-
Name
N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline
- EINECS 240-418-2
- CAS No. 16357-59-8
- Article Data13
- CAS DataBase
- Density 1.16 g/cm3
- Solubility insoluble in water
- Melting Point 62-67 °C(lit.)
- Formula C14H17NO3
- Boiling Point 355.9 °C at 760 mmHg
- Molecular Weight 247.294
- Flash Point 169 °C
- Transport Information
- Appearance White to pale yellow solid
- Safety 22-24/25-36/37/39-26-36
- Risk Codes 38-36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, Xn
Xn
- Synonyms 1-(Ethoxycarbonyl)-2-ethoxy-1,2-dihydroquinoline;2-Ethoxy-1(2H)-quinolinecarboxylic acid ethyl ester;2-Ethoxy-N-(ethoxycarbonyl)-1,2-dihydroquinoline;EEDQ;Ethyl2-ethoxy-1(2H)-quinolinecarboxylate;N-Carbethoxy-2-ethoxy-1,2-dihydroquinoline;NSC 147831;
- PSA 38.77000
- LogP 3.10390
Synthetic route
-
-
91-22-5
quinoline

-
-
64-17-5
ethanol

-
-
541-41-3
chloroformic acid ethyl ester

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In dichloromethane; water at 0℃; for 1.33333h; | 69% |

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With diethylzinc; palladium diacetate; triphenylphosphine In toluene at 20℃; for 4h; Catalytic behavior; Reagent/catalyst; Solvent; Inert atmosphere; regioselective reaction; | 100% |
-
-
107-10-8
propylamine

-
-
3050-37-1
indole-3-glycolic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 99% |

-
-
3050-37-1
indole-3-glycolic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 99% |

-
-
3050-37-1
indole-3-glycolic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 99% |
-
-
107-10-8
propylamine

-
-
617-73-2
2-hydroxy-octanoic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 99% |

-
-
107-10-8
propylamine

-
-
828-01-3
phenyllactic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 99% |

-
-
828-01-3
phenyllactic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 99% |
-
-
828-01-3
phenyllactic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
100-46-9
benzylamine

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 99% |

-
-
4224-69-5
methyl 2-(bromomethyl)propenoate

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| With indium; acetic acid In tetrahydrofuran for 0.5h; | 99% |
-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
128312-11-8
3-methylthiophenylboronic acid

-
-
1497411-35-4
C19H19NO2S

| Conditions | Yield |
|---|---|
| With methallylnickel chloride dimer; sodium phenoxide; (1R,7R)-9,9-dimethyl-2,2,6,6-tetra(napht-2-yl)-4-phenyl-3,5,8,10-tetraoxa-4-phosphabicyclo<5.3.0>decane In 1,4-dioxane; tert-Amyl alcohol at 20℃; for 25h; Suzuki Coupling; Inert atmosphere; enantioselective reaction; | 99% |
-
-
3050-37-1
indole-3-glycolic acid

-
-
142-84-7
di-n-propylamine

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 98% |

-
-
3050-37-1
indole-3-glycolic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
100-46-9
benzylamine

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 98% |

-
-
611-71-2, 611-72-3, 17199-29-0, 90-64-2
MANDELIC ACID

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
100-46-9
benzylamine

-
-
101730-29-4
ethoxycarbonyloxy-phenyl-acetic acid benzylamide

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 98% |

-
-
617-73-2
2-hydroxy-octanoic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 98% |

-
-
142-84-7
di-n-propylamine

-
-
611-71-2, 611-72-3, 17199-29-0, 90-64-2
MANDELIC ACID

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 97% |

-
-
107-10-8
propylamine

-
-
849585-22-4
LACTIC ACID

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 97% |

-
-
828-01-3
phenyllactic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 97% |

-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
166330-10-5
bis[2-(diphenylphosphino)phenyl] ether

| Conditions | Yield |
|---|---|
| Stage #1: bis(1,5-cyclooctadiene)nickel (0); bis[2-(diphenylphosphino)phenyl] ether In toluene at 23℃; for 0.333333h; Inert atmosphere; Stage #2: N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline In toluene for 1h; Inert atmosphere; | 97% |

-
-
13331-23-2
fur-2-ylboronic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 1h; | 96.4% |
-
-
828-01-3
phenyllactic acid

-
-
142-84-7
di-n-propylamine

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 96% |

-
-
13734-41-3
t-Boc-L-valine

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
98807-40-0
Boc-Val-O-COOEt

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one In dichloromethane at 23℃; for 2h; | 95% |
-
-
10303-64-7
2-hydroxy-4-methylpentanoic acid

-
-
142-84-7
di-n-propylamine

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 95% |

-
-
617-73-2
2-hydroxy-octanoic acid

-
-
142-84-7
di-n-propylamine

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 95% |

-
-
107-10-8
propylamine

-
-
10303-64-7
2-hydroxy-4-methylpentanoic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 95% |

-
-
10303-64-7
2-hydroxy-4-methylpentanoic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
100-46-9
benzylamine

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 95% |

-
-
617-73-2
2-hydroxy-octanoic acid

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
100-46-9
benzylamine

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 95% |


-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydrogencarbonate In tetrahydrofuran; water; ethyl acetate; acetonitrile | 95% |

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| With chlorine azide In water; toluene at 20℃; for 0.0188889h; Flow reactor; | 95% |
-
-
107-10-8
propylamine

-
-
611-71-2, 611-72-3, 17199-29-0, 90-64-2
MANDELIC ACID

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 94% |

-
-
611-71-2, 611-72-3, 17199-29-0, 90-64-2
MANDELIC ACID

-
-
16357-59-8
N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline

-
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| In dichloromethane for 5h; Ambient temperature; | 94% |

N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline Chemical Properties
Molar mass: 247.29 g/mol
Density: 1.16 g/cm3
Flash Point: 169 °C
Index of Refraction: 1.564
Boiling Point: 355.9 °C at 760 mmHg
Vapour Pressure: 3.04E-05 mmHg at 25°C
Melting point: 62-67 °C(lit.)
storage temp: 0-6°C
Water solubility: insoluble
Sensitive: Moisture Sensitive
Appearance: White to pale yellow solid
Structure of N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline(16357-59-8):
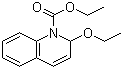
Synonyms: EEDQ;Ethyl 2-ethoxy-1(2H)-quinolinecarboxylate;1(2H)-quinolinecarboxylic acid, 2-ethoxy-, ethyl ester;2-Ethoxy-1(2H)-quinolinecarboxylic Acid Ethyl Ester;Ethyl-2-ethoxychinolin-1(2H)-carboxylat
Product Categories: Heterocycles;Coupling Reagent;Acids and Derivatives;Biochemistry;Condensation & Active Esterification;Synthetic Organic Chemistry;Coupling Reactions (Peptide Synthesis);Peptide Synthesis
N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline Toxicity Data With Reference
| 1. | ipr-mus LD50:32 mg/kg | JMCMAR Journal of Medicinal Chemistry. 14 (1971),49. |
N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline Consensus Reports
N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline Safety Profile
Hazard Codes: Xi
 ,Xn
,Xn
Risk Statements:
20: Harmful by inhalation
21: Harmful in contact with skin
22: Harmful if swallowed
36: Irritating to the eyes
37: Irritating to the respiratory system
38: Irritating to the skin
Safety Statements:
22: Do not breathe dust
24: Avoid contact with skin
25: Avoid contact with eyes
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37: Wear suitable gloves
39: Wear eye/face protection
N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline Specification
Skin: Get medical aid. Should flush skin with plenty of water immediatelyfor at least 15 minutes while removing contaminated clothing and shoes.
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids.Then get medical aid soon.
Inhalation: Remove from exposure and move to fresh air immediately.Give artificial respiration While not breathing. when breathing is difficult, give oxygen.And as soon as to get medical aid if cough or other symptoms appear.
Ingestion: Do not induce vomiting.If victim is conscious and alert, give 2-4 cupfuls of milk or water.If conscious drink water, then induce vomiting. If unconscious, immediately take victim to a physician and do not attempt to induce vomiting. Wash mouth out with water,and get medical aid immediately.
Notes to Physician: Treat supportively and symptomatically.
Stability and Reactivity of N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline(16357-59-8):
Incompatibilities with Other Materials: Strong oxidizing agents.
Conditions to Avoid: Incompatible materials.
Hazardous Decomposition Products: Nitrogen oxides, carbon dioxide,carbon monoxide.
Related Products
- N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline
- 16357-69-0
- 163578-99-2
- 163596-56-3
- 163596-75-6
- 163597-57-7
- 16361-24-3
- 1636-14-2
- 163619-04-3
- 163620-24-4
- 1636-27-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View