-
Name
Leuprorelin
- EINECS 633-395-9
- CAS No. 53714-56-0
- Article Data9
- CAS DataBase
- Density 1.44 g/cm3
- Solubility Soluble in water at 1mg/ml
- Melting Point
- Formula C59H84N16O12
- Boiling Point
- Molecular Weight 1209.42
- Flash Point
- Transport Information
- Appearance Fluffy solid.
- Safety 22-24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Luteinizinghormone-releasing factor (pig),6-D-leucine-9-(N-ethyl-L-prolinamide)-10-deglycinamide-;(D-Leu6,des-Gly-NH210)-LH-RH ethylamide;D-Leu6-des-Gly10-LH-releasing hormone ethylamide;Des-Gly10-[D-Leu6]-LH-releasing hormone ethylamide;Des-Gly10-[D-Leu6]LH-RHethylamide;1-9-Luteinizinghormone-releasing factor (swine), 6-D-leucine-9-(N-ethyl-L-prolinamide)-;Leuprorelin;Lupron SR;NSC 377526;
- PSA 466.34000
- LogP 3.44730
Synthetic route

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With methyl-phenyl-thioether; trifluoroacetic acid at 50℃; for 1h; Yield given; |
-
-
914200-76-3
pGlu-His(Trt)-Trp-Ser(tBu)-Tyr(tBu)-D-Leu-Leu-Arg(Pbf)-Pro-NHEt

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With chlorotriisopropylsilane; ethane-1,2-dithiol; trifluoroacetic acid at 20℃; for 2h; |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide pH=8.5; UV-irradiation; |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With chlorotriisopropylsilane; water; trifluoroacetic acid at 20 - 60℃; |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 20℃; for 2h; |
-
-
53714-56-0
Leuprolide

-
-
92557-81-8
6-carboxyfluorescein N-hydroxysuccinimide ester

| Conditions | Yield |
|---|---|
| With N,N,N′,N′-tetramethyl-N″-tert-butylguanidine; 5-carboxyfluorescein succinimidyl ester In N,N-dimethyl-formamide at 40℃; for 8h; Inert atmosphere; | 77% |

| Conditions | Yield |
|---|---|
| With collidine In water; acetonitrile at 25℃; for 0.416667h; | 69% |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 9h; Irradiation; Inert atmosphere; Sealed tube; chemospecific reaction; | 57% |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 9h; Irradiation; Inert atmosphere; Sealed tube; chemospecific reaction; | 43% |
-
-
1539-59-9
4-cyclohexyl-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylic acid diethyl ester

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 9h; Irradiation; Inert atmosphere; Sealed tube; chemospecific reaction; | 41% |
-
-
1539-32-8
4-isopropyl-2,6-dimethyl-1,4-dihydro-pyridine-3,5-dicarboxylic acid diethyl ester

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 9h; Irradiation; Inert atmosphere; Sealed tube; chemospecific reaction; | 40% |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 9h; Irradiation; Inert atmosphere; Sealed tube; chemospecific reaction; | 39% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 9h; Irradiation; Inert atmosphere; Sealed tube; chemospecific reaction; | 34% |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 9h; Irradiation; Inert atmosphere; Sealed tube; chemospecific reaction; | 33% |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 2,2,2-trifluoroethanol at 35℃; for 9h; Irradiation; Inert atmosphere; Sealed tube; chemospecific reaction; | 30% |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; pH=7.4; Chan-Lam Coupling; |

-
-
53714-56-0
Leuprolide

| Conditions | Yield |
|---|---|
| With Cu(II) salt In dimethyl sulfoxide at 20℃; pH=8.5; |

| Conditions | Yield |
|---|---|
| With silver(I) acetate; copper (I) acetate; acetic acid; N-acetylglycine In water at 60℃; for 12h; |
Leuprorelin Specification
1. Introduction of Leuprorelin
Leuprorelin is one kind of white to off-white powder. The IUPAC Name of this chemical is N-[1-[[1-[[1-[[1-[[1-[[1-[[5-(Diaminomethylideneamino)-1-[2-(ethylcarbamoyl)pyrrolidin-1-yl]-1-oxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-(4-hydroxyphenyl)-1-oxopropan-2-yl]amino]-3-hydroxy-1-oxopropan-2-yl]amino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]amino]-3-(1H-imidazol-5-yl)-1-oxopropan-2-yl]-5-oxopyrrolidine-2-carboxamide. Besides, Leuprorelin belongs Amino Acid Derivatives;Peptide.
Its Classification Code is Antineoplastic Agents; Antineoplastic agents, hormonal; Fertility Agents; Fertility agents, female; Hormone; Human Data; Reproductive Control Agents; Reproductive Effect. Leuprorelin can soluble in water.
2. Properties of Leuprorelin
Physical properties about Leuprorelin are:
(1)Index of Refraction: 1.681; (2)Molar Refractivity: 315.96 cm3; (3)Molar Volume: 834.5 cm3; (4)Surface Tension: 61.7 dyne/cm; (5)Storage Temp.: -20 °C; (6)XLogP3-AA: 0.7; (7)H-Bond Donor: 15; (8)H-Bond Acceptor: 16; (9)Rotatable Bond Count: 32; (10)Tautomer Count: 1000; (11)Exact Mass: 1208.645462; (12)MonoIsotopic Mass: 1208.645462; (13)Topological Polar Surface Area: 432; (14)Heavy Atom Count: 87.
3. Structure Descriptors of Leuprorelin
(1)Canonical SMILES: CCNC(=O)C1CCCN1C(=O)C(CCCN=C(N)N)NC(=O)C(CC(C)C)NC(=O)C(CC(C)C)NC(=O)(CC2=CC=C(C=C2)O)NC(=O)C(CO)NC(=O)C(CC3=CNC4=CC=CC=C43)NC(=O)C(CC5=CN=CN5)NC(=O)C6CCC(=O)N6
(2)InChIKey: GFIJNRVAKGFPGQ-UHFFFAOYSA-N
(3)Smiles: C1[C@@H]([N@@](CC1)[Ar]***[Se]**C([C@@H]1NC(=O)CC1)=O)C(NCC)=O
4. Safety information of Leuprorelin
Safety Statements of Leuprorelin (CAS NO.53714-56-0): 22-24/25
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
WGK Germany: 2
RTECS: OH6390000
5. Uses of Leuprorelin
Leuprorelin (CAS NO.53714-56-0) may be used in treating hormone-responsive cancers such as breast cancer or prostate cancer, estrogen-dependent conditions (such as endometriosis or uterine fibroids), to treat precocious puberty, and to control ovarian stimulation in In Vitro Fertilization (IVF). Leuprorelin is also under investigation for possible use in the treatment of mild to moderate Alzheimer's disease.
6. Production of Leuprorelin
(1)The reaction of phenol and phosgene to generate the phenyl chloroformate, and then tert-butanol, to generate the oxy acid benzyl ester of tert-butoxy. After the hydrazine, then nitrosated generates azido acid tert-butyl ester, and then reacted and leucineobtained leucine side chains.
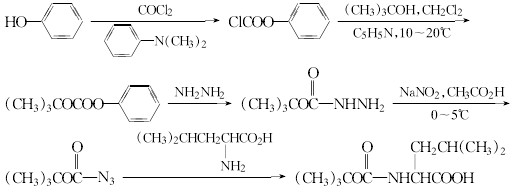
(2)Serine and benzyl chloride to protect the hydroxyl group, and then the azido carboxylic acid tert-butyl ester reaction serine side chain.
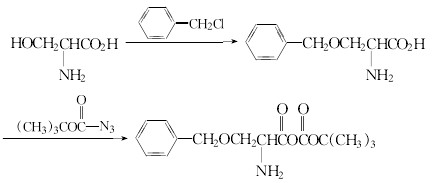
After the reaction of (1) or (2), then pick peptides and ethylamine reaction, hydrolysis or hydrogenolysis to wipe off the protection base and get the Leuprorelin.
Related Products
- Leuprorelin
- Leuprorelin acetate
- 5371-49-3
- 5371-52-8
- 537-15-5
- 5371-55-1
- 53715-64-3
- 53715-67-6
- 53716-49-7
- 53716-50-0
- 53716-53-3
- 53719-45-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View