-
Name
Dibutyl maleate
- EINECS 203-328-4
- CAS No. 105-76-0
- Article Data27
- CAS DataBase
- Density 1.004 g/cm3
- Solubility Insoluble in water
- Melting Point -85 °C
- Formula C12H20O4
- Boiling Point 280 °C at 760 mmHg
- Molecular Weight 228.288
- Flash Point 136.4 °C
- Transport Information
- Appearance clear colourless to slightly yellowish liquid
- Safety 26-36-61-37-29-24
- Risk Codes 36/37/38-51/53-43
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, N
N
- Synonyms Dibutyl maleate;DBM
- PSA 52.60000
- LogP 2.22920
Synthetic route

| Conditions | Yield |
|---|---|
| With diacidic ionic liquid supported on magnetic-silica nanoparticles In neat (no solvent) at 118℃; for 3h; Dean-Stark; | 100% |
| With 3,3′-(2,2-bis(hydroxymethyl)propane-1,3-diyl)bis(1-methyl-1H-imidazol-3-ium) hydrogen sulfate for 4h; Dean-Stark; Reflux; | 98% |
| With N-(4-sulphonic acid)butylpyridinium hydrogen sulphate at 120℃; for 1h; Catalytic behavior; Reagent/catalyst; Microwave irradiation; | 84.38% |
-
-
592-84-7
n-butyl formate

-
-
110-16-7
maleic acid

-
A
-
105-76-0
Dibutyl maleate

-
B
-
925-21-3
mon-n-butyl maleate

| Conditions | Yield |
|---|---|
| With Dowex 50Wx2 In octane at 100℃; for 1.66667h; Esterification; | A 6% B 89% |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 124.85℃; for 2h; | 80% |


| Conditions | Yield |
|---|---|
| With per-rhenic acid In water at 170℃; for 3h; Inert atmosphere; | 75% |

| Conditions | Yield |
|---|---|
| tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In dichloromethane at 20℃; | A 74% B n/a |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 80℃; for 16h; | 60% |
| With sulfuric acid; benzene beim Entfernen des gebildeten Wassers; | |
| With sulfuric acid; toluene beim Entfernen des gebildeten Wassers; | |
| With dodecatungstosilic acid at 95℃; for 5h; Reagent/catalyst; Flow reactor; | |
| With Amberlyst-15 at 80℃; for 4h; Kinetics; Reagent/catalyst; Temperature; Molecular sieve; |

| Conditions | Yield |
|---|---|
| at 80℃; for 6.25h; | 96.5% |


| Conditions | Yield |
|---|---|
| With Benzoylformic acid for 72h; Irradiation; Green chemistry; | 96% |
-
-
75-15-0
carbon disulfide

-
-
105-76-0
Dibutyl maleate

-
-
110-73-6
2-(Ethylamino)ethanol

-
-
885700-65-2
C17H31NO5S2

| Conditions | Yield |
|---|---|
| at 70℃; for 6.66667h; | 94% |


| Conditions | Yield |
|---|---|
| With water; 5-methoxy-1,3,4-triphenyl-4,5-dihydro-1H-1,2-4-triazoline In 1,2-dimethoxyethane at 150℃; for 2h; Inert atmosphere; Microwave irradiation; | 94% |

| Conditions | Yield |
|---|---|
| With sodium hydrogensulfite In ethanol at 119.85℃; | 90.4% |
| With sodium hydrogensulfite In water at 99.85℃; sulfonation; | |
| With sodium hydrogensulfite In ethanol Heating; | |
| With sodium hydrogensulfite In water at 99.84℃; |
-
-
105-76-0
Dibutyl maleate

-
-
301677-76-9
3,4-Dibromo-2,5-dimethylthiophene S-monoxide

-
-
1252031-08-5
dibutyl 2,3-dibromo-1,4-dimethyl-7-thiabicyclo[2.2.1]hept-2-ene-5,6-dicarboxylate 7-oxide

| Conditions | Yield |
|---|---|
| In chloroform for 24h; Reflux; | 89% |
-
-
105-76-0
Dibutyl maleate

-
-
6637-53-2, 144252-21-1, 144252-22-2
3-hydroxy-3-phenyl-2,3-dihydro-isoindol-1-one

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; acetic acid In 1,2-dichloro-ethane at 120℃; for 20h; Sealed tube; | 86% |

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; (p-cymene)ruthenium(II) chloride; copper(II) acetate monohydrate; acetic acid In dichloromethane at 120℃; for 24h; Inert atmosphere; regioselective reaction; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: butan-1-ol With sodium at 80℃; Stage #2: (3-chloro-2-pyridyl)hydrazine With iodotris(triphenylphosphine)silver(I) In butan-1-ol at 30℃; Stage #3: Dibutyl maleate In butan-1-ol at 28 - 32℃; Reagent/catalyst; | 80% |

-
-
105-76-0
Dibutyl maleate

-
-
1436417-01-4
methyl 1-benzoyl-1H-indole-5-carboxylate

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; (p-cymene)ruthenium(II) chloride; copper(II) acetate monohydrate; acetic acid In dichloromethane at 120℃; for 24h; Inert atmosphere; regioselective reaction; | 78% |
-
-
105-76-0
Dibutyl maleate

-
-
1436590-99-6
N-benzoyl-6-chloroindole

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; (p-cymene)ruthenium(II) chloride; copper(II) acetate monohydrate; acetic acid In dichloromethane at 120℃; for 24h; Inert atmosphere; regioselective reaction; | 77% |
-
-
14729-20-5
tetracarbonyl(1,10-phenanthroline)tungsten(0)

-
-
105-76-0
Dibutyl maleate

-
-
225373-58-0, 396727-60-9, 396718-44-8
W(CO)2(C12H8N2)(C12H20O4)2

| Conditions | Yield |
|---|---|
| In toluene (N2(; reflux (19 h); concn., layering with light petroleum (7-10 d), filtration, washing, drying (vac.); elem. anal.; | 75% |
-
-
105-76-0
Dibutyl maleate

-
-
1127562-54-2
(5-bromoindol-1-yl)phenylmethanone

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; (p-cymene)ruthenium(II) chloride; copper(II) acetate monohydrate; acetic acid In dichloromethane at 120℃; for 24h; Inert atmosphere; regioselective reaction; | 75% |

| Conditions | Yield |
|---|---|
| Heating; | 75% |

-
-
15740-78-0
(1,10-phenanthroline)molybdenum tetracarbonyl

-
-
105-76-0
Dibutyl maleate

-
-
60342-24-7, 60384-25-0
Mo(CO)2(C12H8N2)(C12H20O4)2

| Conditions | Yield |
|---|---|
| In toluene (N2(; reflux (17 h); concn., layering with light petroleum (7-10 d), filtration, washing, drying (vac.); elem. anal.; | 72% |
-
-
105-76-0
Dibutyl maleate

-
-
135822-90-1
o-chloro-N-tosylbenzaldimine

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; lithium acetate In 1,2-dichloro-ethane at 80℃; for 16h; Sealed tube; diastereoselective reaction; | 72% |
-
-
105-76-0
Dibutyl maleate

-
-
135822-90-1
o-chloro-N-tosylbenzaldimine

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; lithium acetate In 1,2-dichloro-ethane at 80℃; for 16h; | 72% |
-
-
105-76-0
Dibutyl maleate

-
-
98217-62-0
(E)-1,2-bis(4-(trifluoromethyl)phenyl)diazene

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper diacetate In 1,2-dichloro-ethane at 130℃; for 12h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Sealed tube; chemoselective reaction; | 71% |
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper diacetate In 1,2-dichloro-ethane at 130℃; for 12h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; | 71% |

-
-
105-76-0
Dibutyl maleate

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper diacetate In 1,2-dichloro-ethane at 130℃; for 12h; Sealed tube; chemoselective reaction; | 70% |

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; (p-cymene)ruthenium(II) chloride; copper(II) acetate monohydrate; acetic acid In dichloromethane at 120℃; for 24h; Inert atmosphere; regioselective reaction; | 68% |

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper diacetate In 1,2-dichloro-ethane at 130℃; for 12h; Sealed tube; chemoselective reaction; | 67% |
-
-
105-76-0
Dibutyl maleate

-
-
100-52-7
benzaldehyde

-
-
77425-65-1, 77480-79-6, 91278-68-1
3,6-diphenyltetrahydrofuro[3,4-c]furan-1,4-dione

| Conditions | Yield |
|---|---|
| With samarium; copper(l) iodide; toluene-4-sulfonic acid; potassium iodide In tetrahydrofuran at 20℃; Molecular sieve; Inert atmosphere; diastereoselective reaction; | 65% |

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper diacetate In 1,2-dichloro-ethane at 130℃; for 12h; Sealed tube; chemoselective reaction; | 65% |
-
-
105-76-0
Dibutyl maleate

-
-
15426-14-9, 106131-20-8, 106131-24-2
3,3'-dichloroazobenzene

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper diacetate In 1,2-dichloro-ethane at 130℃; for 12h; Sealed tube; chemoselective reaction; | 64% |
-
-
16632-93-2
tetracarbonyl(4,7-diphenyl-1,10-phenanthroline-N,N')molybdenum(0)

-
-
105-76-0
Dibutyl maleate

-
-
390387-45-8
[molybdenum(CO)2(4,7-diphenyl-1,10-phenanthroline)(dibutylfumarate)2]

| Conditions | Yield |
|---|---|
| In toluene all manipulations under N2 atm.; org. compd. added to soln. of complex, refluxed for 21 h; concd., chromy., solvent removed, dried in vac., elem. anal.; | 63% |
Dibutyl maleate Consensus Reports
Reported in EPA TSCA Inventory.
Dibutyl maleate Specification
The CAS registry number of Dibutyl maleate is 105-76-0. Its EINECS registry number is 203-328-4. The systematic name is dibutyl but-2-enedioate. In addition, the molecular formula is C12H20O4 and the molecular weight is 228.28. What's more, it is a kind of clear colourless to slightly yellowish liquid and belongs to the classes of Plasticizer; Carbonyl Compounds; Esters. It can be used as raw material for synthetic resin, and used as organic synthesis intermediates.
Physical properties about this chemical are: (1)ACD/LogP: 3.81; (2)ACD/LogD (pH 5.5): 3.81; (3)ACD/LogD (pH 7.4): 3.81; (4)ACD/BCF (pH 5.5): 461.68; (5)ACD/BCF (pH 7.4): 461.68; (6)ACD/KOC (pH 5.5): 2810.22; (7)ACD/KOC (pH 7.4): 2810.22; (8)#H bond acceptors: 4; (9)#Freely Rotating Bonds: 10; (10)Polar Surface Area: 52.6 Å2; (11)Index of Refraction: 1.451; (12)Molar Refractivity: 61.25 cm3; (13)Molar Volume: 227.1 cm3; (14)Polarizability: 24.28 ×10-24cm3; (15)Surface Tension: 33.4 dyne/cm; (16)Density: 1.004 g/cm3; (17)Flash Point: 136.4 °C; (18)Enthalpy of Vaporization: 51.87 kJ/mol; (19)Boiling Point: 280 °C at 760 mmHg; (20)Vapour Pressure: 0.00388 mmHg at 25°C.
Preparation of Dibutyl maleate: it can be prepared by cisbutenedioic anhydride and butanol in the presence of sulfuric acid through esterification reaction. After a series of neutralization, washing, dehydroxylation, distillation and filtration you can get the desired product from the reaction product.
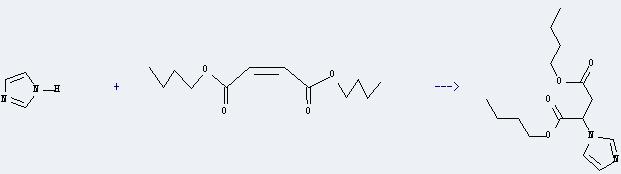
Uses of Dibutyl maleate: it is used as impregnants, dispersants, lubricants and plasticizers. And it can react with 1H-imidazole to get 2-imidazol-1-yl-succinic acid dibutyl ester. The reaction time is 7 hours at reaction temperature of 100 °C. The yield is about 50%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. It may cause sensitization by skin contact. In addition, it is very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. During using it, wear suitable protective clothing and gloves and avoid contact with skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. Moreover, you should avoid release to the environment and you can refer to special instructions safety data sheet. You can not empty it into drains.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(OCCCC)C=CC(=O)OCCCC
(2)InChI: InChI=1/C12H20O4/c1-3-5-9-15-11(13)7-8-12(14)16-10-6-4-2/h7-8H,3-6,9-10H2,1-2H3
(3)InChIKey: JBSLOWBPDRZSMB-UHFFFAOYAY
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 150mg/kg (150mg/kg) | National Technical Information Service. Vol. AD691-490, | |
| rabbit | LD50 | skin | 10gm/kg (10000mg/kg) | Raw Material Data Handbook, Vol.1: Organic Solvents, 1974. Vol. 2, Pg. 19, 1975. | |
| rat | LD50 | oral | 3700mg/kg (3700mg/kg) | Raw Material Data Handbook, Vol.1: Organic Solvents, 1974. Vol. 2, Pg. 19, 1975. |
Related Products
- Dibutyl 2,2-dithiobisbenzoate
- Dibutyl 2,2'-thiobisacetate
- Dibutyl isophthalate
- Dibutyl L-(+)-tartrate
- Dibutyl maleate
- Dibutyl oxalate
- DIBUTYL PHENYL PHOSPHATE
- Dibutyl phosphate
- Dibutyl phosphite
- Dibutyl phthalate
- 10576-12-2
- 105762-16-1
- 105763-77-7
- 1057677-92-5
- 105-77-1
- 105772-14-3
- 105774-05-8
- 105776-13-4
- 10578-06-0
- 10578-12-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View