-
Name
Sodium diethyldithiocarbamate
- EINECS 205-710-6
- CAS No. 148-18-5
- Article Data23
- CAS DataBase
- Density 1.086 g/cm3
- Solubility water: >=10 g/100 mL at 14 °C
- Melting Point 95 °C
- Formula C5H10NNaS2
- Boiling Point 176.4 °C at 760 mmHg
- Molecular Weight 171.263
- Flash Point 60.5 °C
- Transport Information
- Appearance Odorless white or slightly brown or slightly pink crystals
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Sodium N,N-diethyldithiocarbamate;Carbamic acid, diethyldithio-, sodium salt;DEDC;Soxinol ESL;Diethyldithiocarbamic acid, sodium;Usaf ek-2596;DEDK;Ditiocarb sodium;Diethyldithiocarbamic acid, sodium salt;Thiocarb;GS 694A;NCI-C02835;DTC;DDC;
- PSA 60.63000
- LogP 1.81060
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water cooling; | 90% |
| With sodium hydroxide In ethanol at 20 - 40℃; | 73% |
| With sodium hydroxide In water at 20℃; for 1h; Cooling with ice; | 69% |
-
-
97-77-8
disulfiram

-
-
140-90-9
sodium O-ethyl dithiocarbonate

-
A
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
B
-
327989-09-3
C8H15NOS4

| Conditions | Yield |
|---|---|
| With sodium nitrate; C8MoN8(3-)*3Na(1+) In water; acetone at 25℃; Rate constant; Equilibrium constant; μ 0.2 mol/l; |

-
-
97-77-8
disulfiram

-
-
4092-82-4
sodium N,N'-diisopropyldithiocarbamate

-
A
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| With sodium nitrate; potassium hexacyanoferrate(III) In water; acetone at 25℃; Equilibrium constant; μ 0.2 mol/l; |

-
-
140-90-9
sodium O-ethyl dithiocarbonate

-
-
327989-09-3
C8H15NOS4

-
A
-
502-55-6
bis-ethoxythiocarbonyldisulfane

-
B
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| With sodium nitrate; C8MoN8(3-)*3Na(1+) In water; acetone at 25℃; Rate constant; Equilibrium constant; μ 0.2 mol/l; |

-
-
4092-82-4
sodium N,N'-diisopropyldithiocarbamate

-
A
-
4136-91-8
tetraisopropylothiuram disulphide

-
B
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| With sodium nitrate; potassium hexacyanoferrate(III) In water; acetone at 25℃; Equilibrium constant; μ 0.2 mol/l; |

-
-
138523-63-4
Tributyltin diethyldithiocarbamate

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| With sodium hydrogensulfide In methanol at 4℃; |
-
-
80-05-7
BPA

-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
120-32-1
chlorophene

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| Stage #1: sodium N,N-diethyldithiocarbamate; 5'-I-d(TTT)-CPG resin In N,N-dimethyl-formamide at 20℃; for 0.0333333h; Stage #2: With ammonium hydroxide at 60℃; for 17h; | 100% |

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane at 20℃; for 1h; | 100% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
572-09-8
2,3,4,6-tetra-O-acetyl-α-D-glucopyranosyl bromide

| Conditions | Yield |
|---|---|
| In acetone for 1h; Reflux; | 99% |
| In acetonitrile at 25℃; for 1h; | 63% |

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In solid byproducts: Et4NBr, NaBr; mixt. was evacuated at 80 °C for 2 h, mech. activation for 1.5 h in a vac. vibration mill with steel ball (23 Hz); H2O was added, ppt. was washed with hot H2O and EtOH, dried in vac. at 75 °C, residue was treated with hot MeCN, washed with H2O and EtOH, dried in vac. at 75 °C, recrystn. from DMSO, elem. anal.; | 99% |
-
-
1243627-92-0
[AuCl(2-thia-1,3,5-triaza-7-phosphaadamantane-2,2-dioxide)]

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
1243628-11-6
[Au(diethyldithiocarbamate)(2-thia-1,3,5-triaza-7-phosphaadamantane-2,2-dioxide)]

| Conditions | Yield |
|---|---|
| In methanol Na salt was added to suspn. of Au complex in MeOH; stirred for 18 h; filtered; washed (H2O, MeOH, Et2O); dried in air; elem. anal.; | 99% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 12h; stereoselective reaction; | 99% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
112297-29-7
7-[(4-methylphenyl)sulfonyl]-7-azabicyclo[4.1.0]hept-3-ene

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 0.5h; stereoselective reaction; | 99% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
121995-12-8
2-(N-acetylanilino)-4-iodomethyl-1,3-selenazole

| Conditions | Yield |
|---|---|
| In acetone for 0.5h; Heating; | 98% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
13681-87-3
bis(N,N-diethyldithiocarbamato)copper(II)

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) byproducts: NaCl; placing of CuCl2 and NaS2CNEt2 in 1:2 ratio to the reactor; sealing, vibrating for 60 min; sublimating in vac. at 180-190°C; crystn.; elem. anal.; | 98% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
16894-68-1
tetraphenylchloroantimony(V)

-
-
109423-89-4
tetraphenylantimony N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In water byproducts: NaCl; soln. of ligand in water was added to soln. of Sb-complex in water; filtered off, dried, crystd. from toluene-heptane; | 98% |
-
-
1112152-67-6
2-(bromomethyl)-1,3-thiaselenole

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 0.0333333h; regioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: NaBr; under inert atm.; addn. of soln. of Na-compd. to soln. of complex at -78°C, removed from cold bath and stirred at ambient temp. for 1.45 h; evapd. (vac.), dissolved in CH2Cl2, addn. of NEt4Cl, stirred for 45 min, filtered (cellulose), dried (vac., 2.5 h), treated with THF, cooled to -8°C for 30 min, supernatant decanted, , washed (THF), recrystd. (CH2Cl2/Et2O); | 97.5% |

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
7646-85-7
zinc(II) chloride

-
-
14324-55-1
bis(N,N-diethyldithiocarbamato)zinc(II)

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) byproducts: NaCl; placing of ZnCl2 and NaS2CNEt2 in 1:2 ratio to the reactor; sealing, vibrating for 10 min; sublimating at 150-165°C in vac.; crystn.; elem. anal.; | 97% |
-
-
13965-31-6
dichloro(2,2'-bipyridine)platinum(II)

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
15730-38-8
bis(N,N-diethyldithiocarbamate)platinum(II)

| Conditions | Yield |
|---|---|
| In not given stoich. amts., stirring for 1 h (pptn.); collection (filtration), washing (water), drying (vac.); | 97% |

| Conditions | Yield |
|---|---|
| In chloroform equimolar amts. at 25°C; | 97% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
5035-52-9
dichlorophenylstibine

-
-
18615-16-2
phenylantimony diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In chloroform equimolar amts. of educts at 25°C; | 97% |

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In dichloromethane (inert atmosphere); stirring (12 h); filtration (Celite), solvent removal (vac.), washing (light petroleum); elem. anal.; | 97% |

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 12h; stereoselective reaction; | 97% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 0.166667h; stereoselective reaction; | 97% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
81097-48-5
N-tosyl-6-azabicyclo[3.1.0]hexane

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 1h; stereoselective reaction; | 97% |

| Conditions | Yield |
|---|---|
| In acetone at 40℃; for 1h; | 97% |

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; for 6h; | 97% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 97% |
-
-
61272-76-2
4-fluoro-2-iodoaniline

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| With copper diacetate; potassium carbonate In N,N-dimethyl-formamide at 120℃; for 6h; Sealed tube; | 97% |
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
10108-64-2
cadmium(II) chloride

-
-
14239-68-0
cadmium(II) diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) byproducts: NaCl; placing of CdCl2 and NaS2CNEt2 in 1:2 ratio to the reactor; sealing, vibrating for 60 min; sublimating in vac. at 180-230°C; crystn.; elem. anal.; | 96.6% |
| In water byproducts: NaCl; pptn. of complex from soln. at room temp., filtration while hot, drying (80°C, vac.); recrystn. (benzene); (1)H- and (13)C-NMR; | |
| In water byproducts: NaCl; ZnCl2, the thiocarbamate in water, immediately pptn.; filtered hot to remove NaCl and any excess of unreacted thiocarbamate, dried (vac. at 80°C), recrystn. (boiling benzene); | |
| In water solution of CdCl2 added to stirred solution of the ligand; filtered, dried, recrystd. from chloroform; |

| Conditions | Yield |
|---|---|
| With 1,3,5-trichloro-2,4,6-triazine; dimethyl sulfoxide In dichloromethane at 20℃; for 2h; | 96% |
| With iodine In methanol at 0℃; | 95% |
| With oxygen; sodium hydroxide In water at 25℃; for 1.08333h; pH=10; Kinetics; pH-value; Reagent/catalyst; | 94% |
-
-
760-90-7
dimethyl 2-bromosuccinate

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
94725-95-8
dimethyl (diethylthiocarbamoylthio)succinate

| Conditions | Yield |
|---|---|
| In water for 8h; | 96% |
-
-
600-00-0
ethyl 2-bromoisobutyrate

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
120924-70-1
S-(1-methyl-1-ethoxycarbonylethyl) N,N-diethyl-dithiocarbamate

| Conditions | Yield |
|---|---|
| In acetone at 40℃; | 96% |

| Conditions | Yield |
|---|---|
| In acetone at 40℃; | 96% |
| With water In N,N-dimethyl-formamide at 75 - 80℃; | 88% |
-
-
700840-90-0
3-benzyltetrahydro-1,3-oxazine-2-thione

-
-
148-18-5
sodium N,N-diethyldithiocarbamate

- polymer, product of cationic ring-opening polymerization, Mn 20400 Da by SEC in THF, Mn 12200 by light scattering, PDI 1.04; monomer(s): 3-benzyltetrahydro-1,3-oxazine-2-thione; sodium N,N-diethyldithiocarbamate
-
polymer, product of cationic ring-opening polymerization, Mn 20400 Da by SEC in THF, Mn 12200 by light scattering, PDI 1.04; monomer(s): 3-benzyltetrahydro-1,3-oxazine-2-thione; sodium N,N-diethyldithiocarbamate

| Conditions | Yield |
|---|---|
| Stage #1: 3-benzyltetrahydro-1,3-oxazine-2-thione With boron trifluoride diethyl etherate In nitrobenzene at 50℃; for 24h; Stage #2: sodium N,N-diethyldithiocarbamate In nitrobenzene; acetonitrile | 96% |
Sodium diethyldithiocarbamate Chemical Properties
IUPAC Name: sodium N,N-diethylcarbamodithioate
Following is the structure of Sodium diethyldithiocarbamate (CAS NO.148-18-5):
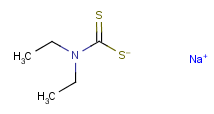
Molecular formula: C5H10NNaS2
Molecular Weight: 171.26
EINECS: 205-710-6
Melting point: 95°C
Flash Point: 60.5 °C
Index of Refraction: 1.555
Molar Refractivity: 44.12 cm3
Molar Volume: 137.3 cm3
Density: 1.086 g/cm3
Surface Tension: 44.6 dyne/cm
Boiling Point: 176.4 °C at 760 mmHg
Enthalpy of Vaporization: 41.27 kJ/mol
Vapour Pressure: 1.1 mmHg at 25 °C
Water Solubility of Sodium diethyldithiocarbamate (CAS NO.148-18-5): >=10 g/100 mL at 14 °C
Product Categories of Sodium diethyldithiocarbamate (CAS NO.148-18-5): Pharmaceutical Intermediates; Aliphatics
Canonical SMILES: CCN(CC)C(=S)[S-].[Na+]
InChI: InChI=1S/C5H11NS2.Na/c1-3-6(4-2)5(7)8;/h3-4H2,1-2H3,(H,7,8);/q;+1/p-1
InChIKey: IOEJYZSZYUROLN-UHFFFAOYSA-M
Sodium diethyldithiocarbamate Uses
Sodium diethyldithiocarbamate (CAS NO.148-18-5) can be used as a pesticide.
Sodium diethyldithiocarbamate Toxicity Data With Reference
| 1. | oms-omi 100 µmol/L | BBACAQ Biochimica et Biophysica Acta. 519 (1978),65. | ||
| 2. | dnd-hmn:hla 100 µmol/L | BBACAQ Biochimica et Biophysica Acta. 519 (1978),65. | ||
| 3. | cyt-rat-orl 5200 mg/kg | MUREAV Mutation Research. 53 (1978),212. | ||
| 4. | dni-ckn:emb 4 µmol/L | BBACAQ Biochimica et Biophysica Acta. 519 (1978),65. | ||
| 5. | oms-ckn:emb 40 µmol/L | BBACAQ Biochimica et Biophysica Acta. 519 (1978),65. | ||
| 6. | orl-rat LD50:1500 mg/kg | DRFUD4 Drugs of the Future. 6 (1981),225. | ||
| 7. | ipr-rat LD50:1250 mg/kg | DRFUD4 Drugs of the Future. 6 (1981),225. | ||
| 8. | orl-mus LD50:1500 mg/kg | DRFUD4 Drugs of the Future. 6 (1981),225. | ||
| 9. | ipr-mus LD50:1302 mg/kg | DRFUD4 Drugs of the Future. 6 (1981),225. | ||
| 10. | scu-rbt LD50:500 mg/kg | APTOA6 Acta Pharmacologica et Toxicologica. 8 (1952),329. |
Sodium diethyldithiocarbamate Consensus Reports
IARC Cancer Review: Group 3 IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 7 ,1987,p. 56.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Inadequate Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 12 ,1976,p. 217.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) . NCI Carcinogenesis Bioassay (feed); No Evidence: mouse, rat NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NCI-CG-TR-172 ,1979. . Reported in EPA TSCA Inventory.
Sodium diethyldithiocarbamate Safety Profile
Moderately is toxic by ingestion, intraperitoneal, and subcutaneous routes. Experimental reproductive effects. Questionable carcinogen with experimental neoplastigenic and teratogenic data. Human mutation data reported. When heated to decomposition it emits very toxic fumes of NOx, SOx, and Na2O. See also CARBAMATES.
Sodium diethyldithiocarbamate Standards and Recommendations
DFG MAK: 2 mg/m3
Sodium diethyldithiocarbamate Specification
Sodium diethyldithiocarbamate , its cas register number is 148-18-5. It also can be called Diethyldithiocarbamic acid sodium salt ; N,N-Diethyldithiocarbamic acid, sodium salt ; and Sodium N,N-diethyldithiocarbamate . Its classification code are Agricultural Chemical; Antidote for nickel and cadmium poisoning; Fungicide, bactericide, wood preservative; Mutation data ; Reproductive Effect and Tumor data.
Related Products
- Sodium
- Sodium (+)-10-camphorsulfonate
- Sodium (C10-16)alkylbenzenesulfonate
- Sodium 2-(4-methoxyphenoxy)propionate
- Sodium 2-(naphthalen-1-ylcarbamoyl)benzoate
- Sodium 2,4-dinitrophenate
- Sodium 2,5-difluorobenzoate
- Sodium 2-bromoethanesulphonate
- Sodium 2-carboxy-4-sulfobenzoate
- Sodium 2-chloro-5-nitrobenzenesulfonate
- 1481-93-2
- 148204-28-8
- 148204-33-5
- 148204-34-6
- 148214-56-6
- 148214-90-8
- 148217-40-7
- 148219-53-8
- 148229-78-1
- 14823-31-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View