-
Name
Dibutyl phosphate
- EINECS 203-509-8
- CAS No. 107-66-4
- Article Data44
- CAS DataBase
- Density 1.078 g/cm3
- Solubility insoluble in water
- Melting Point -13 °C
- Formula C8H19O4P
- Boiling Point 275.3 °C at 760 mmHg
- Molecular Weight 210.21
- Flash Point 120.3 °C
- Transport Information UN 3261
- Appearance clear liquid
- Safety 26-39-45-36/37/39-25
- Risk Codes 37/38-41-38-34
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi,  C
C
- Synonyms Butylphosphate, (BuO)2(HO)PO (6CI);DP 4;DP 4 (coupling agent);Di-n-Butylphosphate;Dibutyl acid phosphate;Dibutyl hydrogen phosphate;HDBP;NSC 203039;NSC 22313;
- PSA 65.57000
- LogP 2.72020
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water for 2.5h; Heating; | 97% |
| With sodium hydroxide | |
| With hydrogenchloride In hydrogenchloride byproducts: C4H9Cl; hydrolysis of TBP in concd. HCl;; hydrolytic decompn. of 18.5 % TBP in 6 days at ambient temp.;; | |
| With HCl In hydrogenchloride byproducts: C4H9Cl; hydrolysis of TBP in concd. HCl;; hydrolytic decompn. of 18.5 % TBP in 6 days at ambient temp.;; |
-
-
71-36-3
butan-1-ol

-
A
-
1623-15-0
n-butyl phosphoric acid

-
B
-
126-73-8
phosphoric acid tributyl ester

-
C
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| With phosphorus; tetraethylammonium iodide In water; acetonitrile electrolysis; | A n/a B 87% C n/a |


| Conditions | Yield |
|---|---|
| With sodium bromate; phosphan at 80℃; Kinetics; Condensation; | A 20% B 80% |
| With sodium hypophosphite; copper dichloride at 24.9℃; for 4h; | A 22.2% B 77.8% |
| With phosphorous; air; ruthenium hydroxide trichloride; copper dichloride at 80℃; oxidative alkoxylation; | A 33.7% B 11.3% |
-
-
38257-85-1
Di-n-butyl-(o-hydroxy-phenyl)-phosphat

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| With [bis(acetoxy)iodo]benzene; water In acetonitrile at 20℃; for 1h; oxidative cleavage; | 72% |

| Conditions | Yield |
|---|---|
| With oxygen; hypophosphorous acid; copper dichloride at 50℃; for 6h; | A 4% B 48% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In tetrahydrofuran; water at 0℃; | 46% |
| With water beim Erhitzen; | |
| With water In toluene for 1h; Reflux; | 29.5 g |
-
-
121252-83-3
N-prolyl>proline

-
A
-
121252-84-4
(R)-1-[(R)-1-(Dibutoxy-phosphoryl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carboxylic acid butyl ester

-
B
-
107-66-4
dibutyl phosphate

-
C
-
19943-27-2
rac-octahydrodipyrrolo[1,2-a:1',2'-d]pyrazine-5,10-dione

| Conditions | Yield |
|---|---|
| With butan-1-ol at 105 - 110℃; for 6h; | A 28.6% B 25% C 42% |

-
-
121252-83-3
N-prolyl>proline

-
-
71-36-3
butan-1-ol

-
A
-
121252-84-4
(R)-1-[(R)-1-(Dibutoxy-phosphoryl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carboxylic acid butyl ester

-
B
-
107-66-4
dibutyl phosphate

-
C
-
19943-27-2
rac-octahydrodipyrrolo[1,2-a:1',2'-d]pyrazine-5,10-dione

| Conditions | Yield |
|---|---|
| at 105 - 110℃; for 6h; | A 28.6% B 25% C 42% |


| Conditions | Yield |
|---|---|
| With ethanol Hydrogenation.an Platin; |

| Conditions | Yield |
|---|---|
| With water; trichlorophosphate | |
| With triethylamine; trichlorophosphate In toluene at 60℃; for 1h; |

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; dimethylsulfide for 18h; Ambient temperature; |
-
-
126-73-8
phosphoric acid tributyl ester

-
A
-
1623-15-0
n-butyl phosphoric acid

-
B
-
674-48-6
phosphorofluoridic acid dibutyl ester

-
C
-
372-03-2
phosphorofluoridic acid monobutyl ester

-
D
-
26078-17-1
phosphorodifluoridic acid butyl ester

-
E
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| In sulfuric acid; hydrogen fluoride Product distribution; time, composition of the aqueous solution; |


-
-
126-73-8
phosphoric acid tributyl ester

-
A
-
1623-15-0
n-butyl phosphoric acid

-
B
-
109-69-3
n-Butyl chloride

-
C
-
107-66-4
dibutyl phosphate

-
D
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 25℃; Mechanism; Product distribution; Rate constant; different HCl concentrations; |


| Conditions | Yield |
|---|---|
| With phosphorus pentaoxide microwave irradiation; |
-
-
126-73-8
phosphoric acid tributyl ester

-
A
-
1623-15-0
n-butyl phosphoric acid

-
B
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 100℃; for 2h; Title compound not separated from byproducts.; | |
| With (238)plutonium; nitric acid for 21h; Concentration; Irradiation; Radiolysis; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 78.6 percent / Et3N, CCl4 / ethanol; H2O / 14 h / 0 °C 2: Et3N, CCl4 / CH2Cl2 / 10 h / 0 °C 3: Et3N / 6 h / 0 °C 4: 76.3 percent / LiOH*H2O, H2O / tetrahydrofuran; methanol / 5 h / 60 °C / various bases, other time and temperature 5: 25 percent / butanol / 6 h / 105 - 110 °C View Scheme | |
| With copper diacetate In tetrahydrofuran-d8 at 20℃; Inert atmosphere; | |
| Multi-step reaction with 2 steps 1: copper dichloride / tetrahydrofuran / 0.42 h / 0 - 20 °C 2: sodium hydroxide / tetrahydrofuran; water / 0 °C View Scheme | |
| With Nitrogen dioxide at 35℃; Cooling; |
-
-
121252-81-1
N-(dibutyloxyphosphinyl)proline

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: Et3N, CCl4 / CH2Cl2 / 10 h / 0 °C 2: Et3N / 6 h / 0 °C 3: 76.3 percent / LiOH*H2O, H2O / tetrahydrofuran; methanol / 5 h / 60 °C / various bases, other time and temperature 4: 25 percent / butanol / 6 h / 105 - 110 °C View Scheme |
-
-
121252-82-2
N-proline methyl ester

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 76.3 percent / LiOH*H2O, H2O / tetrahydrofuran; methanol / 5 h / 60 °C / various bases, other time and temperature 2: 25 percent / butanol / 6 h / 105 - 110 °C View Scheme |

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Et3N / 6 h / 0 °C 2: 76.3 percent / LiOH*H2O, H2O / tetrahydrofuran; methanol / 5 h / 60 °C / various bases, other time and temperature 3: 25 percent / butanol / 6 h / 105 - 110 °C View Scheme |
-
-
7647-01-0
hydrogenchloride

-
-
80937-33-3
oxygen

-
-
71-36-3
butan-1-ol

-
A
-
126-73-8
phosphoric acid tributyl ester

-
B
-
109-69-3
n-Butyl chloride

-
C
-
107-66-4
dibutyl phosphate

-
D
-
7646-85-7
zinc(II) chloride

| Conditions | Yield |
|---|---|
| copper dichloride In butan-1-ol Kinetics; |

-
-
1369964-80-6
dibutyl (1,1-dioxido-4-oxo-3-phenyl-4H-thiochromen-2-yl)methyl phosphate

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| In chloroform-d1 for 0.183333h; Irradiation; | > 99 %Spectr. |
-
-
107-66-4
dibutyl phosphate

-
-
16298-74-1
sodium salt of dibutyl phosphate

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water at 20℃; for 1h; | 100% |
| With sodium hydroxide | |
| With sodium hydrogencarbonate In water |

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In dichloromethane at 20℃; for 1h; | 100% |

-
-
107-66-4
dibutyl phosphate

-
-
208656-42-2
dibutyl (2-O-benzoyl-3,4,6-tri-O-benzyl-β-D-glucopyranosyl) phosphate

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In dichloromethane at 20℃; for 0.5h; | 100% |
-
-
32353-64-3
1-butyl-4-methylpyridin-1-ium iodide

-
-
107-66-4
dibutyl phosphate

-
-
1289675-21-3
1-butyl-4-methylpyridinium dibutylphosphate

| Conditions | Yield |
|---|---|
| Stage #1: dibutyl phosphate With Amberlyst A-26 (OH- form) In methanol; water at 20℃; Stage #2: 1-butyl-4-methylpyridin-1-ium iodide In methanol | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: dibutyl phosphate With Amberlyst A-26 (OH- form) In methanol; water at 20℃; Stage #2: 1,3-dimethylimidazolim iodide In methanol | 100% |
-
-
65039-05-6
1-methyl-3-(n-butyl)imidazolium iodide

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| Stage #1: dibutyl phosphate With Amberlyst A-26 (OH- form) In methanol; water at 20℃; Stage #2: 1-methyl-3-(n-butyl)imidazolium iodide In methanol | 100% |
-
-
143085-46-5
1,3-bis(n-butyl)imidazolium iodide

-
-
107-66-4
dibutyl phosphate

-
-
654058-05-6
C8H18O4P(1-)*C11H21N2(1+)

| Conditions | Yield |
|---|---|
| Stage #1: dibutyl phosphate With Amberlyst A-26 (OH- form) In methanol; water at 20℃; Stage #2: 1,3-dibutyl-1,3-imidazolium iodide In methanol | 100% |
-
-
107-66-4
dibutyl phosphate

-
-
1289675-18-8
1-butyl-2,3-dimethylimidazolium dibutylphosfonate

| Conditions | Yield |
|---|---|
| Stage #1: dibutyl phosphate With Amberlyst A-26 (OH- form) In methanol; water at 20℃; Stage #2: 1-butyl-2,3-dimethylimidazolium bromide In methanol | 100% |
-
-
1448263-56-6
methyl 3-O-benzyl-4-O-tert-butyldimethylsilyl-β-L-idopyranuronate 1,2-(methyl-orthoacetate)

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| Stage #1: methyl 3-O-benzyl-4-O-tert-butyldimethylsilyl-β-L-idopyranuronate 1,2-(methyl-orthoacetate) In dichloromethane at 20℃; for 0.25h; Inert atmosphere; Molecular sieve; Stage #2: dibutyl phosphate In dichloromethane Inert atmosphere; Molecular sieve; | 100% |
-
-
1448263-56-6
methyl 3-O-benzyl-4-O-tert-butyldimethylsilyl-β-L-idopyranuronate 1,2-(methyl-orthoacetate)

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Molecular sieve; | 100% |
| In dichloromethane at 20℃; Molecular sieve; | 100% |
-
-
78-73-9
cholinium hydrogen carbonate

-
-
107-66-4
dibutyl phosphate

-
-
1241962-99-1
cholinium dibutyl phosphate

| Conditions | Yield |
|---|---|
| In water at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| In methanol | 100% |
| In methanol at 0 - 20℃; | 98% |
| In methanol at 0 - 20℃; for 1h; | 98% |
-
-
373606-55-4
2-O-benzyl-3,4-di-O-pivaloyl-α-L-fucopyranosyl trichloroacetimidate

-
-
107-66-4
dibutyl phosphate

-
-
373606-28-1
dibutyl 2-O-benzyl-3,4-di-O-pivaloyl-β-L-fucopyranoside phosphate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.5h; | 99% |

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2,4-di-O-benzyl-3,6-di-O-picoloyl-1-thio-α-D-mannopyranoside; dibutyl phosphate In dichloromethane at 20℃; for 1h; Molecular sieve; Inert atmosphere; Stage #2: With N-iodo-succinimide; trifluorormethanesulfonic acid In dichloromethane at 20℃; for 1h; Molecular sieve; Inert atmosphere; diastereoselective reaction; | 99% |
-
-
107-66-4
dibutyl phosphate

-
-
208047-71-6
3,4-di-O-benzyl-2-deoxy-6-O-levulinyl-2-phthalimido-β-D-glucopyranosyl trichloroacetamidate

| Conditions | Yield |
|---|---|
| In toluene for 14h; | 98% |

| Conditions | Yield |
|---|---|
| In various solvent(s) at 80℃; for 1h; | 98% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 0.25h; | 98% |
| With triethylamine |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 12h; Inert atmosphere; | 98% |

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In dichloromethane at 20℃; for 0.5h; | 97% |

-
-
107-66-4
dibutyl phosphate

-
-
1059116-10-7
dibutyl (2-O-benzoyl-3,4-di-O-benzyl-α-D-mannopyranosyl) phosphate

| Conditions | Yield |
|---|---|
| Molecular sieve; Inert atmosphere; | 97% |
-
-
1382354-48-4
1,3-dibutyl-5,6-dimethylbenzimidazolium iodide

-
-
107-66-4
dibutyl phosphate

-
-
1382354-51-9
C8H18O4P(1-)*C17H27N2(1+)

| Conditions | Yield |
|---|---|
| Stage #1: dibutyl phosphate With Amberlyst A-26 (OH- form) In methanol; water at 20℃; Stage #2: 1,3-dibutyl-5,6-dimethylbenzimidazolium iodide In methanol | 97% |
-
-
62774-33-8
phenyl 2,3,4,6-tetra-O-benzoyl-1-thio-β-D-glucopyranoside

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| Stage #1: phenyl 2,3,4,6-tetra-O-benzoyl-1-thio-β-D-glucopyranoside; dibutyl phosphate In dichloromethane at 20℃; for 1h; Molecular sieve; Inert atmosphere; Stage #2: With N-iodo-succinimide; trifluorormethanesulfonic acid In dichloromethane at 20℃; for 18h; Molecular sieve; Inert atmosphere; | 97% |

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; trifluorormethanesulfonic acid In dichloromethane at 0 - 20℃; for 1.33333h; Molecular sieve; Inert atmosphere; | 97% |
-
-
75-30-9
2-iodo-propane

-
-
107-66-4
dibutyl phosphate

-
-
60140-72-9
phosphoric acid dibutyl ester isopropyl ester

| Conditions | Yield |
|---|---|
| With cesium fluoride In acetonitrile for 6h; Ambient temperature; | 96% |

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethylchlorformamidinium chloride; triethylamine In dichloromethane at 0 - 20℃; for 2h; | 96% |
-
-
16697-49-7
3,4,6-tri-O-benzyl-1,2-O-(methoxyethylidene)-β-D-mannopyranose

-
-
107-66-4
dibutyl phosphate

-
-
604767-74-0
di-n-butyl (2-O-acetyl-3,4,6-tri-O-benzyl-α-D-mannopyranosyl) phosphate

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In dichloromethane at 20℃; for 0.366667h; | 96% |
| Molecular sieve; Inert atmosphere; | 93% |
| With triethylamine In dichloromethane at 0 - 25℃; Molecular sieve; Inert atmosphere; |

-
-
107-66-4
dibutyl phosphate

| Conditions | Yield |
|---|---|
| Stage #1: C23H24Cl3NO11S; dibutyl phosphate In dichloromethane at 20℃; for 3h; Inert atmosphere; Molecular sieve; Stage #2: With N-iodo-succinimide; trifluorormethanesulfonic acid In diethyl ether; dichloromethane at 0 - 4℃; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; trifluorormethanesulfonic acid In dichloromethane at 0℃; | 96% |
-
-
1572443-60-7
C62H66O7SSi

-
-
107-66-4
dibutyl phosphate

-
-
1572443-75-4
dibutyl 6-O-triisopropylsilyl-2-O-(2-((4-methoxybenzyloxy)methyl)benzoyl)-2,3-di-O-(2-methylnaphthyl)-α-D-mannopyranoside phosphate

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; trimethylsilyl trifluoromethanesulfonate In dichloromethane Inert atmosphere; | 96% |
Dibutyl phosphate Consensus Reports
Reported in EPA TSCA Inventory.
Dibutyl phosphate Standards and Recommendations
OSHA PEL: TWA 1 ppm; STEL 2 ppm
ACGIH TLV: TWA 1 ppm; STEL 2 ppm
Dibutyl phosphate Analytical Methods
For occupational chemical analysis use NIOSH: Dibutyl Phosphate, 5017.
Dibutyl phosphate Specification
The Dibutyl phosphate is an organic compound with the formula C8H19O4P. The IUPAC name of this chemical is dibutyl hydrogen phosphate. With the CAS registry number 107-66-4 and EINECS 203-509-8, it is also named as Di-n-butyl hydrogen phosphate. It is clear liquid which is insoluble in water and soluble in diethyl ether, carbon tetrachloride and other organic solvents. What's more, it is toxic and flammable. When heated to decomposition it emits toxic fumes of POx. So the storage environment should be well-ventilated, low-temperature and dry. Keep Dibutyl phosphate separate from raw materials of food.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 2.20; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -1.17; (4)ACD/LogD (pH 7.4): -1.3; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 4; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 8; (12)Polar Surface Area: 54.57 Å2; (13)Index of Refraction: 1.436; (14)Molar Refractivity: 51.01 cm3; (15)Molar Volume: 194.9 cm3; (16)Polarizability: 20.22×10-24 cm3; (17)Surface Tension: 36.3 dyne/cm; (18)Density: 1.078 g/cm3; (19)Flash Point: 120.3 °C; (20)Enthalpy of Vaporization: 56.54 kJ/mol; (21)Boiling Point: 275.3 °C at 760 mmHg; (22)Vapour Pressure: 0.00141 mmHg at 25°C.
Uses of Dibutyl phosphate: It is used as extraction agent of uranium and thorium. It also can react with 2-iodo-propane to get phosphoric acid dibutyl ester isopropyl ester. This reaction needs reagent CsF and solvent acetonitrile at ambient temperature. The reaction time is 6 hours. The yield is 96%.
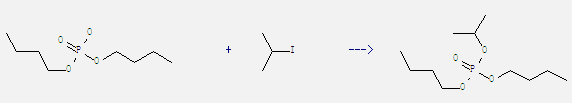
When you are using this chemical, please be cautious about it as the following:
It can cause burns and is irritating to respiratory system and skin. It also has risk of serious damage to the eyes, so people should avoid contact with eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If you want to contact this product, you must wear suitable protective clothing, gloves and eye/face protection. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
People can use the following data to convert to the molecule structure.
1. SMILES:O=P(OCCCC)(OCCCC)O
2. InChI:InChI=1/C8H19O4P/c1-3-5-7-11-13(9,10)12-8-6-4-2/h3-8H2,1-2H3,(H,9,10)
3. InChIKey:JYFHYPJRHGVZDY-UHFFFAOYAZ
The following are the toxicity data which has been tested.
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 3200mg/kg (3200mg/kg) | National Technical Information Service. Vol. OTS0528344 |
Related Products
- Dibutyl (diethylene glycol bisphthalate)
- Dibutyl 2,2-dithiobisbenzoate
- Dibutyl 2,2'-thiobisacetate
- Dibutyl adipate
- Dibutyl butanephosphonate
- Dibutyl chlorendate
- Dibutyl chlorophosphate
- Dibutyl D-(-)-tartrate
- Dibutyl fumarate
- Dibutyl isophthalate
- 107665-59-8
- 107666-25-1
- 107667-60-7
- 107668-79-1
- 107671-54-5
- 1076-74-0
- 107674-15-7
- 107680-18-2
- 107682-63-3
- 107-68-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View