-
Name
Ethyl valerate
- EINECS 208-726-1
- CAS No. 539-82-2
- Article Data90
- CAS DataBase
- Density 0.882 g/cm3
- Solubility Insoluble in water, miscible with organic solvents
- Melting Point -92 ~ -90 °C
- Formula C7H14O2
- Boiling Point 145.9 °C at 760 mmHg
- Molecular Weight 130.187
- Flash Point 38.9 °C
- Transport Information UN 3272 3/PG 3
- Appearance Clear colorless liquid
- Safety 16
- Risk Codes 10
-
Molecular Structure
- Hazard Symbols R10:;
- Synonyms Valeric acid,ethyl ester (6CI,8CI);Ethyl n-pentanoate;Ethyl n-valerate;Ethyl pentanoate;NSC 8868;Pentanoic acid, ethyl ester;
- PSA 26.30000
- LogP 1.73970
Synthetic route

| Conditions | Yield |
|---|---|
| With sulfuric acid at 100℃; for 3h; | 99% |
| With sulfuric acid at 100℃; for 3h; | 99% |
| With Rhizomucor miehei lipase In n-heptane at 40℃; for 24h; Enzymatic reaction; | 63.1% |

| Conditions | Yield |
|---|---|
| With 0.08%-SO3H functionalized-benzylated Al-SBA-15 nanoporous catalyst at 79.84℃; for 2h; | 95% |

| Conditions | Yield |
|---|---|
| With 1H-imidazole In neat (no solvent) at 125℃; for 0.0666667h; Temperature; Wavelength; Microwave irradiation; | 86% |

| Conditions | Yield |
|---|---|
| at 120℃; for 4h; | 86% |
-
-
64-17-5
ethanol

-
-
111-26-2
hexan-1-amine

-
-
201230-82-2
carbon monoxide

-
A
-
123-66-0
Ethyl hexanoate

-
B
-
539-82-2
ethyl n-valerate

-
C
-
106-30-9
ethyl heptanoate

-
D
-
7451-47-0
N-hexylcarbamic acid ethyl ester

| Conditions | Yield |
|---|---|
| With oxygen; Sulfate; zirconium(IV) oxide; palladium dichloride at 170℃; under 45003.6 Torr; for 3h; Further byproducts given. Title compound not separated from byproducts; | A n/a B n/a C n/a D 81% |

-
-
64-17-5
ethanol

-
-
201230-82-2
carbon monoxide

-
-
109-73-9
N-butylamine

-
A
-
871-71-6
N-butylformamide

-
B
-
539-82-2
ethyl n-valerate

-
C
-
591-62-8
N-(ethoxycarbonyl)butylamine

-
D
-
105-54-4
butanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With oxygen; Sulfate; zirconium(IV) oxide; palladium dichloride at 170℃; under 45003.6 Torr; for 3h; Title compound not separated from byproducts; | A n/a B n/a C 80% D n/a |

-
-
14660-52-7
ethyl 5-bromovalerate

-
-
539-82-2
ethyl n-valerate

| Conditions | Yield |
|---|---|
| In hexane; ethyl acetate; mineral oil | 80% |
-
-
539-88-8
4-oxopentanoic acid ethyl ester

-
A
-
539-82-2
ethyl n-valerate

-
B
-
108-29-2
5-methyl-dihydro-furan-2-one

-
C
-
109-52-4
valeric acid

| Conditions | Yield |
|---|---|
| With hydrogen; 10 wtpercent nickel/0.1 wtpercent platinum on silica Product distribution / selectivity; | A n/a B 77% C n/a |
| With hydrogen; 10 wtpercent rhenium/0.1 wtpercent platinum on silica Product distribution / selectivity; | A n/a B 68% C n/a |
| With hydrogen; 1 wtpercent ruthenium on silica Product distribution / selectivity; | A n/a B 54% C n/a |


| Conditions | Yield |
|---|---|
| With 2,6-dimethyl-1,4-benzoquinone In dichloromethane at 20℃; for 0.5h; | 76% |

| Conditions | Yield |
|---|---|
| With hydrogen at 240℃; under 22502.3 Torr; for 3h; Reagent/catalyst; Autoclave; | A 74% B 23% |


| Conditions | Yield |
|---|---|
| With 3Pd-10Nb-500AC; hydrogen In ethanol at 250℃; under 15001.5 Torr; for 10h; Autoclave; | 70% |
-
-
64-17-5
ethanol

-
-
108-29-2
5-methyl-dihydro-furan-2-one

-
A
-
539-82-2
ethyl n-valerate

-
B
-
109-52-4
valeric acid

| Conditions | Yield |
|---|---|
| With hydrogen at 240℃; under 22502.3 Torr; for 3h; Autoclave; | A 69% B 22% |
| 10 wt% nickel/0.1 wt% platinum on silica and zeolite β Product distribution / selectivity; Heating / reflux; | |
| 1 wt% platinum on silica and zeolite β Product distribution / selectivity; | |
| nickel on silica Product distribution / selectivity; | |
| With hydrogen at 250℃; under 760.051 Torr; for 50h; Catalytic behavior; Reagent/catalyst; Temperature; Flow reactor; |

-
-
64-17-5
ethanol

-
-
123-76-2
levulinic acid

-
A
-
539-82-2
ethyl n-valerate

-
B
-
108-29-2
5-methyl-dihydro-furan-2-one

| Conditions | Yield |
|---|---|
| With hydrogen at 210℃; under 22502.3 Torr; for 3h; Autoclave; | A 13% B 68% |

-
-
14660-52-7
ethyl 5-bromovalerate

-
-
124-38-9
carbon dioxide

-
-
584-08-7
potassium carbonate

-
A
-
539-82-2
ethyl n-valerate

-
B
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With nickel(II) bromide dimethoxyethane; 2.9-dimethyl-1,10-phenanthroline; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; C60H36N2 In N,N-dimethyl-formamide at 20℃; for 24h; Molecular sieve; Irradiation; | A 25 %Spectr. B 66% |


-
-
539-82-2
ethyl n-valerate

| Conditions | Yield |
|---|---|
| With mercury(II) diacetate; tetrabutyl ammonium fluoride In tetrahydrofuran; methanol at 0℃; for 0.583333h; | 62% |

| Conditions | Yield |
|---|---|
| With bis(acetylacetonate)oxovanadium; dihydrogen peroxide at 60℃; for 4h; | 60% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; 18-crown-6 ether In ethanol; benzene 1.) room temperature, 9 h, 2.) reflux, 13 h; | 59% |
| Multi-step reaction with 2 steps 1: potassium hydroxide / ethanol / 72 h / 20 °C 2: 1H-imidazole / neat (no solvent) / 0.07 h / 125 °C / Microwave irradiation View Scheme |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; sodium hydrogencarbonate; polymer-anchored palladium(II) chloride at 80℃; under 1551.4 Torr; for 8h; | 56% |


| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium carbonate In water; benzene for 6h; Ambient temperature; | 23% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 25℃; Inert atmosphere; | 17% |

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
682802-51-3
1,1-diethoxy-pent-1-ene

-
-
539-82-2
ethyl n-valerate

| Conditions | Yield |
|---|---|
| With water |
-
-
42367-85-1
bis-(5-ethoxycarbonyl-valeryl)-peroxide

-
A
-
539-82-2
ethyl n-valerate

-
B
-
1968-40-7
ethyl 4-pentenoate

-
C
-
141-28-6
diethyl adipate

-
D
-
110-40-7
diethyl sebacate

| Conditions | Yield |
|---|---|
| bei der explosiven Zersetzung; |

-
-
855597-17-0
O1-benzoyl-monoperoxyadipic acid-6-ethyl ester

-
-
121-69-7
N,N-dimethyl-aniline

-
-
539-82-2
ethyl n-valerate

| Conditions | Yield |
|---|---|
| Pyrolysis; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; amalgamated zinc; ethanol at 20℃; | |
| With hydrogen | |
| Multi-step reaction with 2 steps 1: hydrogen; 3Pd-10Nb-500AC / water / 5 h / 100 °C / 3750.38 Torr 2: 10Nb-500-AC; water / 5 h / 100 °C / 3750.38 Torr / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: hydrogen; 3Pd-10Nb-500AC / water / 5 h / 100 °C / 3750.38 Torr 2: hydrogen; 3Pd-10Nb-500AC / ethanol / 10 h / 250 °C / 15001.5 Torr / Autoclave View Scheme | |
| Multi-step reaction with 3 steps 1: hydrogen; 3Pd-10Nb-500AC / water / 5 h / 100 °C / 3750.38 Torr 2: hydrogen; 3Pd-10Nb-500AC / water / 5 h / 100 °C / 3750.38 Torr 3: hydrogen; 3Pd-10Nb-500AC / ethanol / 10 h / 250 °C / 15001.5 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| With magnesium In benzene |

| Conditions | Yield |
|---|---|
| With water 1.) diethyl ether, room temp., 2 h, 2.) 1N HCl; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With NaH-t-AmONa-Ni(OAc)2-bpy In tetrahydrofuran at 63℃; for 1h; Product distribution; Mechanism; other thiophenecarboxylates; other reagents, and solvents, var. reaction times; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; titanium tetrachloride for 25h; Heating; Yield given; |
-
-
539-82-2
ethyl n-valerate

-
-
38291-82-6
valeric acid hydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol for 12h; Reflux; | 99% |
| With hydrazine hydrate In ethanol at 100℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With ethanol; sodium for 20h; Heating / reflux; | 98% |

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 100℃; | 93.8% |

| Conditions | Yield |
|---|---|
| Stage #1: chlorobromomethane With lithium diisopropyl amide In tetrahydrofuran at -95℃; for 0.25h; Stage #2: ethyl n-valerate In tetrahydrofuran at -95 - -60℃; for 2.5h; | 92% |
-
-
539-82-2
ethyl n-valerate

-
-
19259-70-2
n-hexyldiphenylphosphine oxide

-
-
201872-03-9
6-Diphenylphosphinoylundecan-5-one

| Conditions | Yield |
|---|---|
| With 2,2,6,6-tetramethyl-piperidine; n-butyllithium In tetrahydrofuran; hexane 1.) -30 deg C, 1 h, 2.) -78 deg C, 2 h; | 90% |
-
-
539-82-2
ethyl n-valerate

-
-
3815-69-8
5-amino-1-(phenylmethyl)-1H-imidazole-4-carboxamide

-
-
640274-67-5
9-benzyl-2-butylhypoxanthine

| Conditions | Yield |
|---|---|
| With ethanol; sodium for 10h; Heating; | 89% |
-
-
539-82-2
ethyl n-valerate

-
-
35658-10-7
[1,1-2H2]-1-pentanol

| Conditions | Yield |
|---|---|
| With lithium aluminium deuteride In diethyl ether at 20℃; | 88% |
| With lithium aluminium deuteride |
-
-
539-82-2
ethyl n-valerate

-
-
66769-61-7
1-(iodomethyl)-3,5-dimethoxybenzene

-
-
196926-60-0
2-(3,5-Dimethoxy-benzyl)-pentanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane for 2h; Ambient temperature; | 88% |
-
-
2564-83-2, 45842-10-2
2,2,6,6-Tetramethyl-1-piperidinyloxy free radical

-
-
539-82-2
ethyl n-valerate

-
A
-
1377149-79-5
ethyl 2-((2,2,6,6-tetramethylpiperidin-1-yl)oxy)pentanoate

-
B
-
1357253-56-5
methyl 2-methyl-2-(2,2,6,6-tetramethylpiperidin-1-yloxy)butyrate

| Conditions | Yield |
|---|---|
| Stage #1: ethyl n-valerate With n-butyllithium; diisopropylamine; lithium chloride In tetrahydrofuran; hexane at -78℃; for 0.333333h; Inert atmosphere; Stage #2: 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical With ferrocenium hexafluorophosphate In tetrahydrofuran; hexane at -78℃; Inert atmosphere; | A 87% B 4% |

-
-
539-82-2
ethyl n-valerate

-
-
70770-06-8
1-benzyloxy-3-phenylpropane

-
-
5451-88-7
3-phenyl-1-propyl pentanoate

| Conditions | Yield |
|---|---|
| With Bromotrichloromethane; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate In dichloromethane at 20℃; for 12h; Inert atmosphere; Irradiation; | 87% |
-
-
539-82-2
ethyl n-valerate

-
-
1356411-42-1
1-{4-[3-(dibutylamino)propyl]phenyl}ethanone

-
-
1356411-41-0
1-{4-[3-(dibutylamino)propyl]phenyl}heptane-1,3-dione

| Conditions | Yield |
|---|---|
| With sodium methylate In 1-methyl-pyrrolidin-2-one at 5 - 20℃; | 82% |

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 70℃; | 81% |
| With sodium hydride In tetrahydrofuran |

| Conditions | Yield |
|---|---|
| for 0.166667h; microwave irradiation; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-phenylimidazole With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 1h; Inert atmosphere; Stage #2: ethyl n-valerate In tetrahydrofuran; hexane at -78 - 20℃; for 8h; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-methyl-1H-imidazole With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 1h; Inert atmosphere; Stage #2: ethyl n-valerate In tetrahydrofuran; hexane at -78 - 20℃; for 8h; Inert atmosphere; | 81% |
-
-
539-82-2
ethyl n-valerate

-
-
683-08-9
Diethyl methylphosphonate

-
-
3450-63-3
2-oxohexylphosphonate de diethyle

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; | 80.5% |
-
-
539-82-2
ethyl n-valerate

-
-
34837-55-3
Phenylselenyl bromide

-
-
308335-49-1
ethyl 2-phenylselanylpentanoate

| Conditions | Yield |
|---|---|
| With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 0.25h; selenenylation; | 78% |
-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
539-82-2
ethyl n-valerate

-
A
-
597-93-3
tri-n-butyl-carbinol

-
B
-
502-56-7
5-Nonanone

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In tetrahydrofuran 1.) -100 deg C, 10 min, 2.) -78 deg C, 10 min; | A n/a B 77% |
| With hydrogenchloride; chloro-trimethyl-silane multistep reaction, acylation of organolithium reagents by esters; |


| Conditions | Yield |
|---|---|
| With n-butyllithium; tetramethylpiperidine 1.) -30 deg C, 1 h; 2.) -78 deg C, 2 h; | 77% |
| With 2,2,6,6-tetramethyl-piperidine; n-butyllithium In tetrahydrofuran; hexane 1.) -30 deg C, 1 h, 2.) -78 deg C, 2 h; | 77% |
-
-
539-82-2
ethyl n-valerate

-
-
111198-02-8
1-(phenoxymethyl)-1H-benzo[d][1,2,3]triazole

-
-
189343-52-0
1-Benzotriazol-1-yl-1-phenoxy-hexan-2-one

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 12h; | 75% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In tetrahydrofuran; water; ethyl acetate | 74% |
Ethyl valerate Specification
The Ethyl valerate with CAS registry number of 539-82-2 is also known as Pentanoic acid, ethyl ester. The IUPAC name is Ethyl pentanoate. It belongs to product categories of Fatty & Aliphatic Acids, Esters, Alcohols & Derivatives; Organics; Alphabetical Listings; E-F; Flavors and Fragrances; EstersSaturated Fatty Acids and Derivatives; Ethyl Ester; Alpha Sort; Chemical Class; E; E-LAlphabetic; EQ - EZAnalytical Standards; Esters; Volatiles/ Semivolatiles; C6 to C7Saturated Fatty Acids and Derivatives; Carbonyl Compounds. Its EINECS registry number is 208-726-1. In addition, the formula is C7H14O2 and the molecular weight is 130.18. This chemical is a clear colorless liquid and should be stored in cool, ventilated, dry room away from heat and fire. What's more, it is poorly soluble in water but miscible with organic solvents. During using it, keep away from sources of ignition as it is flammable.
Physical properties about Ethyl valerate are: (1)ACD/LogP: 2.30; (2)ACD/LogD (pH 5.5): 2.3; (3)ACD/LogD (pH 7.4): 2.3; (4)ACD/BCF (pH 5.5): 33.1; (5)ACD/BCF (pH 7.4): 33.1; (6)ACD/KOC (pH 5.5): 426.09; (7)ACD/KOC (pH 7.4): 426.09; (8)#H bond acceptors: 2 ; (9)#Freely Rotating Bonds: 5; (10)Index of Refraction: 1.406; (11)Molar Refractivity: 36.25 cm3; (12)Molar Volume: 147.5 cm3; (13)Surface Tension: 26.6 dyne/cm; (14)Density: 0.882 g/cm3; (15)Flash Point: 38.9 °C; (16)Enthalpy of Vaporization: 36.96 kJ/mol; (17)Boiling Point: 145.9 °C at 760 mmHg; (18)Vapour Pressure: 4.75 mmHg at 25 °C.
Preparation of Ethyl valerate: it is prepared by reaction of pentanoic acid with ethanol. The reaction needs reagent conc. H2SO4 and solvent benzene with other condition of heating for 48 hours. The yield is about 62.3%.
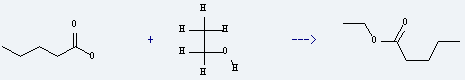
Uses of Ethyl valerate: it is used to produce 3-chloro-oct-1-en-4-one by reaction with 3-chloroallyllithium. The reaction occurs with solvent tetrahydrofuran at the temperature of -90 °C for 30 minutes. The yield is about 72%.

You can still convert the following datas into molecular structure:
1. Canonical SMILES: CCCCC(=O)OCC
2. InChI: InChI=1S/C7H14O2/c1-3-5-6-7(8)9-4-2/h3-6H2,1-2H3
3. InChIKey: ICMAFTSLXCXHRK-UHFFFAOYSA-N
Related Products
- Ethyl (13-cis)-9-(4-methoxy-2,3,6-trimethylphenyl)-3,7-dimethyl-2,4,6,8-nonatetraenoate
- Ethyl (2-bromopropionamido)acetate
- Ethyl (2-cyanoimino-5,6-dichloro-1,2,3,4-tetrahydroquinazolin-3-yl)acetate
- Ethyl (2Z)-chloro[(4-methoxyphenyl)hydrazono]acetate
- Ethyl (3R)-piperidine-3-carboxylate
- Ethyl (3R,S)-2,2-difluoro-3-hydroxy-3-(2,2-dimethyldioxolan-4-yl)propionate
- Ethyl (3S)-4-bromo-3-hydroxybutanoate
- Ethyl (3S)-piperidine-3-carboxylate
- Ethyl (3-trifluoromethylbenzoyl)acetate
- Ethyl (3α,5β,7α,12α)-3,7,12-trihydroxycholan-24-oate
- 5398-23-2
- 5398-24-3
- 5398-25-4
- 5398-29-8
- 539834-19-0
- 5398-34-5
- 5398-36-7
- 53984-03-5
- 53984-36-4
- 5398-44-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View