blocks;Heterocycles;Quinolines;pharmacetical;Tinibs;Intermediates;Intermediates & Fine Chemicals;Pharmaceuticals
6,7-Dimethoxy-3,4-dihydroquinazoline-4-one Uses
6,7-Dimethoxy-3,4-dihydroquinazoline-4-one (CAS NO.13794-72-4) is used ad Gefitinib intermediate.
6,7-Dimethoxy-3,4-dihydroquinazoline-4-one Safety Profile
Safety Information about 6,7-Dimethoxy-3,4-dihydroquinazoline-4-one (CAS NO.13794-72-4):
Hazard Codes: Xn,Xi
Xn,Xi
Risk Statements: 20/21/22-36/37/38
R20/21/22: Harmful by inhalation, in contact with skin and if swallowed.
R36/37/38: Irritating to eyes, respiratory system and skin.
Safety Statements: 26-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39: Wear suitable gloves and eye/face protection.
Hazard Note: Irritant
HazardClass: IRRITANT
Related Products
- 6-α,21-DIMETHYLETHISTERONE
- 6′-CHLORO-2-(ETHYLAMINO)-o-VALERO-TOLUIDIDE HYDROCHLORIDE
- 6′-CHLORO-2-(METHYLAMINO)-o-PROPIONO TOLUIDIDE HYDROCHLORIDE
- 6′-CHLORO-3-(2,6-DIMETHYLPIPERIDINO)-o-PROPIONOTOLUIDIDE HYDROCHLORIDE
- 6((-4-(Diethylamino)-1-methylbutyl))-5,8-dimethoxy-2,4-dimethylquinoline-1,5-naphthalene disulfonate
- 6-((4-(Diethylamino)-1-methylbutyl)-amino)-2-methyl-4,5,8-trimethoxy-quinoline-1,5-naphthalene disulfonate
- 6-((p-(Dimethylamino)phenyl)azo)benzothiazole
- 6-((p-(Dimethylamino)phenyl)azo)quinoline
- 6-((p-(Dimethylamino)phenyl)azo)quinoline 1-oxide
- 6-((p-Hydroxyphenyl)azo)uracil
- 137953-98-1
- 137958-96-4
- 13796-22-0
- 137967-81-8
- 137-97-3
- 137973-76-3
- 137975-06-5
- 137976-09-1
- 137976-61-5
- 137977-97-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View