-
Name
ATRANORIN
- EINECS
- CAS No. 479-20-9
- Density 1.404g/cm3
- Solubility
- Melting Point 156-158°C
- Formula C19H18 O8
- Boiling Point 535.7°C at 760 mmHg
- Molecular Weight 374.347
- Flash Point 189.3°C
- Transport Information
- Appearance
- Safety A poison by skin contact. A skin irritant. When heated to decomposition it emits acrid smoke and irritating vapors.
- Risk Codes
-
Molecular Structure
- Hazard Symbols A poison by skin contact. A skin irritant.
- Synonyms Atranoricacid (6CI); Isophthalaldehydic acid, 2,4-dihydroxy-6-methyl-, 4-ester withmethyl 3,6-dimethyl-b-resorcylate (8CI); Antranorin; Atranorin; Atranorine; NSC 249980; NSC685591; NSC 87512; Parmelin; Parmelin acid; Usnarin; Usnarin acid
- PSA 130.36000
- LogP 2.54690
Synthetic route
-
-
479-20-9
atranorin

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; ethanol In 1,2-dimethoxyethane for 16h; | 99% |
| With sodium tetrahydroborate In methanol at 0 - 20℃; for 1h; | 84% |
-
-
479-20-9
atranorin

-
-
90689-60-4
physodalic acid

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In 1,4-dioxane for 4h; Heating; | 91% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 89% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 86.6% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 86% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 83% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 83% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 82.6% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 82% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 81.2% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 73.3% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 73.3% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 52.7% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 50℃; for 2h; | 50.7% |

| Conditions | Yield |
|---|---|
| With alkali at 150℃; im Rohr; |
-
-
64-17-5
ethanol

-
-
479-20-9
atranorin

-
A
-
4707-47-5
2,4-dihydroxy-3,6-dimethyl-benzoic acid methyl ester

-
B
-
39503-14-5
ethyl hematommate

| Conditions | Yield |
|---|---|
| With alkali at 150℃; im Rohr; |

-
-
123-51-3
i-Amyl alcohol

-
-
479-20-9
atranorin

-
A
-
4707-47-5
2,4-dihydroxy-3,6-dimethyl-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With alkali at 150℃; im Rohr; |


| Conditions | Yield |
|---|---|
| With formic acid | |
| With water at 150℃; | |
| With sodium carbonate |

| Conditions | Yield |
|---|---|
| With acetic acid | |
| With water | |
| With ethanol |
-
-
479-20-9
atranorin

-
A
-
4707-47-5
2,4-dihydroxy-3,6-dimethyl-benzoic acid methyl ester

-
B
-
34874-90-3
methyl haematommate

| Conditions | Yield |
|---|---|
| With methanol at 150℃; im Rohr; |
-
-
479-20-9
atranorin

-
A
-
4707-47-5
2,4-dihydroxy-3,6-dimethyl-benzoic acid methyl ester

-
B
-
62392-82-9
2,4-dihydroxy-6-methyl-3-(phenylimino-methyl)-benzoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With ethanol; aniline |

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
479-20-9
atranorin

-
-
5038-46-0
methyl 4-(2,4-dihydroxy-3,6-dimethylbenzoyloxy)-2-hydroxy-3,6-dimethylbenzoate

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; acetic acid Hydrogenation; |
-
-
479-20-9
atranorin

| Conditions | Yield |
|---|---|
| With acetic acid at 80℃; |
-
-
479-20-9
atranorin

-
-
108-24-7
acetic anhydride

-
-
15257-10-0
2-acetoxy-4-(2,4-diacetoxy-3-diacetoxymethyl-6-methyl-benzoyloxy)-3,6-dimethyl-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With perchloric acid |
-
-
479-20-9
atranorin

-
-
108-24-7
acetic anhydride

-
-
15222-51-2
2-acetoxy-4-(2,4-diacetoxy-3-formyl-6-methyl-benzoyloxy)-3,6-dimethyl-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With pyridine |

| Conditions | Yield |
|---|---|
| at 80℃; beim Eindunsten; |
-
-
64-17-5
ethanol

-
-
479-20-9
atranorin

-
-
62-53-3
aniline

-
A
-
4707-47-5
2,4-dihydroxy-3,6-dimethyl-benzoic acid methyl ester

-
B
-
62392-82-9
2,4-dihydroxy-6-methyl-3-(phenylimino-methyl)-benzoic acid ethyl ester


| Conditions | Yield |
|---|---|
| With methanol |

ATRANORIN Chemical Properties
Atranorin is also named as Parmelin;Usnarin;3-hydroxy-4-(methoxycarbonyl)-2,5-dimethylphenyl 3-formyl-2,4-dihydroxy-6-methylbenzoate;3-Formyl-2,4-dihydroxy-6-methylbenzoic acid 3-hydroxy-4-(methoxycarbonyl)-2,5-dimethylphenyl ester;NSC-685591,and so on. Atranorin is usually colorless crystal or crystalline powder.
CAS: 479-20-9
Molecular Formula: C19H18O8
Molecular Weight: 374.34
EINECS: 207-527-7
Molecular structure: 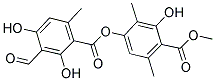
Melting point: 156-158°C
ATRANORIN Uses
ATRANORIN Toxicity Data With Reference
| 1. | skn-gpg 5% | CODEDG Contact Dermatitis. Environmental and Occupational Dermatitis. 11 (1984),168. | ||
| 2. | skn-gpg TDLo:1 pph | CODEDG Contact Dermatitis. Environmental and Occupational Dermatitis. 11 (1984),168. |
ATRANORIN Safety Profile
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View