-
Name
ANTHRANILIC ACID CINNAMYL ESTER
- EINECS
- CAS No. 87-29-6
- Article Data3
- CAS DataBase
- Density 1,18 g/cm3
- Solubility 0.23mg/L(temperature not stated)
- Melting Point 64-66°C
- Formula C16H15 N O2
- Boiling Point 322°C
- Molecular Weight 253.301
- Flash Point 322°C
- Transport Information
- Appearance
- Safety Suspected carcinogen with experimental carcinogenic and neoplastigenic data. Mutation data reported. See also ESTERS. Combustible liquid. When heated to decomposition it emits toxic fumes of NOx.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 2-Propen-1-ol,3-phenyl-, 2-aminobenzoate (9CI); Anthranilic acid, cinnamyl ester(6CI,7CI,8CI); Cinnamyl alcohol, anthranilate (8CI); 3-Phenyl-2-propenyl2-aminobenzoate; 3-Phenyl-2-propenyl anthranilate; Cinnamyl anthranilate; NSC46286
- PSA 52.32000
- LogP 3.72020
Anthranilic acid, cinnamyl ester Chemical Properties
Molecule structure of Anthranilic acid, cinnamyl ester (CAS NO.87-29-6):
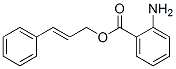
IUPAC Name: [(E)-3-Phenylprop-2-enyl] 2-aminobenzoate
Molecular Weight: 253.2958 g/mol
Molecular Formula: C16H15NO2
Density: 1.178 g/cm3
Melting Point: 64-66 °C
Boiling Point: 449.1 °C at 760 mmHg
Flash Point: 269.4 °C
Index of Refraction: 1.642
Molar Refractivity: 77.72 cm3
Molar Volume: 214.9 cm3
Polarizability: 30.81×10-24 cm3
Surface Tension: 51.6 dyne/cm
Enthalpy of Vaporization: 70.77 kJ/mol
Vapour Pressure: 2.95E-08 mmHg at 25 °C
XLogP3: 4
H-Bond Donor: 1
H-Bond Acceptor: 3
Rotatable Bond Count: 5
Tautomer Count: 2
Exact Mass: 253.110279
MonoIsotopic Mass: 253.110279
Topological Polar Surface Area: 52.3
Heavy Atom Count: 19
Complexity: 308
Canonical SMILES: C1=CC=C(C=C1)C=CCOC(=O)C2=CC=CC=C2N
Isomeric SMILES: C1=CC=C(C=C1)/C=C/COC(=O)C2=CC=CC=C2N
InChI: InChI=1S/C16H15NO2/c17-15-11-5-4-10-14(15)16(18)19-12-6-9-13-7-2-1-3-8-13/h1-11H,12,17H2/b9-6+
InChIKey: GABQNAFEZZDSCM-RMKNXTFCSA-N
EINECS: 201-738-8
Anthranilic acid, cinnamyl ester Toxicity Data With Reference
| 1. | mmo-esc 10 µg/plate | ENMUDM Environmental Mutagenesis. 7 (Suppl 5)(1985),1. | ||
| 2. | mic-mus:lym 10 mg/L | SCIEAS Science. 236 (1987),933. | ||
| 3. | orl-rat LD50:5000 mg/kg | FCTXAV Food and Cosmetics Toxicology. 13 (1975),681. | ||
| 4. | skn-rbt LD50:5000 mg/kg | FCTXAV Food and Cosmetics Toxicology. 13 (1975),681. |
Anthranilic acid, cinnamyl ester Consensus Reports
IARC Cancer Review: Group 3 IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 7 (1987),p. 56.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Limited Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 31 (1983),p. 133.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Inadequate Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 16 (1978),p. 287.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; NCI Carcinogenesis Bioassay (feed); Clear Evidence: mouse, rat NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NCI-CG-TR-196,80 ,1980. . Reported in EPA TSCA Inventory.
Anthranilic acid, cinnamyl ester Safety Profile
Safety Statements: 22-24/25
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
RTECS: CB2725000
Suspected carcinogen with experimental carcinogenic and neoplastigenic data. Mutation data reported. See also ESTERS. Combustible liquid. When heated to decomposition it emits toxic fumes of NOx.
Anthranilic acid, cinnamyl ester Specification
Anthranilic acid, cinnamyl ester (CAS NO.87-29-6) is also named as 2-Aminobenzoic acid 3-phenyl-2-propenyl ester ; 2-Propen-1-ol, 3-phenyl-, 2-aminobenzoate ; 3-Phenyl-2-propen-1-ol 2-aminobenzoate ; 3-Phenyl-2-propen-1-yl 2-aminobenzoate ; 3-Phenyl-2-propen-1-yl anthranilate ; 3-Phenyl-2-propenyl anthranilate ; 3-Phenyl-2-propenylanthranilate ; AI3-36203 ; Benzoic acid, 2-amino-, 3-phenyl-2-propenyl ester ; CCRIS 171 ; Cinnamyl 2-aminobenzoate ; Cinnamyl alcohol, anthranilate ; Cinnamyl anthranilate ; Cinnamyl anthranylate ; Cinnamyl o-aminobenzoate ; Cinnamylester kyseliny anthranilove ; Cinnamylester kyseliny anthranilove [Czech] ; FEMA No. 2295 ; HSDB 4127 ; NCI-C03510 ; NSC 46286 . Anthranilic acid, cinnamyl ester (CAS NO.87-29-6) is brownish or reddish-yellow powder or light yellow solid with a fruity taste. And it is insoluble in water. Anthranilic acid, cinnamyl ester is sensitive to oxidation and hydrolysis. It is subject to photooxidation. Anthranilic acid, cinnamyl ester reacts with various aldehydes, forming highly colored compounds. Anthranilic acid, cinnamyl ester emits toxic fumes of nitrogen oxides when heated to decomposition. Flash point data for Anthranilic acid, cinnamyl ester emits toxic fumes of nitrogen oxides are not available; however, it is probably combustible.
Related Products
- Anthranilic acid
- Anthranilic acid, 3,5-diiodo-
- Anthranilic acid, cinnamyl ester
- ANTHRANILIC ACID, LINALYL ESTER
- Anthranilic acid, methylenedi-, dimethyl ester
- ANTHRANILIC ACID, PHENETHYL ESTER
- 87296-66-0
- 872-98-0
- 87298-93-9
- 87299-57-8
- 872998-61-3
- 873001-54-8
- 873054-44-5
- 873054-97-8
- 873055-22-2
- 873075-54-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View