-
Name
Chlorodiphenylphosphine
- EINECS 214-093-2
- CAS No. 1079-66-9
- Article Data87
- CAS DataBase
- Density 1.229 g/mL at 25 °C(lit.)
- Solubility reacts violently with water
- Melting Point 14-16 °C
- Formula C12H10ClP
- Boiling Point 319.998 °C at 760 mmHg
- Molecular Weight 220.638
- Flash Point 147.33 °C
- Transport Information UN 3265 8/PG 2
- Appearance Colorless to light yellow liquid
- Safety 26-36/37/39-45-8-30-25-27
- Risk Codes 14-34-29-36
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms Phosphinouschloride, diphenyl- (6CI,8CI,9CI);Chlorodiphenylphosphine;Diphenylchlorophosphine;Diphenylphosphine chloride;Diphenylphosphinouschloride;Diphenylphosphorus chloride;NSC 60407;Phosphine, chlorodiphenyl-;
- PSA 13.59000
- LogP 3.27310
Synthetic route

| Conditions | Yield |
|---|---|
| With acetyl chloride In tetrahydrofuran at 100℃; for 12h; Temperature; Reagent/catalyst; Inert atmosphere; Sealed tube; | 99% |
| With acetyl chloride In tetrahydrofuran at 25℃; Solvent; Temperature; Reagent/catalyst; Inert atmosphere; | 93% |
| With phosphorus trichloride In toluene at 20℃; for 1h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With zinc trifluoromethanesulfonate; phosphorus trichloride In tetrachloromethane at 0 - 60℃; for 12h; | 93% |
| With chlorine Destillation des entstandenen Triphenylphosphindichlorids; |

| Conditions | Yield |
|---|---|
| at 350℃; for 5h; | 85% |
-
-
97108-40-2
2-chloro-2,2-diphenyl-3,5-bis(1,1,2,2-tetrafluoroethyl)-1,4,2-dioxaphospholane

-
A
-
756-04-7
2,2,3,3-tetrafluoropropionaldehyde

-
B
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| under 0.06 Torr; Heating; | A 55.5% B 80% |
-
-
97108-41-3
2-chloro-3,5-bis(1,1,2,2,3,3,4,4-octafluorobutyl)-2,2-diphenyl-1,4,2-dioxaphospholane

-
A
-
2648-47-7
2,2,3,3,4,4,5,5-octafluorovaleraldehyde

-
B
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| under 0.06 Torr; Heating; | A 45.7% B 75% |
| under 0.06 Torr; Heating; | A 45.7% B 75% |
-
-
1159415-91-4, 1306641-30-4, 1306641-41-7
[Pd(BIMIONAP)Cl2]

-
B
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| With tetraethylammonium chloride In acetonitrile at 50℃; for 3h; Inert atmosphere; | A 62% B n/a |
-
-
75-77-4
chloro-trimethyl-silane

-
-
20472-49-5
ethyl diphenylthiophosphinite

-
-
100-52-7
benzaldehyde

-
A
-
107-46-0
Hexamethyldisiloxane

-
B
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| at 200℃; for 2h; | A 57% B n/a C 30% |


-
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| With aluminium trichloride; phosphorus trichloride at 140 - 150℃; for 5h; | 52% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride; phosphorus trichloride for 15h; Heating; | 52% |
| Stage #1: benzene With aluminum (III) chloride; phosphorus trichloride at 140 - 150℃; for 8.8h; Stage #2: With α-picoline at 25 - 40℃; | 47.5% |
| Stage #1: benzene With aluminum (III) chloride; trichlorophosphate at 60℃; for 2h; Inert atmosphere; Stage #2: at 116 - 120℃; under 1275.13 - 1350.14 Torr; for 5h; Temperature; Autoclave; |
-
-
104892-86-6
α-ethylthio-β,β,β-trichloroethyl diphenylphosphonite

-
A
-
18271-61-9
α,β,β,β-tetrachloroethyl ethyl sulfide

-
B
-
95028-66-3
β,β-dichlorovinyl diphenyl phosphonate

-
C
-
20472-49-5
ethyl diphenylthiophosphinite

-
D
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| Product distribution; Heating; thermal decomposition; | A 41.6% B 33.3% C 42.2% D 38.7% |

-
-
17564-64-6
N-(chloromethyl)phthalimide

-
-
1636-15-3
(diethylamino)diphenylphosphine

-
A
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| In Petroleum ether at 90 - 100℃; for 2.5h; | A n/a B 33.7% |


| Conditions | Yield |
|---|---|
| In dichloromethane Ar atmosphere; addn. of SnCl2 to org. compd. in CH2Cl2 with stirring; filtration, washing(CH2Cl2), drying (vac.); elem. anal.; | A 31% B n/a |


| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane at 200℃; for 2h; Yields of byproduct given; | A n/a B 30% |


| Conditions | Yield |
|---|---|
| With aluminium trichloride; phosphorus trichloride at 140 - 150℃; for 5h; Product distribution; Mechanism; var. temps; var. molar proportion of reactants; var. time; | A 14% B 20% |

| Conditions | Yield |
|---|---|
| With chlorine beim anschliessenden Erhitzen mit rotem Phosphor; |
-
-
28557-00-8
phenylzinc chloride

-
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| With phosphorus trichloride |

| Conditions | Yield |
|---|---|
| at 220 - 230℃; |
-
-
644-97-3
Dichlorophenylphosphine

-
-
587-85-9
diphenylmercury(II)

-
A
-
1079-66-9
chloro-diphenylphosphine

-
B
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| at 230 - 240℃; | |
| at 230 - 240℃; |


| Conditions | Yield |
|---|---|
| at 300℃; | |
| With phosphorus; chlorobenzene at 380℃; for 4h; Temperature; Autoclave; Large scale; | 219.5 kg |

| Conditions | Yield |
|---|---|
| With alkali metal | |
| With magnesium | |
| With pyrographite | |
| With phosphorus | |
| With phosphorous acid trimethyl ester; triethyl phosphite |
-
-
56372-47-5
N,N-diethyl P,P-diphenylphosphinamide

-
A
-
1079-66-9
chloro-diphenylphosphine

-
B
-
1101-41-3
Tetraphenyldiphosphin

-
C
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With pyridine; trichlorosilane In benzene Title compound not separated from byproducts; |

-
-
63507-05-1
2,2-dimethyl-1-propyl diphenylphosphinite

-
A
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether; dichloromethane-d2 at 25℃; Product distribution; Mechanism; Variation of temperature.; |
-
-
81456-78-2
(Dichloracetyl)diphenylphosphan

-
A
-
29804-89-5
chloroketene

-
B
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| at 80℃; |
-
-
81456-79-3
(2,2-Dichlorpropionyl)diphenylphosphan

-
A
-
1079-66-9
chloro-diphenylphosphine

-
B
-
13363-86-5
chloro-methyl-ketene

| Conditions | Yield |
|---|---|
| at 90℃; |
-
-
81456-80-6
Diphenyl(trichloracetyl)phosphan

-
A
-
1079-66-9
chloro-diphenylphosphine

-
B
-
1017-89-6
diphenyltrichlorophosphorane

| Conditions | Yield |
|---|---|
| at 80℃; Product distribution; in dependence on temperature, time; |
-
-
4559-70-0
Diphenylphosphine oxide

-
-
1499-21-4
Diphenylphosphinic chloride

-
A
-
1707-03-5
diphenyl-phosphinic acid

-
B
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| Heating; | |
| Heating; | A 0.209 g B n/a |


-
A
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| at -80 - -20℃; |
-
-
756-04-7
2,2,3,3-tetrafluoropropionaldehyde

-
-
1079-66-9
chloro-diphenylphosphine

-
-
97108-40-2
2-chloro-2,2-diphenyl-3,5-bis(1,1,2,2-tetrafluoroethyl)-1,4,2-dioxaphospholane

| Conditions | Yield |
|---|---|
| In diethyl ether at 0℃; for 1h; | 100% |
-
-
2648-47-7
2,2,3,3,4,4,5,5-octafluorovaleraldehyde

-
-
1079-66-9
chloro-diphenylphosphine

-
-
97108-41-3
2-chloro-3,5-bis(1,1,2,2,3,3,4,4-octafluorobutyl)-2,2-diphenyl-1,4,2-dioxaphospholane

| Conditions | Yield |
|---|---|
| for 1h; | 100% |
-
-
1079-66-9
chloro-diphenylphosphine

-
-
18156-74-6
1-(Trimethylsilyl)imidazole

-
A
-
75-77-4
chloro-trimethyl-silane

-
B
-
54877-59-7
1-(di-phenylphosphino)imidazole

| Conditions | Yield |
|---|---|
| In dichloromethane for 1h; Ambient temperature; | A n/a B 100% |

-
-
1079-66-9
chloro-diphenylphosphine

-
-
55249-23-5
diphenylselenophosphinic chloride

| Conditions | Yield |
|---|---|
| With selenium In tetrahydrofuran at 120℃; for 3h; | 100% |
| With selenium In toluene at 120℃; for 3h; | 100% |
| With selenium | |
| With selenium at 120℃; for 6h; |

| Conditions | Yield |
|---|---|
| With bis(trimethylsilyl) telluride In acetonitrile Inert atmosphere; | 100% |
| With lithium In tetrahydrofuran | 90% |
| With 1,1',3,3'-tetraethyl-2,2'-biimidazolidinylidene In benzene at 0℃; for 2h; | 88% |
-
-
1079-66-9
chloro-diphenylphosphine

-
-
23356-96-9
(S)-1-Pyrrolidin-2-yl-methanol

-
-
83728-79-4
(S)-(N-diphenylphosphino)(2-diphenylphosphinoxymethyl)pyrrolidine

| Conditions | Yield |
|---|---|
| With triethylamine In toluene Ambient temperature; | 100% |
| With triethylamine In toluene at 0℃; for 16h; Yield given; | |
| With triethylamine In toluene |

| Conditions | Yield |
|---|---|
| 100% |
-
-
67175-80-8
1-methoxy-4-(2-hydroxyethyl)-cyclohexa-1,4-diene

-
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 25℃; for 13h; Condensation; | 100% |
-
-
1079-66-9
chloro-diphenylphosphine

-
-
347143-19-5
bis[(diphenylphosphino)(2-pyridyl)amino]dimethylsilane

-
-
125291-85-2
N-(diphenylphosphino)-P,P-diphenyl-N-2-pyridinylphosphinous amide

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 0℃; for 2h; | 100% |
-
-
1079-66-9
chloro-diphenylphosphine

-
-
366491-14-7
1-n-butyl-3-methyl-2,3-dihydro-imidazol-2-ylidene

| Conditions | Yield |
|---|---|
| In hexane; dichloromethane at -78℃; for 1h; | 100% |
-
-
1202-34-2
di(pyridin-2-yl)amine

-
-
1079-66-9
chloro-diphenylphosphine

-
-
472959-76-5
(bis(2-pyridyl)amino)diphenylphosphane

| Conditions | Yield |
|---|---|
| With n-butyllithium In diethyl ether; hexane at 20℃; for 48h; | 100% |

| Conditions | Yield |
|---|---|
| With dmap In diethyl ether at 20℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With dmap In diethyl ether at 20℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With dmap In diethyl ether at 20℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With dmap In diethyl ether at 20℃; for 0.5h; | 100% |
-
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| With dmap In diethyl ether at 20℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With dmap In diethyl ether at 20℃; for 0.5h; | 100% |
-
-
60-12-8
2-phenylethanol

-
-
1079-66-9
chloro-diphenylphosphine

-
-
849604-79-1
2-phenylethyl diphenylphosphinite

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 1h; | 100% |
| With n-butyllithium In tetrahydrofuran at 0℃; for 1h; | |
| With triethylamine In dichloromethane at 20℃; for 2h; Inert atmosphere; | |
| Stage #1: 2-phenylethanol With n-butyllithium In tetrahydrofuran at 0℃; for 1h; Inert atmosphere; Schlenk technique; Stage #2: chloro-diphenylphosphine In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; Schlenk technique; |
-
-
6928-85-4
N-amino-N'-methylpiperazine

-
-
1079-66-9
chloro-diphenylphosphine

-
-
849371-46-6
1-diphenylphosphinoamino-4-methylpiperazine

| Conditions | Yield |
|---|---|
| With triethylamine In diethyl ether at 20℃; for 1h; | 100% |
| With triethylamine In tetrahydrofuran for 5h; | 85% |
-
-
2213-43-6
1-Aminopiperidine

-
-
1079-66-9
chloro-diphenylphosphine

-
-
849371-47-7
1-diphenylphosphinoaminopiperidine

| Conditions | Yield |
|---|---|
| With triethylamine In diethyl ether at -78 - 20℃; for 1.5h; | 100% |
-
-
103-05-9
4-phenyl-2-methyl-2-butanol

-
-
1079-66-9
chloro-diphenylphosphine

-
-
820961-79-3
1,1-dimethyl-3-phenylpropyl diphenylphosphinite

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 100% |
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 98% |
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 95% |
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 91% |
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 100% |
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 100% |
-
-
2344-70-9
1-phenyl-3-butanol

-
-
1079-66-9
chloro-diphenylphosphine

-
-
820961-78-2
4-phenylbutan-2-yl diphenylphosphinite

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 100% |
| With n-butyllithium In tetrahydrofuran at 0℃; for 1h; | |
| With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 1h; | |
| With triethylamine In chloroform-d1 at 20℃; for 2h; Reagent/catalyst; Concentration; Inert atmosphere; | 86 %Spectr. |

| Conditions | Yield |
|---|---|
| With dmap In tetrahydrofuran at 0℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With dmap In tetrahydrofuran at 0℃; for 1h; | 100% |
-
-
114079-44-6
1-O-(tert-butyl)diphenylsilyl-1,3-butanediol

-
-
1079-66-9
chloro-diphenylphosphine

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 100% |
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 100% |

-
-
1079-66-9
chloro-diphenylphosphine

-
-
864848-06-6
(para-diphenylphosphanyl-phenyl)-(1-isopropyl-2-methyl-propyl)-amine

| Conditions | Yield |
|---|---|
| In diethyl ether; hexane at -78 - 20℃; for 18h; | 100% |
-
-
1079-66-9
chloro-diphenylphosphine

-
-
1730-25-2
allylmagnesium bromide

-
-
127686-75-3
allyl(diphenyl)phosphine-borane

| Conditions | Yield |
|---|---|
| Stage #1: chloro-diphenylphosphine With dimethylsulfide borane complex In diethyl ether at 0℃; for 2h; Stage #2: allylmagnesium bromide In diethyl ether at 0 - 20℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 100% |
Chlorodiphenylphosphine Specification
The Chlorodiphenylphosphine, with the CAS registry number 1079-66-9, is also known as Phosphine, chlorodiphenyl-. It belongs to the product categories of Pharmaceutical Intermediates; Chlorophosphines; Phosphines; Ligand; Organophosphine halide. Its EINECS number is 214-093-2. This chemical's molecular formula is C12H10ClP and molecular weight is 220.64. What's more, its systematic name is Diphenylphosphinous chloride. It is a colourless oily liquid with a pungent odor that is often described as being garlic-like and detectable even in the ppb range. It is useful reagent for introducing the Ph2P group into molecules, which includes many ligands. This product is used in the synthesis of various phosphines. This chemical is stable at common pressure and temperature, and it should be sealed and stored in containers with dry inert gas which are placed in a cool and dry place. Moreover, it should be protected from oxides, water, alcohol and reactive metal.
Physical properties of Chlorodiphenylphosphine are: (1)ACD/LogP: 3.601; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 3.60; (4)ACD/LogD (pH 7.4): 3.60; (5)ACD/BCF (pH 5.5): 321.32; (6)ACD/BCF (pH 7.4): 321.32; (7)ACD/KOC (pH 5.5): 2168.05; (8)ACD/KOC (pH 7.4): 2168.05; (9)#H bond acceptors: 0; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 13.59 Å2; (13)Flash Point: 147.33 °C; (14)Enthalpy of Vaporization: 53.928 kJ/mol; (15)Boiling Point: 319.998 °C at 760 mmHg ; (16)Vapour Pressure: 0.001 mmHg at 25°C.
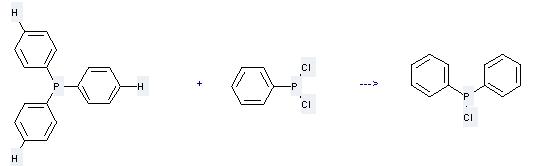
Preparation of Chlorodiphenylphosphine: this chemical can be prepared by diphenylphosphinous acid chloride at the temperature of 350 °C. This reaction time is 5 hours. The yield is about 85%.
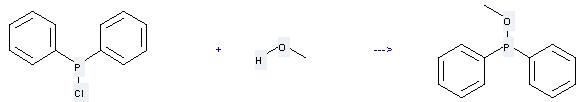
Uses of Chlorodiphenylphosphine: it can be used to produce diphenylphosphinous acid methyl ester. It will need reagent triethylamine and solvent diethyl ether with the reaction time of 3 hours. The yield is about 82%.
When you are using this chemical, please be cautious about it as the following:
This chemical can cause buins, and it will react violently with water. When contact with water, it will liberate toxic gas. You must keep the container dry and never add water to this product. This product is irritating to eyes. You should avoid contact with eyes. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection. You should take off immediately all contaminated clothing. In case of accident or if you feel unwell, you need to seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: ClP(c1ccccc1)c2ccccc2
(2)Std. InChI: InChI=1S/C12H10ClP/c13-14(11-7-3-1-4-8-11)12-9-5-2-6-10-12/h1-10H
(3)Std. InChIKey: XGRJZXREYAXTGV-UHFFFAOYSA-N
Related Products
- Chlorodiphenylphosphine
- 107-97-1
- 107971-01-7
- 107978-77-8
- 107979-42-0
- 107-98-2
- 107983-78-8
- 107990-50-1
- 107991-51-5
- 107-99-3
- 1079950-08-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View