-
Name
PERFLUOROOCTYL BROMIDE
- EINECS 207-028-4
- CAS No. 423-55-2
- Article Data11
- CAS DataBase
- Density 1.87 g/cm3
- Solubility Not miscible or difficult to mix in water.
- Melting Point 6 °C
- Formula C8BrF17
- Boiling Point 144 °C at 760 mmHg
- Molecular Weight 498.965
- Flash Point 40.9 °C
- Transport Information
- Appearance clear colorless liquid
- Safety 26-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Octane,1-bromoheptadecafluoro- (8CI);1-Bromoheptadecafluorooctane;1-Bromoperfluorooctane;AF 0104;AFO 150;FO 6167;Foralkyl Br 8;Imagent;Imagent BP;Oxygent;Oxygent CA;Oxygent HT;PFOB;Perflubron;Perfluoroctylbromide;Perfluorooctyl bromide;n-Perfluorooctyl bromide;
- PSA 0.00000
- LogP 6.34820
Synthetic route

| Conditions | Yield |
|---|---|
| 58% | |
| With carbon tetrabromide at 300℃; for 24h; | 40% |
-
-
507-63-1
1-iodoheptadecafluorooctane

-
-
558-13-4
carbon tetrabromide

-
-
7757-83-7
sodium sulfite

-
-
423-55-2
1-perfluorooctyl bromide

| Conditions | Yield |
|---|---|
| In 1,2-dibromo-3-fluorobenzene; water |

-
-
507-63-1
1-iodoheptadecafluorooctane

-
-
3115-68-2
tetrabutyl phosphonium bromide

-
-
423-55-2
1-perfluorooctyl bromide

| Conditions | Yield |
|---|---|
| In water |
-
-
507-63-1
1-iodoheptadecafluorooctane

-
-
149-44-0
rongalite

-
A
-
335-65-9
hydroperfluorooctane

-
B
-
423-55-2
1-perfluorooctyl bromide

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium bromide In N-methyl-acetamide; water |

-
-
21652-55-1
perfluoro-n-octylsulfopentafluoride

-
-
307-35-7
perfluorooctyl sulfofluorure

-
-
7726-95-6
bromine

-
-
423-55-2
1-perfluorooctyl bromide

| Conditions | Yield |
|---|---|
| 490°C , lead on aluminiumoxide tube filled with nickel turnings; |


| Conditions | Yield |
|---|---|
| at 500°C in aluminiumoxide-tube filled with nickel; |
-
-
40630-63-5
octanesulfonyl fluoride

-
-
423-55-2
1-perfluorooctyl bromide
-
-
71053-03-7
butyltetramethylene sulfone

-
-
423-55-2
1-perfluorooctyl bromide

| Conditions | Yield |
|---|---|
| With bromine at 140 - 142℃; Reflux; Large scale; |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
52112-19-3
n-C8H17TMS

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; magnesium In tetrahydrofuran | 99.8% |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
93324-65-3
1-methylaminopyrene hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 1-methylaminopyrene hydrochloride; Poly(acrylic acid), Mw = 1250000 With dicyclohexyl-carbodiimide In water; N,N-dimethyl-formamide at 60℃; for 2h; Polymerization; Stage #2: 1-perfluorooctyl bromide With sodium carbonate In water; N,N-dimethyl-formamide at 60℃; for 12h; Polymerization; Further stages.; | 99% |

-
-
423-55-2
1-perfluorooctyl bromide

-
-
93324-65-3
1-methylaminopyrene hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 1-methylaminopyrene hydrochloride; Poly(acrylic acid), Mw = 1250000 With dicyclohexyl-carbodiimide In water; N,N-dimethyl-formamide at 60℃; for 2h; Polymerization; Stage #2: 1-perfluorooctyl bromide With sodium carbonate In water; N,N-dimethyl-formamide at 60℃; for 12h; Polymerization; Further stages.; | 98% |

-
-
423-55-2
1-perfluorooctyl bromide

| Conditions | Yield |
|---|---|
| Stage #1: 1-perfluorooctyl bromide With diethylzinc In hexane at 20 - 77℃; for 3h; Inert atmosphere; Stage #2: dodecyl 2-iodobenzoate With copper(l) iodide In hexane at 90℃; for 13h; Inert atmosphere; | 94% |
| With copper(l) iodide; diethylzinc at 90℃; for 16h; Sealed tube; Inert atmosphere; Schlenk technique; | 94% |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
A
-
335-65-9
hydroperfluorooctane

-
B
-
63967-40-8
perfluorononanal

| Conditions | Yield |
|---|---|
| With lead(II) bromide; aluminium for 4h; Ambient temperature; | A n/a B 90.2% C n/a |

-
-
111-66-0
oct-1-ene

-
-
423-55-2
1-perfluorooctyl bromide

-
-
6145-05-7
1,1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8-heptadecafluorohexadecane

| Conditions | Yield |
|---|---|
| With tris(bipyridine)ruthenium(II) dichloride hexahydrate; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate In N,N-dimethyl-formamide for 12h; Reagent/catalyst; Irradiation; | 90% |
| With tris-(trimethylsilyl)silane; 1,1'-azobis(1-cyanocyclohexanenitrile) In water at 70℃; for 3h; Inert atmosphere; | 63% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; fluorine In sulfolane at 20 - 200℃; for 5.5h; Autoclave; Inert atmosphere; | 88.5% |
| With BrF2(1+)*(x)F6Sb; hydrogen fluoride at -40 - 24℃; Inert atmosphere; | 100 %Spectr. |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
93324-65-3
1-methylaminopyrene hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 1-methylaminopyrene hydrochloride; Poly(acrylic acid), Mw = 1250000 With dicyclohexyl-carbodiimide In water; N,N-dimethyl-formamide at 60℃; for 2h; Polymerization; Stage #2: 1-perfluorooctyl bromide With sodium carbonate In water; N,N-dimethyl-formamide at 60℃; for 12h; Polymerization; Further stages.; | 88% |

-
-
27845-47-2
N-benzylidene ethylamine

-
-
423-55-2
1-perfluorooctyl bromide

-
-
118825-48-2
N-Ethyl-2,2,3,3,4,4,5,5,6,6,7,7,8,8,9,9,9-heptedecafluoro-1-phenylnonylamine

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; methyllithium; lithium bromide In diethyl ether at -78℃; for 1h; | 86% |
-
-
423-55-2
1-perfluorooctyl bromide

-
A
-
335-65-9
hydroperfluorooctane

-
B
-
335-95-5
sodium perfluorooctanoate

| Conditions | Yield |
|---|---|
| With rongalite; sodium hydrogencarbonate In water; N,N-dimethyl-formamide at 85℃; for 20h; | A n/a B 86% |

| Conditions | Yield |
|---|---|
| With sodium methylate for 7h; | 81% |
| With 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride In tetrahydrofuran at 60℃; for 4h; | 77.5% |
-
-
292638-84-7
styrene

-
-
423-55-2
1-perfluorooctyl bromide

-
-
6145-09-1
(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluorodecyl)benzene

| Conditions | Yield |
|---|---|
| With tris-(trimethylsilyl)silane; 1,1'-azobis(1-cyanocyclohexanenitrile) In water at 70℃; for 3h; Inert atmosphere; | 81% |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
140-88-5
ethyl acrylate

-
A
-
335-65-9
hydroperfluorooctane

-
B
-
40599-19-7
Ethyl 4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,11-heptadecafluoroundecanoate

| Conditions | Yield |
|---|---|
| With [Co(HDm)2Py]Br; zinc In ethanol at 30℃; for 3h; | A n/a B 79% |

-
-
423-55-2
1-perfluorooctyl bromide

-
-
97-63-2
methyl methacrylate

-
A
-
335-65-9
hydroperfluorooctane

-
B
-
126331-02-0
Ethyl 4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,11-heptadecafluoro-2-methylundecanoate

| Conditions | Yield |
|---|---|
| With [Co(HDm)2Py]Br; zinc In ethanol at 30℃; for 3h; | A n/a B 78% |

-
-
423-55-2
1-perfluorooctyl bromide

-
-
68555-67-9
Sodium n-perfluorooctanesulfinate

| Conditions | Yield |
|---|---|
| With sodium disulfite In water; N,N-dimethyl-formamide at 80℃; for 6h; | 78% |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

-
-
1423439-31-9
(E)-1-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluoro-1-decen-1-yl)-4-methoxybenzene

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; copper(I) trifluoromethanesolfonate toluene complex; Dimethyl phosphite In acetonitrile at 80℃; for 72h; Schlenk technique; Inert atmosphere; Sealed tube; stereoselective reaction; | 78% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; caesium carbonate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In 1,2-dichloro-ethane at 80℃; for 24h; Heck Reaction; Schlenk technique; Inert atmosphere; | 75% |

| Conditions | Yield |
|---|---|
| With triethylamine; cobalt acetylacetonate In 1,1,1-trichloroethane at 25℃; for 10h; | 75% |

-
-
140-10-3
(E)-3-phenylacrylic acid

-
-
423-55-2
1-perfluorooctyl bromide

-
-
82497-73-2, 106873-72-7, 106873-83-0
(E)-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluoro-1-decen-1-yl)benzene

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; copper(I) trifluoromethanesolfonate toluene complex; Dimethyl phosphite In acetonitrile at 80℃; for 72h; Schlenk technique; Inert atmosphere; Sealed tube; stereoselective reaction; | 73% |

| Conditions | Yield |
|---|---|
| With o-phenylenebis(diphenylphosphine); cobalt(II) bromide; zinc In water; acetone at 20℃; for 3h; Inert atmosphere; stereoselective reaction; | 73% |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; tert.-butylhydroperoxide; cobalt(II) acetylacetonate In 1,1,1-trichloroethane; water at 80℃; for 4h; diastereoselective reaction; | 71% |

-
-
423-55-2
1-perfluorooctyl bromide

-
-
107-13-1
acrylonitrile

-
A
-
335-65-9
hydroperfluorooctane

-
B
-
26649-26-3
4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,11-heptadecafluoroundecanenitrile

| Conditions | Yield |
|---|---|
| With [Co(HDm)2Py]Br; zinc In ethanol at 30℃; for 4h; | A n/a B 70% |


| Conditions | Yield |
|---|---|
| With tris-(trimethylsilyl)silane; 1,1'-azobis(1-cyanocyclohexanenitrile) In water at 70℃; for 3h; Inert atmosphere; | 70% |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
78-94-4
methyl vinyl ketone

-
-
82486-22-4
5,5,6,6,7,7,8,8,9,9,10,10,11,11,12,12,12-heptadecafluorododecan-2-one

| Conditions | Yield |
|---|---|
| With tris-(trimethylsilyl)silane; 1,1'-azobis(1-cyanocyclohexanenitrile) In water at 70℃; for 3h; Inert atmosphere; | 70% |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
93324-65-3
1-methylaminopyrene hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 1-methylaminopyrene hydrochloride; Poly(acrylic acid), Mw = 1250000 With dicyclohexyl-carbodiimide In water; N,N-dimethyl-formamide at 60℃; for 2h; Polymerization; Stage #2: 1-perfluorooctyl bromide With sodium carbonate In water; N,N-dimethyl-formamide at 60℃; for 12h; Polymerization; Further stages.; | 68% |

-
-
423-55-2
1-perfluorooctyl bromide

-
-
1428264-89-4
2-isocyano-5-methyl-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With disodium hydrogenphosphate; fac-tris[2-phenylpyridinato-C2,N]iridium(III) In N,N-dimethyl-formamide at 26℃; for 48h; Inert atmosphere; Irradiation; | 67% |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
174312-75-5
diethyl 2-allyl-2-(2-phenylethyl)malonate

| Conditions | Yield |
|---|---|
| With fac-tris(2-phenylpyridinato-N,C2')iridium(III); silver benzoate; dimethyl sulfoxide at 25℃; for 24h; Schlenk technique; Inert atmosphere; Irradiation; | 66% |
-
-
423-55-2
1-perfluorooctyl bromide

-
-
33453-95-1
lithium p-toluenethiolate

-
-
138451-19-1
Perfluoro-n-octyl p-tolyl sulfide

| Conditions | Yield |
|---|---|
| With lithium perchlorate; triethylamine In N,N-dimethyl-formamide at 20℃; electrolysis; | 63% |

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride In benzene at 60℃; for 8h; | 60.4% |
Perfluorooctyl bromide Specification
The Perfluorooctyl bromide, with the CAS registry number 423-55-2, is also known as 1-Bromoheptadecafluorooctane. It belongs to the product categories of Fluorous Chemistry; Fluorous Solvents; Synthetic Organic Chemistry. Its EINECS registry number is 207-028-4. This chemical's molecular formula is C8BrF17 and molecular weight is 498.96. Its IUPAC name is called 1-bromo-1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluorooctane. This chemical's classification codes are Anti-obesity agents; Blood substitute; Central Nervous System Agents; Contrast media; Radiation-sensitizing agents. It is clear colorless liquid.
Physical properties of Perfluorooctyl bromide: (1)ACD/LogP: 8.62; (2) # of Rule of 5 Violations: 1 ; (3)ACD/LogD (pH 5.5): 8.62; (4) ACD/LogD (pH 7.4): 8.62 ; (5)ACD/BCF (pH 5.5): 1000000; (6) ACD/BCF (pH 7.4): 1000000 ; (7)ACD/KOC (pH 5.5): 1168259.25; (8) ACD/KOC (pH 7.4): 1168259.25; (9)#Freely Rotating Bonds: 6; (10)Index of Refraction: 1.296; (11) Molar Refractivity: 49.35 cm3; (12)Molar Volume: 266.7 cm3; (13)Surface Tension: 14.9 dyne/cm; (14) Density: 1.87 g/cm3; (15)Melting Point: 6 °C; (16)Flash Point: 40.9 °C; (17) Enthalpy of Vaporization: 36.53 kJ/mol ; (18)Boiling Point: 144 °C at 760 mmHg; (19) Vapour Pressure: 6.54 mmHg at 25°C.
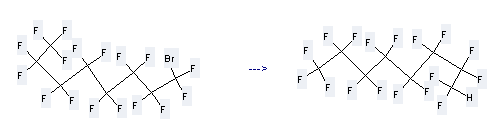
Uses of Perfluorooctyl bromide: it can be used to produce 8H-heptadecafluoro-octane at temperature of 60 °C. This reaction will need reagent n-Bu3SnH, AIBN and solvent tetrahydrofuran with reaction time of 4 hours. The yield is about 77.5%.
When you are using this chemical, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: C(C(C(C(C(F)(F)Br)(F)F)(F)F)(F)F)(C(C(C(F)(F)F)(F)F)(F)F)(F)F
(2)InChI: InChI=1S/C8BrF17/c9-7(22,23)5(18,19)3(14,15)1(10,11)2(12,13)4(16,17)6(20,21)8(24,25)26
(3)InChIKey: WTWWXOGTJWMJHI-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intravenous | 14720mg/kg (14720mg/kg) | Microvascular Research. Vol. 8, Pg. 320, 1974. | |
| mouse | LD50 | unreported | 42500mg/kg (42500mg/kg) | Biomaterials, Artificial Cells, and Artificial Organs. Vol. 16, Pg. 441, 1988. |
Related Products
- Perfluorooctyl bromide
- Perfluorooctyl iodide
- 423-56-3
- 4235-95-4
- 423-60-9
- 423-62-1
- 4236-41-3
- 423-65-4
- 42365-62-8
- 42365-68-4
- 42366-72-3
- 4236-73-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View