-
Name
17a-Hydroxyprogesterone caproate
- EINECS 211-138-8
- CAS No. 630-56-8
- Article Data28
- CAS DataBase
- Density 1.1 g/cm3
- Solubility
- Melting Point 119 °C
- Formula C27H40O4
- Boiling Point 536.4 °C at 760 mmHg
- Molecular Weight 428.612
- Flash Point 227.6 °C
- Transport Information
- Appearance White or almost White Crystalline power
- Safety 53-22-36/37/39-45
- Risk Codes 61
-
Molecular Structure
-
Hazard Symbols
 T
T
- Synonyms Pregn-4-ene-3,20-dione,17-hydroxy-, hexanoate (7CI,8CI);Progesterone, 17-hydroxy-, hexanoate (6CI);Hexanoic acid, ester with 17-hydroxypregn-4-ene-3,20-dione (8CI);17a-Caproyloxypregn-4-ene-3,20-dione;17a-Hydroxypregn-4-ene-3,20-dionecaproate;17a-Hydroxypregn-4-ene-3,20-dionehexanoate;17a-Hydroxyprogesteronecaproate;17a-Hydroxyprogesteronen-caproate;Delalutin;Depo-proluton;Hormofort;Pregn-4-ene-3,20-dione,17-[(1-oxohexyl)oxy]-;NSC 17592;Neolutin;Primolut Depot;Procyte Depo;Proge;Syngynon;Teralutil;
- PSA 60.44000
- LogP 5.96960
Synthetic route
-
-
2051-49-2
n-hexanoic anhydride

-
-
68-96-2
17-hydroxyprogesterone

-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid |
-
-
94201-44-2
17-hexanoyloxy-3β-hydroxy-pregn-5-en-20-one

-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| With cyclohexanone; aluminum isopropoxide | |
| With cyclohexanone; aluminum isopropoxide |
-
-
6084-02-2
3β-formyloxy-17-hexanoyloxy-pregn-5-en-20-one

-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| With cyclohexanone; aluminum isopropoxide | |
| With cyclohexanone; aluminum isopropoxide |
-
-
68-96-2
17-hydroxyprogesterone

-
-
142-62-1
hexanoic acid

-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| With trifluoroacetic anhydride | |
| Stage #1: 17-hydroxyprogesterone; hexanoic acid With pyridine; toluene-4-sulfonic acid In toluene at 110 - 120℃; for 3h; Stage #2: With hydrogenchloride In ethanol; water for 2h; Reagent/catalyst; Solvent; Reflux; | 19.8 g |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: toluene-4-sulfonic acid; benzene 2: cyclohexanone; aluminium isopropylate View Scheme | |
| Multi-step reaction with 3 steps 1: toluene-4-sulfonic acid 2: hydrogenchloride 3: cyclohexanone; aluminum isopropoxide View Scheme |
-
-
20867-15-6
3β-formyloxy-17-hydroxy-pregn-5-en-20-one

-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: toluene-4-sulfonic acid; benzene 2: cyclohexanone; aluminium isopropylate View Scheme |
-
-
86790-83-2
3β-acetoxy-17-hexanoyloxy-pregn-5-en-20-one

-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogenchloride 2: cyclohexanone; aluminum isopropoxide View Scheme |
-
-
68-96-2
17-hydroxyprogesterone

-
-
142-62-1
hexanoic acid

-
A
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| With pyridine; toluene-4-sulfonic acid In toluene at 110 - 120℃; for 3h; Solvent; |


| Conditions | Yield |
|---|---|
| 83% |
-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

-
-
4975-65-9
17α-Hydroxypregna-4,6-diene-3,20-dione 17-capronate

| Conditions | Yield |
|---|---|
| With acetic anhydride; chloranil; 5-sulfosalicylic Acid In xylene for 6h; Heating; | 40% |
-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| (i) MeNH2, MeOH, (ii) aq. Ca(OCl)2, NH3, OTs; Multistep reaction; |
-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione
-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione


| Conditions | Yield |
|---|---|
| at 37℃; for 0.166667h; in incubation mixture, with rat liver microsomes; or perbenzoic acid oxidation of dienol acetate; |
-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

-
-
81089-15-8
3β,17α-Dihydroxypregna-4,6-dien-20-one 17-caproate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 40 percent / chloranil, sulfosalicylic acid, acetic anhydride / xylene / 6 h / Heating 2: 55 percent / sodium borohydride / methanol / 0.5 h / Ambient temperature View Scheme |
-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: acetic acid / Ambient temperature; anodic oxidation; platinum electrode, potassium acetate electrolyte View Scheme |
-
-
630-56-8
17-hexanoyloxy-pregn-4-ene-3,20-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: acetic acid / Ambient temperature; anodic oxidation; platinum electrode, potassium acetate electrolyte View Scheme |
Hydroxyprogesterone caproate Chemical Properties
Molecule structure of Hydroxyprogesterone caproate (CAS NO.630-56-8):
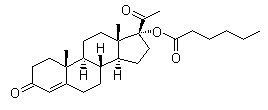
IUPAC Name: [(8R,9S,10R,13S,14S,17R)-17-Acetyl-10,13-dimethyl-3-oxo-2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthren-17-yl] hexanoate
Molecular Weight: 428.6041 g/mol
Molecular Formula: C27H40O4
Density: 1.1 g/cm3
Melting Point: 119 °C
Boiling Point: 536.4 °C at 760 mmHg
Flash Point: 227.6 °C
Index of Refraction: 1.533
Molar Refractivity: 120.62 cm3
Molar Volume: 388.6 cm3
Surface Tension: 43.1 dyne/cm
Enthalpy of Vaporization: 81.31 kJ/mol
Vapour Pressure: 1.4E-11 mmHg at 25 °C
XLogP3: 5.7
H-Bond Acceptor: 4
Rotatable Bond Count: 7
Tautomer Count: 10
Exact Mass: 428.29266
MonoIsotopic Mass: 428.29266
Topological Polar Surface Area: 60.4
Heavy Atom Count: 31
Canonical SMILES: CCCCCC(=O)OC1(CCC2C1(CCC3C2CCC4=CC(=O)CCC34C)C)C(=O)C
Isomeric SMILES: CCCCCC(=O)O[C@@]1(CC[C@@H]2[C@@]1(CC[C@H]3[C@H]2CCC4=CC(=O)CC[C@]4C)C)C(=O)C
InChI: InChI=1S/C27H40O4/c1-5-6-7-8-24(30)31-27(18(2)28)16-13-23-21-10-9-19-17-20(29)11-14-25(19,3)22(21)12-15-26(23,27)4/h17,21-23H,5-16H2,1-4H3/t21-,22+,23+,25+,26+,27+/m1/s1
InChIKey: DOMWKUIIPQCAJU-LJHIYBGHSA-N
EINECS: 211-138-8
Product Categories: Biochemistry; Hydroxyketosteroids; Steroids
Hydroxyprogesterone caproate Uses
Hydroxyprogesterone caproate (CAS NO.630-56-8) is used as progesterone medicine.
Hydroxyprogesterone caproate Production
Hydroxyprogesterone caproate can be prepared by the following sequence:
.gif)
Hydroxyprogesterone caproate Consensus Reports
IARC Cancer Review: Animal Inadequate Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 21 , 1979,p. 399.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) .
Hydroxyprogesterone caproate Safety Profile
Hazard Codes:  T
T
Risk Statements: 61
R61:May cause harm to the unborn child.
Safety Statements: 53-22-36/37/39-45
S53:Avoid exposure - obtain special instructions before use.
S22:Do not breathe dust.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
WGK Germany: 3
RTECS: TU5085000
Human reproductive effects by an unknown route: behavioral effects on newborn. Experimental teratogenic and reproductive effects. Questionable carcinogen. A steroid. Used to treat menstrual disorders, threatened abortion, and sterility. When heated to decomposition it emits acrid smoke and fumes.
Hydroxyprogesterone caproate Specification
Hydroxyprogesterone caproate (CAS NO.630-56-8) is also named as 17-((1-Oxohexyl)oxy)pregn-4-ene-3,20-dione ; 17-Hydroxypregn-4-ene-3,20-dione hexanoate ; 17-alpha-Hexanoyloxypregn-4-ene-3,20-dione ; 17-alpha-Hydroxy progesterone n-caproate ; 17-alpha-Hydroxyprogesterone caproate ; 17-alpha-Hydroxyprogesterone hexanoate ; 17alpha-Caproyloxypregn-4-ene-3,20-dione ; 17alpha-Hydroxyprogesterone caproate ; Caproate d'hydroxyprogesterone ; Caproate d'hydroxyprogesterone [INN-French] ; Caproato de hidroxiprogesterona ; Caproato de hidroxiprogesterona [INN-Spanish] ; Corlutin L.A. ; Delalutin ; Depo-Proluton ; Duraluton ; Estralutin ; Gesterol LA 250 ; Hylutin ; Hyproval ; Hyproval-PA ; Hyroxon ; Idrogestene ; Neolutin ; Neolutin forte ; Proge ; Progesterone caproate ; Progesterone retard pharlon ; Progesterone, 17-hydroxy-, hexanoate ; Proluton depot ; Relutin . Hydroxyprogesterone caproate (CAS NO.630-56-8) is an ester derivative of 17α-hydroxyprogesterone formed from caproic acid (hexanoic acid).
Related Products
- Hydroxyprogesterone
- Hydroxyprogesterone acetate
- Hydroxyprogesterone caproate
- 63-05-8
- 63059-25-6
- 63059-29-0
- 63059-40-5
- 6305-95-9
- 6305-99-3
- 6306-00-9
- 630-60-4
- 6306-05-4
- 6306-34-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View