-
Name
TRIS(TRIMETHYLSILYL) PHOSPHATE
- EINECS 234-028-1
- CAS No. 10497-05-9
- Article Data40
- CAS DataBase
- Density 0.945 g/mL at 25 °C(lit.)
- Solubility 935.5μg/L at 20℃
- Melting Point 3-4 °C(lit.)
- Formula C9H27 O4 P Si3
- Boiling Point 228-229 °C720 mm Hg(lit.)
- Molecular Weight 314.541
- Flash Point 76 °F
- Transport Information UN 1993 3
- Appearance
- Safety Moderately toxic by ingestion. When heated to decomposition it emits toxic fumes of POx. See also ESTERS.
- Risk Codes 11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F
F Xi
Xi
- Synonyms Silanol,trimethyl-, phosphate (3:1) (8CI,9CI); Tris(trimethylsiloxy)phosphine oxide;Tris(trimethylsilyl) phosphate
- PSA 54.57000
- LogP 4.64900
Synthetic route
-
-
3385-94-2
hexamethyldisilathiane

-
A
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
B
-
20152-28-7
O,O,O-tris(trimethylsilyl) phosphorothioate

| Conditions | Yield |
|---|---|
| With phosphorus pentoxide at 170 - 180℃; for 10h; | A 85% B 98% |

-
-
75-77-4
chloro-trimethyl-silane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| In formamide for 1h; Inert atmosphere; Schlenk technique; | 98% |
-
-
107-46-0
Hexamethyldisiloxane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| Stage #1: Hexamethyldisiloxane With phosphoric acid at 160 - 170℃; under 4500.45 - 6000.6 Torr; for 5h; Autoclave; Reflux; Stage #2: N-trimethylsilyldihydropyrrole at 100℃; under 760.051 Torr; | 96.52% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With potassium dihydrogenphosphate In hexane Reflux; | 96% |
| With ammonium dihydrogen phosphate at 78℃; for 3h; Temperature; Reagent/catalyst; Time; | 89.6% |
| With phosphoric acid | |
| With phosphoric acid Schlenk technique; Inert atmosphere; | |
| With phosphoric acid Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With saccharin In acetonitrile for 1h; Heating; | 93% |
-
-
14468-90-7
N-trimethylsilyl-pyrrolidin-2-one

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With lithium dihydrogenphosphate at 20℃; for 1h; | 86% |
-
-
53380-41-9
tetrakis(trimethylsiloxy)phosphonium iodide

-
A
-
16029-98-4
trimethylsilyl iodide

-
B
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| In benzene Heating; during 5 h the solvent was distilled off, the temp. rose to 110 deg C; | A n/a B 85% |
-
-
1416767-26-4
C22H50O10P2SSi5

-
A
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
B
-
1416767-27-5
bis(trimethylsilyl) (1,1-dioxido-3Н-2,1-benzoxathiol-3-yl)phosphonate

| Conditions | Yield |
|---|---|
| at 90 - 110℃; under 1 Torr; | A n/a B 52% |
-
-
10416-59-8
N,O-bis-(trimethylsilyl)-acetamide

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 120℃; for 1h; | 26% |
-
-
1795-31-9
tris(trimethylsilyl) phosphite

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With trityl-glycidol at 130 - 150℃; | 24% |
| With styrene oxide at 130 - 150℃; |
-
-
999-97-3
1,1,1,3,3,3-hexamethyl-disilazane

-
A
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
B
-
18395-45-4
Tetrakis(trimethylsilyl) pyrophosphate

-
C
-
32284-31-4
1,1,3,5,7,7-hexakis(trimethylsilyl) pyrophosphate

| Conditions | Yield |
|---|---|
| With phosphorus pentoxide at 100 - 140℃; for 2h; Further byproducts given; | A 21% B 6% C 3% D 3% |

-
-
1825-61-2
Methoxytrimethylsilane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With phosphonic Acid; air |
-
-
107-46-0
Hexamethyldisiloxane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With phosphorus pentoxide | |
| With phosphorus pentoxide durch langsame Destillation; | |
| With phosphorus pentoxide In neat (no solvent) very slow distn. of (CH3)3SiOSi(CH3)3 over P2O5;; | |
| With P4O10 In neat (no solvent) very slow distn. of (CH3)3SiOSi(CH3)3 over P2O5;; |
-
-
18291-81-1
phosphoric acid, bis(trimethylsilyl)monomethyl ester

-
-
4551-15-9
phenylthiotrimethylsilane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With perhydrodibenzo-18-crown-6; potassium thiophenolate In benzene |
-
-
17882-98-3
phosphoric acid ethyl ester-bis-trimethylsilanyl ester

-
-
4551-15-9
phenylthiotrimethylsilane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With perhydrodibenzo-18-crown-6; potassium thiophenolate In benzene |
-
-
4551-15-9
phenylthiotrimethylsilane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With perhydrodibenzo-18-crown-6; potassium thiophenolate In benzene |
-
-
2754-27-0
trimethylsilyl acetate

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With phosphorus(V) oxybromide at 90℃; for 3h; |

| Conditions | Yield |
|---|---|
| durch langsame Destillation; |
-
-
999-97-3
1,1,1,3,3,3-hexamethyl-disilazane

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With ammonium hypophosphite |
-
-
30148-50-6
bis(trimethylsilyl)phosphonite

-
-
762-72-1
allyl-trimethyl-silane

-
A
-
3663-52-3
bis(trimethylsilyl) phosphonate

-
B
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
C
-
1219077-76-5
trimethylsilyl [3-(trimethylsilyl)propyl]phosphinate

-
D
-
1219077-75-4
bis(trimethylsilyl) [3-(trimethylsilyl)propyl]phosphonate

-
E
-
38049-93-3
trimethylsilylhypophosphite

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); water for 7h; Reflux; | A n/a B 0.1 g C 0.45 g D 0.12 g E n/a |

-
-
1795-31-9
tris(trimethylsilyl) phosphite

-
A
-
3663-52-3
bis(trimethylsilyl) phosphonate

-
B
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
C
-
18395-45-4
Tetrakis(trimethylsilyl) pyrophosphate

-
D
-
4414-25-9
Trimethylsilyldifluorophosphat

-
E
-
5668-94-0
bis(trimethylsilyl) phosphorofluoridate

| Conditions | Yield |
|---|---|
| With [1,3]-dioxolan-2-one; lithium hexafluorophosphate; water; Diethyl carbonate In [D3]acetonitrile at 20℃; Glovebox; Inert atmosphere; |

-
-
1795-31-9
tris(trimethylsilyl) phosphite

-
A
-
3663-52-3
bis(trimethylsilyl) phosphonate

-
B
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| With nickel(II) chloride hexahydrate at 160℃; for 42h; Schlenk technique; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| In hexane N2-atmosphere; addn. of soln. of GaMe3 (10% excess) to phosphate ester soln.; concn., crystd. on cooling (-24°C), collection (filtration), drying (vac.); elem. anal.; | 95% |
-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
420-56-4
trimethylsilyl fluoride

| Conditions | Yield |
|---|---|
| With sodium fluoride 150 to 200 deg C; | 91.5% |
-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
942422-03-9
(fluoro)bis(cyclopentadienyl)vanadium

-
-
1109-15-5
tris(pentafluorophenyl)borate

| Conditions | Yield |
|---|---|
| Stage #1: Tris(trimethylsilyl) phosphate; tris(pentafluorophenyl)borate In dichloromethane at 20℃; for 1h; Inert atmosphere; Glovebox; Stage #2: (fluoro)bis(cyclopentadienyl)vanadium In dichloromethane at 20℃; for 24h; Inert atmosphere; Glovebox; | 90% |

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
4648-54-8
trimethylsilylazide

| Conditions | Yield |
|---|---|
| With sodium azide at 150℃; for 0.166667h; | 87% |

| Conditions | Yield |
|---|---|
| With sodium azide at 150℃; for 0.166667h; other substrates, other reagents; | A 87% B n/a |

| Conditions | Yield |
|---|---|
| In hexane N2-atmosphere; addn. of soln. of AlMe3 (10% excess) to phosphate ester soln. at cooling, warming to room temp.; evapn. (vac.); elem. anal.; | 87% |
-
-
3663-52-3
bis(trimethylsilyl) phosphonate

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
6780-49-0
ethyl N-phenylformimidate

-
-
1227665-11-3
O,O',O'',O'''-tetra(trimethylsilyl) N-anilinomethylenebisphosphonate

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 150 - 160℃; for 1h; Reflux; | 86% |

-
-
17455-13-9
18-crown-6 ether

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
865-47-4
potassium tert-butylate

| Conditions | Yield |
|---|---|
| In 1,2-dimethoxyethane for 0.166667h; | 85% |

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
1331770-02-5
trimethylsilane adduct of trimethylsilylium tetrakis(pentafluorophenyl)borate

| Conditions | Yield |
|---|---|
| In toluene for 1h; Sonication; | 83% |

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran N2-atmosphere; refluxing for 5 h; elem. anal.; | 82% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran N2-atmosphere; refluxing for 5 h; evapn., recrystn. (hexane); elem. anal.; | 81% |

| Conditions | Yield |
|---|---|
| In hexane N2-atmosphere; addn. of soln. of AlMe3 (10% excess) to phosphate ester soln. at cooling, warming to room temp.; concn., crystd. on cooling (-24°C), collection (filtration), drying (vac.); elem. anal.; | 74% |

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| In toluene at 20℃; Schlenk technique; Inert atmosphere; | 69% |
-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
7550-45-0
titanium tetrachloride

| Conditions | Yield |
|---|---|
| In further solvent(s) boiling gently soln. of OP(OSi(CH3)3)3 (0.51 mmol) and TiCl4 (0.50 mmol) in ClSi(CH3)3; allowing to cool;; pptn.;; | 66% |
| In benzene-d6 byproducts: ClSi(CH3)3; briefly heating to boiling in benzene-d6;; not isolated, detected by 1H- and 31P-NMR;; |

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

| Conditions | Yield |
|---|---|
| In acetonitrile inert atmosphere; crystn. (40-50°C, 4 h, room temp., 2 d); elem. anal.; | 59% |
-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
50307-34-1
tert-butylaluminium dichloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran dropwise addn. of 1 equiv. P-compd. to Al-compd. (stirring), refluxing for 2 h; vol. reduction (vac.), crystn. (-20°C, overnight); elem. anal.; | 56% |
-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
22086-73-3
bis(tert-butyl)aluminium chloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran dropwise addn. of 1 equiv. P-compd. to Al-compd. (stirring), refluxing for 2 h; vol. reduction (vac.), crystn. (-20°C, overnight); elem. anal.; | 56% |

| Conditions | Yield |
|---|---|
| 150 to 200 deg C; | 45.5% |
-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
942422-03-9
(fluoro)bis(cyclopentadienyl)vanadium

-
-
1109-15-5
tris(pentafluorophenyl)borate

| Conditions | Yield |
|---|---|
| Stage #1: Tris(trimethylsilyl) phosphate; tris(pentafluorophenyl)borate In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; Glovebox; Stage #2: (fluoro)bis(cyclopentadienyl)vanadium In tetrahydrofuran at 20℃; for 48h; Inert atmosphere; Glovebox; | 43.2% |

-
-
117960-70-0
tris[μ-[2,6-bis(1-methylethyl)benzenaminato]]trimethyltricycloaluminum

-
-
10497-05-9
Tris(trimethylsilyl) phosphate

-
-
482315-48-0
(MeAl)[2,6-(i-Pr)2C6H3N]3(Al[OP(OSiMe3)3])2(O3POSiMe3)

| Conditions | Yield |
|---|---|
| In toluene under N2 atm. OP(OSiMe3)3 was added dropwise to alumazene in toluene andrefluxed for 24 h; react. mixt. was cooled to room temp.; elem. anal.; | 36% |
PHOSPHORIC ACID, TRIS(TRIMETHYLSILYL) ESTER Chemical Properties
IUPAC Name: Tris(trimethylsilyl) phosphate
Synonyms: Phosphoric acid tris(trimethylsilyl) ester ; Timtec-bb sbb001292 ; Tris(trimethylsilyl) phosphate ; Ester ; Phosphoric acid, tms ; Phosphoric acid, tristrimethylsilyl ; Phosphoric acid, tritms ; Silanol, trimethyl-, phosphate (3:1)
Product Categories: Si (Classes of Silicon Compounds);Silicon Compounds (for Synthesis);Silyl Esters;Si-O Compounds;Synthetic Organic Chemistry
CAS NO: 10497-05-9
Molecular Formula of Phosphoric acid, tris(trimethylsilyl) ester (CAS NO.10497-05-9) : C9H27O4PSi3
Molecular Weight of Phosphoric acid, tris(trimethylsilyl) ester (CAS NO.10497-05-9) :314.54
Molecular Structure of Phosphoric acid, tris(trimethylsilyl) ester (CAS NO.10497-05-9) :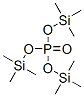
EINECS: 234-028-1
Mol File: 10497-05-9.mol
Index of Refraction: 1.414
Surface Tension: 20.7 dyne/cm
Density: 0.96 g/cm3
Flash Point: 110.8 °C
Enthalpy of Vaporization: 44.85 kJ/mol
Boiling Point: 230.8 °C at 760 mmHg
Vapour Pressure: 0.0977 mmHg at 25°C
Melting point: 3-4 °C(lit.)
PHOSPHORIC ACID, TRIS(TRIMETHYLSILYL) ESTER Toxicity Data With Reference
| 1. | orl-rat LD50:3440 mg/kg | MarJV# Personal Communication from Josef V. Marhold, VUOS, 539-18, Pardubice, Czechoslovakia, to the Editor of RTECS, Cincinnati, OH, March 29, 1977 29MAR77 . |
PHOSPHORIC ACID, TRIS(TRIMETHYLSILYL) ESTER Consensus Reports
Reported in EPA TSCA Inventory.
PHOSPHORIC ACID, TRIS(TRIMETHYLSILYL) ESTER Safety Profile
Moderately toxic by ingestion. When heated to decomposition it emits toxic fumes of POx. See also ESTERS.
Hazard Codes  F,
F, Xi
Xi
Risk Statements 11-36/37/38
R11:Highly flammable.
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements 16-26-36
S16:Keep away from sources of ignition.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
RIDADR UN 1993 3/PG 2
WGK Germany 2
RTECS TC9700000
F 10-21
TSCA Yes
HazardClass 3.2
PackingGroup III
Related Products
- Phosphoric acid
- Phosphoric acid diethyl ester 5-phenyl-isoxazol-3-yl ester
- Phosphoric acid dimethyl-p-(methylthio)phenyl ester
- Phosphoric acid lanthanum salt cerium terbium-doped
- Phosphoric acid magnesium strontium salt tin-doped
- Phosphoric acid trisodium salt
- Phosphoric acid, ammonium salt (1:3)
- Phosphoric acid, bariumsalt (1:1)
- Phosphoric acid, bis(2-chloropropyl) p-nitrophenyl ester
- Phosphoric acid, bis(3-chloropropyl) p-nitrophenyl ester
- 104-97-2
- 1049730-10-0
- 1049730-81-5
- 1049730-83-7
- 104974-44-9
- 104975-02-2
- 1049755-65-8
- 104-98-3
- 10498-35-8
- 104983-85-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View