-
Name
Cyclooctanone
- EINECS 207-940-2
- CAS No. 502-49-8
- Article Data386
- CAS DataBase
- Density 0.912 g/cm3
- Solubility Soluble in water at 20°C 15g/L. Soluble in acetone, alcohol, chloroform, methanol and benzene.
- Melting Point 32-41 °C(lit.)
- Formula C8H14O
- Boiling Point 193.229 °C at 760 mmHg
- Molecular Weight 126.199
- Flash Point 72.778 °C
- Transport Information
- Appearance colorless to white crystalline low melting solid
- Safety 45-36/37/39-26
- Risk Codes 34
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms NSC 9475;cyclooctan-1-one;
- PSA 17.07000
- LogP 2.29980
Synthetic route

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; tetra(n-butyl)ammonium hydrogensulfate; sodium tungstate In tert-butyl alcohol at 90℃; for 0.5h; | 100% |
| With ruthenium trichloride; iodobenzene; potassium peroxomonosulfate In water; acetonitrile at 20℃; for 0.5h; | 100% |
| With ruthenium trichloride; iodobenzene; potassium peroxymonosulfate In water; acetonitrile at 20℃; for 0.5h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; FeH6Mo6O24(3-)*3H3N*3H(1+)*7H2O; tetrabutylammomium bromide; dihydrogen peroxide In 1,4-dioxane; water at 85℃; for 24h; | 98% |
| With oxygen; isobutyraldehyde; vanadium-substituted V-MCM-41 (A) zeolite In acetone at 25℃; for 24h; | 83% |
| With potassium permanganate; borontrifluoride acetic acid In acetonitrile at 23℃; for 0.166667h; | 55% |

| Conditions | Yield |
|---|---|
| With lithium iodide In xylene at 140℃; for 6h; | 98% |
| With lithium iodide In xylene at 140℃; for 6h; Product distribution; other temperatures, other times; various concentrations of catalyst; other catalyst; without a solvent; |

| Conditions | Yield |
|---|---|
| With zinc dichromate trihydrate at 20℃; grinding; neat (no solvent); chemoselective reaction; | 98% |
| With 3-carboxypyridinium dichromate In acetonitrile at 20℃; for 0.25h; | 97% |
| With potassium permanganate; copper(II) sulfate In dichloromethane for 24h; Heating; | 90% |
-
-
13213-32-6
cyclooctyl methyl ether

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| Stage #1: cyclooctyl methyl ether With bromine In dichloromethane; water for 1h; Reflux; Stage #2: With dihydrogen peroxide In dichloromethane; water for 4h; Reflux; | 97% |

| Conditions | Yield |
|---|---|
| With tetrachloromethane; copper(II) choride dihydrate; chromium(III) acetylacetonate; water at 150℃; for 12h; Sealed tube; | A 4% B 96% |

| Conditions | Yield |
|---|---|
| With sodio-propane-1,2-diol in anhydrous propane-1,2-diol at 80 - 85℃; for 0.5h; | 95% |
-
-
40338-24-7
cyclooctanone semicarbazone

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; Tonsil In ethyl acetate for 2h; Heating; | 94.1% |

| Conditions | Yield |
|---|---|
| With [(salph)Cr(THF)2][Co(CO)4] In 1,2-dimethoxyethane at 60℃; under 5171.48 Torr; for 24h; | A 3% B 94% |

-
-
25632-03-5
1,1-dimethoxycyclooctane

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With dimethylboron bromide In dichloromethane; 1,2-dichloro-ethane at -78℃; for 1h; | 93% |
| With dimethylboron bromide; sodium hydrogencarbonate 1) CH2Cl2, -78 deg C, 1h, 2) THF, 5 min; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With dimethylboron bromide In dichloromethane; 1,2-dichloro-ethane at -78℃; for 1h; | 92% |
| With Montmorillonite K 10; water In acetone for 0.5h; Heating; | 87% |
| indium(III) chloride In methanol; water for 1.5h; Heating; | 85% |
| With dimethylboron bromide; sodium hydrogencarbonate 1) CH2Cl2, -78 deg C, 1h, 2) THF, 5 min; Yield given. Multistep reaction; |
-
-
27889-58-3
3,3-dimethyl-1,5-dioxaspiro<5.7>tridecane

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With dimethylboron bromide In dichloromethane; 1,2-dichloro-ethane at -78℃; for 1h; | 91% |
| With dimethylboron bromide; sodium hydrogencarbonate 1) CH2Cl2, -78 deg C, 1h, 2) THF, 5 min; Yield given. Multistep reaction; |
-
-
39261-18-2
2-bromocyclooctan-1-one

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With diphosphorus tetraiodide In dichloromethane at 25℃; for 7h; | 91% |
-
-
931-88-4, 931-87-3
(Z)-Cyclooctene

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; C21H19N5Pd(2+)*2BF4(1-) In decane; acetonitrile at 70℃; for 24h; Temperature; Wacker Oxidation; | 90% |
| With dihydrogen peroxide; tetraphenylphosphonium; sodium hydrogencarbonate; WO(O2)(QO)4 In acetonitrile at 25℃; for 0.5h; | 74% |
| Stage #1: cis-Cyclooctene With dimethylsulfide borane complex In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; Stage #2: With tetrapropylammonium perruthennate; 4-methylmorpholine N-oxide In tetrahydrofuran; dichloromethane at 20℃; Molecular sieve; | 53% |
-
-
85895-35-8
1,1-bis(phenylthio)cyclooctane

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid; trifluoroacetic acid In dichloromethane for 1h; Ambient temperature; | 87% |

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In toluene at 0℃; for 2h; | 87% |
| t-BuOSmI2 In tetrahydrofuran at 60℃; for 4h; | 83% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 25℃; for 2.1h; Product distribution; Mechanism; var. primary and secondary alcohols, var. 12-I-5 species; other solvents and relation time, var. concentrations of oxidant, in the presence of different labile groups in the molecule; | 86% |
-
-
40756-11-4
(cyclooctyloxy)trimethylsilane

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With sodium bromate; ammonium chloride In water; acetonitrile at 80℃; for 0.833333h; | 82% |

| Conditions | Yield |
|---|---|
| With oxygen; magnesium oxide supported polytitazane-tin tetrachloride In 1,4-dioxane at 80℃; for 20h; | A n/a B 81% |

| Conditions | Yield |
|---|---|
| With CoO40W12(5-)*3C30H24CoN6*9.5H2O*2H(1+); dihydrogen peroxide In acetonitrile at 80℃; for 9h; Inert atmosphere; | A 80% B 19% |
| With [Fe4III(μ-O)2(μ-acetate)6(2,2'-bipyridine)2(H2O)2](NO3-)(OH-); dihydrogen peroxide; acetic acid In water; acetonitrile at 32℃; for 3h; Catalytic behavior; Overall yield = 89 %Spectr.; | A 77% B 13% |
| With oxygen; copper diacetate; acetaldehyde In dichloromethane; acetonitrile at 70℃; under 760.051 Torr; for 24h; Catalytic behavior; Inert atmosphere; | A 74% B 8% |
-
-
53867-57-5
1,2-dicyclooctylidenehydrazine

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With HOF* CH3CN In dichloromethane at 0℃; for 0.0166667h; | 80% |

| Conditions | Yield |
|---|---|
| With hydrogen In 1,2-dichloro-benzene | 80% |

| Conditions | Yield |
|---|---|
| With HO40PVW11(14-)*4C16H36N(1+); dihydrogen peroxide In acetonitrile at 80℃; for 12h; Inert atmosphere; | A 23% B 77% |
| With 5,10,15,20-tetrakis(4-chlorophenyl)porphyrinatocopper(II); T(o-Cl)PPCo; oxygen at 110℃; under 10501.1 Torr; for 8h; |

| Conditions | Yield |
|---|---|
| With formaldehyd; water; acetone; Amberlyst 15 at 80℃; for 10h; | 76% |
-
-
1502-14-3, 74305-06-9, 84402-56-2
(+/-)-trans-2-bromo-1-hydroxycyclooctane

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Ambient temperature; Irradiation; | 76% |
| With di-tert-butyl diperoxyoxalate In cyclohexane Heating; | 93 % Chromat. |

| Conditions | Yield |
|---|---|
| 74% | |
| In not given IR, PMR, mass spectral data, GLC;; | 74% |
-
-
76902-54-0
(Pyrrolidinylmethylene)cyclooctane

-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With potassium dichromate; sulfuric acid In diethyl ether at 25℃; for 1h; | 73% |

| Conditions | Yield |
|---|---|
| at 600℃; | A 71% B 14% |
| With aluminum oxide; lithium bromide at 180℃; for 0.5h; Yield given. Yields of byproduct given; |
-
-
50338-42-6
(E)-1-trimethylsilyloxy-1-cyclooctene

-
A
-
502-49-8
cycloactanone

-
B
-
496-82-2
2-hydroxycylooctanone

| Conditions | Yield |
|---|---|
| With tris(cetylpyridinium) 12-tungstophosphate; dihydrogen peroxide In dichloromethane for 6h; Heating; | A 14% B 71% |
| With tris(cetylpyridinium) 12-tungstophosphate; dihydrogen peroxide In dichloromethane at 20℃; for 16h; oxidative cleavage; | A 20 % Chromat. B 45 % Chromat. |

| Conditions | Yield |
|---|---|
| With pyridine; hydroxylamine hydrochloride In ethanol at 90 - 100℃; for 2h; | 100% |
| With ammonium chloride; Amberlyst A-21 In ethanol for 1h; Ambient temperature; | 95% |
| With hydroxylamine hydrochloride; sodium acetate In ethanol; water Reflux; | 90% |

| Conditions | Yield |
|---|---|
| With aluminum oxide; sodium tetrahydroborate In hexane at 30℃; for 3h; | 100% |
| With hydrogen In ethanol at 130℃; under 30003 Torr; for 20h; Autoclave; | 97% |
| With chloro-trimethyl-silane; calcium hydride; zinc(II) chloride In tetrahydrofuran at 40℃; for 0.5h; | 92% |

| Conditions | Yield |
|---|---|
| Stage #1: cycloactanone With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: With methyl chlorosulfate In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; | 100% |
| With ammonium cerium(IV) nitrate at 25 - 28℃; for 5.5h; | 86% |
| With p-toluenesulfonyl chloride; lithium diisopropyl amide In tetrahydrofuran -78 deg C to room t., 1 h; | 69% |

| Conditions | Yield |
|---|---|
| With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78 - 20℃; | 100% |
| Stage #1: cycloactanone With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 1h; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran at -78℃; for 0.666667h; | 62% |
| With sodium amide | |
| With lithium diisopropyl amide 1.) THF, -78 deg C, 30 min, 2.) THF, RT, 40 h; Yield given. Multistep reaction; | |
| Stage #1: cycloactanone With lithium diisopropyl amide In tetrahydrofuran; hexane at 0℃; for 0.5h; Stage #2: methyl iodide In tetrahydrofuran; hexane at -78℃; Further stages.; |
-
-
502-49-8
cycloactanone

-
-
50635-71-7, 50635-72-8, 31350-93-3
1-chloroethyl p-tolyl sulfoxide

-
-
159763-14-1
1-<1-chloro-1-(p-tolylsulfinyl)ethyl>-1-cyclooctanol

| Conditions | Yield |
|---|---|
| at -70℃; | 100% |
| With n-butyllithium; diisopropylamine In tetrahydrofuran at -65℃; for 0.416667h; | 37% |
-
-
502-49-8
cycloactanone

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
2567-85-3
cyclooctanone p-tolylsulfonylhydrazone

| Conditions | Yield |
|---|---|
| In ethanol at 100℃; for 1h; | 100% |
| In ethanol at 100℃; for 1.66667h; Inert atmosphere; | 100% |
| In methanol at 20℃; Schlenk technique; | 100% |
-
-
502-49-8
cycloactanone

-
-
616-38-6
carbonic acid dimethyl ester

-
-
5452-73-3
methyl 2-oxocyclooctanecarboxylate

| Conditions | Yield |
|---|---|
| With sodium hydride In 1,4-dioxane at 90℃; Reflux; | 100% |
| Stage #1: carbonic acid dimethyl ester With sodium hydride In tetrahydrofuran at 5 - 10℃; for 0.5h; Inert atmosphere; Stage #2: cycloactanone In tetrahydrofuran for 4.5h; Reflux; Inert atmosphere; | 88% |
| Stage #1: carbonic acid dimethyl ester With sodium hydride In tetrahydrofuran at 5 - 10℃; for 0.5h; Stage #2: cycloactanone In tetrahydrofuran for 4.5h; Reflux; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: cycloactanone With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at 0℃; Stage #2: acrolein In tetrahydrofuran; hexane at -78℃; Further stages.; | 100% |
| 98% | |
| Stage #1: cycloactanone With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at 0℃; for 1h; Stage #2: acrolein In tetrahydrofuran; hexane at -78℃; for 0.0166667h; | |
| Stage #1: cycloactanone With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 0.75h; Stage #2: acrolein In tetrahydrofuran; hexane at -78℃; |

| Conditions | Yield |
|---|---|
| Stage #1: Bromoform With lithium diisopropyl amide In tetrahydrofuran; hexane at -100℃; for 0.166667h; Metallation; Stage #2: cycloactanone With boron trifluoride diethyl etherate In tetrahydrofuran; diethyl ether; hexane at -90℃; for 4h; Addition; | 100% |
-
-
502-49-8
cycloactanone

-
-
870-46-2
t-butoxycarbonylhydrazine

-
-
339058-79-6
N'-cyclooctylidene-hydrazinecarboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 100% |
-
-
502-49-8
cycloactanone

-
-
124476-10-4
3-amino-4-cyano-5-methylsulfanyl-1H-pyrrole-2-carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With aluminium trichloride In 1,2-dichloro-ethane at 115 - 120℃; for 2h; Friedlaender cyclization reaction; | 100% |
-
-
502-49-8
cycloactanone

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate at 60℃; for 0.5h; Ball milling; neat (no solvent); regiospecific reaction; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; orthoformic acid triethyl ester In 1,4-dioxane; N,N-dimethyl-formamide at 20℃; for 4h; | 100% |
-
-
502-49-8
cycloactanone

-
-
4640-67-9
3-(4-fluorophenyl)-3-oxopropionitrile

-
-
132812-72-7
4-(4-fluorophenyl)-5,6,7,8,9,10-hexahydrocyclooctane[b]pyridine-(1H)-ketone

| Conditions | Yield |
|---|---|
| With 1-ethyl-3-methylimidazolium tetrafluoroborate at 120℃; for 1h; | 99.8% |
| With methanesulfonic acid; phosphorus pentoxide at 50℃; for 15h; | 95% |
| With PPA Heating; | 64% |
| With phosphoric acid; toluene-4-sulfonic acid In toluene Reflux; Dean-Stark; Large scale; | 54% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
502-49-8
cycloactanone

-
-
50338-42-6
(cyclooct-1-enyloxy)-trimethylsilane

| Conditions | Yield |
|---|---|
| With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; Inert atmosphere; | 99% |
| With triethylamine In N,N-dimethyl-formamide for 15h; Reflux; | 93% |
| With triethylamine In N,N-dimethyl-formamide |
-
-
502-49-8
cycloactanone

-
-
7677-24-9
trimethylsilyl cyanide

-
-
50361-50-7
1-((trimethylsilyl)oxy)cyclooctanecarbonitrile

| Conditions | Yield |
|---|---|
| With C29H46LaN3Si2 at 15℃; for 1h; Inert atmosphere; Glovebox; Schlenk technique; | 99% |
| With n-butyllithium In tetrahydrofuran; hexane for 2h; Ambient temperature; | 96.7% |
| potassium-exchanged zirconium hydrogen phosphate In dichloromethane for 20h; Heating; | 95% |
-
-
502-49-8
cycloactanone

-
-
30354-18-8
N,N-dimethyl(methylene)ammonium chloride

-
-
28118-62-9
2-(Dimethylamino)methylcyclooctan-1-one

| Conditions | Yield |
|---|---|
| In acetonitrile for 72h; Ambient temperature; | 99% |
-
-
502-49-8
cycloactanone

-
-
502-42-1
cycloheptanone

-
A
-
187819-58-5
α-(α'-cycloheptenyl)-β-hydroxycarbonyl-ethyl propionate

-
B
-
32405-96-2
2,4,5,6,7,8-hexahydroazulene

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water; N,N-dimethyl-formamide | A 99% B n/a |

-
-
502-49-8
cycloactanone

-
-
50361-50-7
1-((trimethylsilyl)oxy)cyclooctanecarbonitrile

| Conditions | Yield |
|---|---|
| n-butyllithium In tetrahydrofuran | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: potassium cyanide; cycloactanone In ethanol; water at 60℃; for 11h; Inert atmosphere; Stage #2: With hydrogenchloride In ethanol; water at 0℃; for 15h; | 99% |
| With ammonium carbonate In methanol; water at 140℃; for 0.133333h; Sealed vial; Microwave irradiation; | 85% |
-
-
502-49-8
cycloactanone

-
-
1066-54-2
trimethylsilylacetylene

-
-
1403824-58-7
1-((trimethylsilyl)ethynyl)cyclooctanol

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: cycloactanone In tetrahydrofuran at -78 - 20℃; Inert atmosphere; | 99% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 0.5h; Inert atmosphere; Stage #2: cycloactanone In tetrahydrofuran; hexane at 0 - 25℃; for 2h; Inert atmosphere; | |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: cycloactanone In tetrahydrofuran; hexane at 20℃; for 2h; |

| Conditions | Yield |
|---|---|
| With oxygen; trifluoroacetic acid; sodium nitrite at 0 - 20℃; for 5.25h; Product distribution / selectivity; | 98% |
| With dihydrogen peroxide In water; acetonitrile at 90℃; for 11h; Green chemistry; | 92% |
| With dihydrogen peroxide; ortho-tungstic acid In water at 90℃; for 20h; | 85% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium nitrite In tetrahydrofuran at 20℃; for 0.2h; | 98% |
| With perchloric acid; tripropylammonium fluorochromate (VI) In water; acetic acid at 24.9℃; Rate constant; Thermodynamic data; ΔH(activ.), ΔS(activ.), ΔG(activ.); further temperatures (308-328 K); | |
| With selenium(IV) oxide In ethanol |

| Conditions | Yield |
|---|---|
| Stage #1: furan With n-butyllithium; N,N,N,N,-tetramethylethylenediamine In tetrahydrofuran; hexanes at -78℃; for 1h; Inert atmosphere; Stage #2: cycloactanone In tetrahydrofuran; hexanes at -78℃; for 1h; Inert atmosphere; Stage #3: With ammonium chloride In tetrahydrofuran; hexanes; water | 98% |
| With n-butyllithium; toluene-4-sulfonic acid Multistep reaction; | |
| Stage #1: furan With n-butyllithium In diethyl ether; hexane at 0℃; Stage #2: cycloactanone In diethyl ether; hexane at 0 - 20℃; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 60℃; for 3h; | 98% |

| Conditions | Yield |
|---|---|
| With ethanol; sodium In diethyl ether | 98% |
| With ethanol; sodium In diethyl ether at 20℃; for 48h; Inert atmosphere; |
Cyclooctanone Consensus Reports
Reported in EPA TSCA Inventory.
Cyclooctanone Specification
The Cyclooctanone, with the CAS registry number 502-49-8 and EINECS registry number 207-940-2, is also known as cyclooctan-1-one. It is a kind of colorless to white crystalline low melting solid, and belongs to the following product categories: C7 to C8; Carbonyl Compounds; Ketones. And the molecular formula of this chemical is C8H14O. What's more, it is usually used as intermediate in the organic and pharmaceutical synthesis, and it is also used to produce Blonanserin-a kind of anti-schizophrenic drugs.
The physical properties of Cyclooctanone are as following: (1)ACD/LogP: 1.88; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.875; (4)ACD/LogD (pH 7.4): 1.875; (5)ACD/BCF (pH 5.5): 15.659; (6)ACD/BCF (pH 7.4): 15.659; (7)ACD/KOC (pH 5.5): 249.367; (8)ACD/KOC (pH 7.4): 249.367; (9)#H bond acceptors: 1; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 17.07 ?2; (13)Index of Refraction: 1.448; (14)Molar Refractivity: 37.024 cm3; (15)Molar Volume: 138.428 cm3; (16)Polarizability: 14.678 10-24cm3; (17)Surface Tension: 30.761 dyne/cm; (18)Density: 0.912 g/cm3; (19)Flash Point: 72.778 °C; (20)Enthalpy of Vaporization: 42.943 kJ/mol; (21)Boiling Point: 193.229 °C at 760 mmHg; (22)Vapour Pressure: 0.47 mmHg at 25°C.
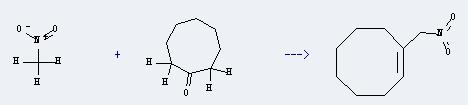
Uses of Cyclooctanone: It can react with nitromethane to produce 1-nitromethyl-cis-cyclooctene. This reaction will need reagent N,N-dimethylethylenediamine, and the solvent benzene. The reaction time is 14 hours with heating, and the yield is about 83%.
You should be cautious while dealing with this chemical. It may cause burns. Therefore, you had better take the following instructions: Wear suitable protective clothing, gloves and eye/face protection, and in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice; In case of accident or if you feel unwell, seek medical advice immediately (show label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: C1CCCC(=O)CCC1
(2)InChI: InChI=1/C8H14O/c9-8-6-4-2-1-3-5-7-8/h1-7H2
(3)InChIKey: IIRFCWANHMSDCG-UHFFFAOYAI
Related Products
- Cyclooctanone
- Cyclooctanone,5-hydroxy-
- 502-50-1
- 502509-05-9
- 502-52-3
- 502546-41-0
- 502-55-6
- 502-56-7
- 50257-39-1
- 50257-40-4
- 5025-82-1
- 502-61-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View