 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
-
Name
Sodium 2,4-dinitrophenate
- EINECS 213-786-7
- CAS No. 1011-73-0
- Article Data13
- CAS DataBase
- Density
- Solubility
- Melting Point
- Formula C6H3N2NaO5
- Boiling Point 312.1 °C at 760 mmHg
- Molecular Weight 206.09
- Flash Point 142.8 °C
- Transport Information UN 1321
- Appearance Brown Yellow Flake Crystal
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Phenol,2,4-dinitro-, sodium deriv. (6CI);Phenol, 2,4-dinitro-, sodium salt (8CI,9CI);2,4-Dinitrophenol sodium salt;Sodium 2,4-dinitrophenate;Sodium2,4-dinitrophenol;Sodium 2,4-dinitrophenoxide;
- PSA 114.70000
- LogP 2.69320
Synthetic route

| Conditions | Yield |
|---|---|
| With Cumene hydroperoxide; cumene hydroperoxide sodium salt In toluene Ambient temperature; | 90% |
| With sodium hydroxide In water Rate constant; variation of surfactants; | |
| With sodium hydroxide In water; dimethyl sulfoxide at 25℃; Rate constant; Mechanism; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 25℃; Rate constant; Mechanism; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 25℃; Rate constant; Mechanism; Equilibrium constant; |
-
-
75735-29-4
(E)-O-(2,4-dinitrophenyl)benzaldoxime

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
B
-
100-47-0
benzonitrile

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Rate constant; Kinetics; Mechanism; isotope effect; other (E)-O-arylbenzaldoximes; |
-
-
75735-26-1
O-(2,4-dinitrophenyl)-p-nitrobenzaldoxime

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
B
-
619-72-7
4-nitrobenzonitrile

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Rate constant; |
-
-
115828-58-5
4-methoxy-benzaldehyde-[O-(2,4-dinitro-phenyl)-seqtrans-oxime ]

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
B
-
874-90-8
4-methoxybenzonitrile

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Rate constant; |
-
-
70-34-8
2,4-Dinitrofluorobenzene

-
-
100-51-6
benzyl alcohol

-
A
-
2734-78-3
1-(benzyloxy)-2,4-dinitrobenzene

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In octane; water at 25℃; |

-
-
71-23-8
propan-1-ol

-
-
75-85-4
tert-Amyl alcohol

-
-
71-41-0
pentan-1-ol

-
-
70-34-8
2,4-Dinitrofluorobenzene

-
-
75-65-0
tert-butyl alcohol

-
A
-
33696-26-3
tert-Butyl-2,4-dinitrophenylether

-
B
-
37445-91-3
1,1-Dimethylpropyl-2,4-dinitrophenolat

-
C
-
10242-17-8
1-propoxy-2,4-dinitrobenzene

-
D
-
13417-45-3
(2,4-dinitro-phenyl)-pentyl ether

-
E
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sodium hydroxide; cetyltrimethylammonim bromide In octane; water at 25℃; Kinetics; Product distribution; |

-
-
115828-59-6
3-Bromo-benzaldehyde O-(2,4-dinitro-phenyl)-oxime

-
A
-
6952-59-6
3-cyanobromobenzene

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Rate constant; |

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Rate constant; |
-
-
97-00-7
1-chloro-2,4-dinitro-benzene

-
-
71-36-3
butan-1-ol

-
A
-
13417-44-2
n-Butyl 2,4-dinitrophenyl ether

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With cetyltrimethylammonim bromide In octane; water at 25℃; Kinetics; Product distribution; |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In water Rate constant; variation of surfactants and hydroxide ion concentration; | |
| With hydroxide In water at 25℃; Rate constant; variation of surfactants and hydroxide ion concentration; | |
| With sodium hydroxide In water; dimethyl sulfoxide at 25℃; Rate constant; Mechanism; Equilibrium constant; |
-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
97-00-7
1-chloro-2,4-dinitro-benzene

-
A
-
1670-17-3
N,N-dimethyl-2,4-dinitroaniline

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydride; 5-amino-3-methylthio-1,2,4-triazole 1.) 0 deg C, 1 h, 2.) 60 deg C, 2 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
45534-08-5
5-amino-3-methylthio-1,2,4-triazole

-
-
97-00-7
1-chloro-2,4-dinitro-benzene

-
A
-
1670-17-3
N,N-dimethyl-2,4-dinitroaniline

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
C
-
82117-99-5
5-amino-1-(2,4-dinitrophenyl)-3-methylthio-1H-1,2,4-triazole

| Conditions | Yield |
|---|---|
| With sodium hydride 1.) DMF, 0 deg C, 1 h, 2.) DMF, 60 deg C, 2 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
45534-08-5
5-amino-3-methylthio-1,2,4-triazole

-
-
97-00-7
1-chloro-2,4-dinitro-benzene

-
A
-
1670-17-3
N,N-dimethyl-2,4-dinitroaniline

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydride 1.) DMF, 0 deg C, 1 h, 2.) DMF, 60 deg C, 2 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
45534-08-5
5-amino-3-methylthio-1,2,4-triazole

-
-
97-00-7
1-chloro-2,4-dinitro-benzene

-
A
-
1670-17-3
N,N-dimethyl-2,4-dinitroaniline

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydride 1.) DMF, 0 deg C, 1 h, 2.) DMF, 60 deg C, 2 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
97-00-7
1-chloro-2,4-dinitro-benzene

-
A
-
1670-17-3
N,N-dimethyl-2,4-dinitroaniline

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydride; 5-amino-3-methylthio-1,2,4-triazole 1.) DMF, 0 deg C, 1 h, 2.) DMF, 60 deg C, 2 h; Yield given. Multistep reaction. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 25℃; Rate constant; Equilibrium constant; other alkali metal hydroxides; |

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In dimethyl sulfoxide at 25℃; Rate constant; Equilibrium constant; other alkali metal hydroxides; |
-
-
191226-78-5
1-(2,4-dinitrophenyl)-1-aza-4,7,10,13-tetraoxapentadecane

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 25℃; Rate constant; Equilibrium constant; other alkali metal hydroxides; |

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 25℃; Rate constant; Equilibrium constant; other alkali metal hydroxides; |

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| In neat (no solvent) Product distribution; Mechanism; Heating; thermal stability; |
-
-
205432-08-2, 351405-62-4
O2-(2,4-dinitrophenyl) 1-(N,N-diethylamino)diazen-1-ium-1,2-diolate

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 25℃; Kinetics; Thermodynamic data; Further Variations:; Temperatures; |

-
B
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With ethylenediaminetetraacetic acid; sodium taurocholate at 37℃; for 0.75h; Kinetics; aq. phosphate buffer; |

-
-
18962-97-5
bis(2,4-dinitrophenyl)phosphate

-
A
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With C38H45N4O6Zn2(1+)*ClO4(1-); sodium perchlorate; water In acetonitrile at 25℃; pH=7.5; Kinetics; Reagent/catalyst; pH-value; Glovebox; Schlenk technique; |

| Conditions | Yield |
|---|---|
| Stage #1: 4BF4(1-)*C86H162N6O2(4+) With cobalt(II) acetate In ethanol at 20℃; for 3h; Stage #2: 2,4-Dinitrophenol With oxygen In dichloromethane for 3h; Stage #3: sodium 2,4-dinitrophenoxide With oxygen In dichloromethane Product distribution / selectivity; | 100% |

-
-
51-28-5
2,4-Dinitrophenol

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| Stage #1: C36H58N4O2(2+)*2BF4(1-); cobalt(II) acetate In ethanol at 20℃; for 2h; Glovebox; Inert atmosphere; Schlenk technique; Stage #2: 2,4-Dinitrophenol With oxygen In dichloromethane for 2h; Stage #3: sodium 2,4-dinitrophenoxide In dichloromethane at 20℃; | 100% |

-
-
51-28-5
2,4-Dinitrophenol

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| Stage #1: C36H52N4O2(2+)*2BF4(1-); cobalt(II) acetate In ethanol at 20℃; for 2h; Glovebox; Inert atmosphere; Schlenk technique; Stage #2: 2,4-Dinitrophenol With oxygen In dichloromethane for 2h; Stage #3: sodium 2,4-dinitrophenoxide In dichloromethane at 20℃; | 100% |


| Conditions | Yield |
|---|---|
| With 5%-palladium/activated carbon; hydrogen at 70℃; under 4500.45 Torr; for 2h; Temperature; Pressure; Time; Autoclave; | 97.1% |
-
-
57933-83-2
Isopropenyl chloroformate

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
96916-43-7
Carbonic acid 2,4-dinitro-phenyl ester isopropenyl ester

| Conditions | Yield |
|---|---|
| In acetonitrile Ambient temperature; | 87% |
-
-
51-28-5
2,4-Dinitrophenol

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| Stage #1: C38H62N4O2(2+)*2BF4(1-); cobalt(II) acetate In methanol at 20℃; for 2h; Inert atmosphere; Schlenk technique; Glovebox; Stage #2: 2,4-Dinitrophenol With oxygen In dichloromethane for 2h; Schlenk technique; Glovebox; Stage #3: sodium 2,4-dinitrophenoxide In dichloromethane at 20℃; Schlenk technique; Glovebox; | 85.84% |

-
-
51-28-5
2,4-Dinitrophenol

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| Stage #1: C34H56N4O2(2+)*2BF4(1-); cobalt(II) acetate In methanol at 20℃; for 6h; Inert atmosphere; Schlenk technique; Glovebox; Stage #2: 2,4-Dinitrophenol With oxygen In dichloromethane at 20℃; for 12h; Inert atmosphere; Schlenk technique; Glovebox; Stage #3: sodium 2,4-dinitrophenoxide In dichloromethane for 24h; Inert atmosphere; Schlenk technique; Glovebox; | 82.2% |


-
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| In water Co complex dissolved in hot H2O by stirring; mixed with soln. of Na saltof ligand in hot H2O with stirring; ppt. washed with ice-cold H2O; air dried; elem. anal.; | 79% |

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| In water byproducts: NaCl; addn. of soln. of phenolate deriv. to aq. soln. of cobalt compd.; filtration, washing with cold water, drying in air, recrystn. by keepingsoln. in water and acetone at room temp. for 2 ds, drying in air, elem. anal.; | 75% |
-
-
1011-73-0
sodium 2,4-dinitrophenoxide


-
-
207975-84-6
2,4-dinitrophenyl-3,4,6-tri-O-acetyl-2-fluoro-2-deoxy-β-D-galactopyranoside

| Conditions | Yield |
|---|---|
| In acetonitrile Heating; | 61% |

| Conditions | Yield |
|---|---|
| N,N,N,N,-tetramethylethylenediamine In benzene at 25℃; for 4h; | 50% |
| N,N,N,N,-tetramethylethylenediamine In benzene at 25℃; for 4h; Rate constant; different mol ratio of reactants and various solublizing agent; in the presence and absence of TEMEDA or tertiary amine as phase transfer catalysts.; | 50% |
| With Aliquat 336 In benzene at 25℃; Rate constant; | |
| With didecyldimethylammonium chloride In benzene at 25℃; Rate constant; | |
| With tetrabutyl-ammonium chloride In benzene at 25℃; Rate constant; |
-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
63752-85-2
N-(4-Chlor-phenylsulfonyl)iminothiokohlensaeuremethylester-chlorid

-
-
108428-77-9
4-Chloro-N-[1-(2,4-dinitro-phenoxy)-1-methylsulfanyl-meth-(Z)-ylidene]-benzenesulfonamide

| Conditions | Yield |
|---|---|
| In benzene for 4h; Heating; | 46% |
-
-
2973-83-3
N-(p-Toluensulfonyl)iminothiokohlensaeuremethylester-chlorid

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
108428-71-3
N-[1-(2,4-Dinitro-phenoxy)-1-methylsulfanyl-meth-(Z)-ylidene]-4-methyl-benzenesulfonamide

| Conditions | Yield |
|---|---|
| In benzene for 4h; Heating; | 41% |
-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
51164-80-8
5-Acetyl-4-chloro-6-methyl-2-phenyl-pyrimidine

-
-
133761-08-7
1-[4-(2,4-Dinitro-phenoxy)-6-methyl-2-phenyl-pyrimidin-5-yl]-ethanone

| Conditions | Yield |
|---|---|
| In ethanol for 2h; Heating; | 40% |
-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
4225-84-7
2-chloro-2-oxo-cis-4,6-dimethyl-1,3,2λ5-dioxaphosphorinane

-
-
132155-38-5
2-(2,4-dinitrophenoxy)-2-oxo-cis-4,6-dimethyl-1,3,2λ5-dioxaphosphorinane

| Conditions | Yield |
|---|---|
| In toluene at 90℃; for 0.0833333h; | 30.1% |
-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
3068-34-6
Acetic acid (2R,3S,4R,5R,6R)-3-acetoxy-2-acetoxymethyl-5-acetylamino-6-chloro-tetrahydro-pyran-4-yl ester

-
-
52621-80-4
2,4-dinitrophenyl 2-acetamido-3,4,6-tri-O-acetyl-2-deoxy-D-glucopyranoside

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 64h; Ambient temperature; | 9% |
-
-
75-44-5
phosgene

-
-
2475-71-0
N,N-bis(2-chloro-n-propyl)amine

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
13723-42-7
N,N-Bis-(2-chlorpropyl)-2,4-dinitrophenyl-carbamat

| Conditions | Yield |
|---|---|
| (i) PhNMe2, toluene, (ii) /BRN= 1736481/, Et3N, CHCl3; Multistep reaction; |

-
-
21139-44-6
2-(chloromethyl)-2-methyl-1,3-propanediol

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
50378-54-6
5-Chloromethyl-2-(2,4-dinitro-phenoxy)-5-methyl-[1,3,2]dioxaphosphinane 2-sulfide

| Conditions | Yield |
|---|---|
| (i) PSCl3, Py, Et2O, (ii) /BRN= 3782558/, MeCN; Multistep reaction; |
-
-
872-99-1
2-chloro-2-oxo-1,3,2-dioxaphosphorinane

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
4225-82-5
2-(2,4-Dinitrophenoxy)-<1.3.2>dioxaphosphorinan-2-oxid

| Conditions | Yield |
|---|---|
| In toluene |
-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
1003-18-5
1-chloro-2-phospholene 1-oxide

-
-
25133-50-0
1-(2,4-dinitro-phenoxy)-2,3-dihydro-1H-phosphole 1-oxide
-
-
75-77-4
chloro-trimethyl-silane

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
62947-23-3
2,4-dinitrophenoxy-trimethylsilane

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; Thermodynamic data; heat of reaction; |
-
-
13154-24-0
triisopropylsilyl chloride

-
-
1011-73-0
sodium 2,4-dinitrophenoxide

| Conditions | Yield |
|---|---|
| With 15-crown-5 In tetrahydrofuran at 25℃; Rate constant; Thermodynamic data; withot reagent; heat of reaction; |

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; Thermodynamic data; heat of reaction; |
-
-
1011-73-0
sodium 2,4-dinitrophenoxide

-
-
74410-72-3
2-chloro-2-oxo-5,6-tetramethylene-1,3,2-dioxaphosphorinane

-
-
74431-09-7
2-(2,4-dinitrophenoxy)-2-oxo-trans-5,6-tetramethylene-1,3,2-dioxaphosphorinane

-
-
74378-78-2, 74410-70-1, 74431-09-7
2-(2,4-dinitrophenoxy)-2-oxo-trans-5,6-tetramethylene-1,3,2-dioxaphosphorinane

| Conditions | Yield |
|---|---|
| In toluene at 90℃; for 0.0666667h; | |
| In toluene at 90℃; |

Sodium 2,4-dinitrophenate Standards and Recommendations
Content(%): ≥70
Purity(%): ≥98.0
Sodium 2,4-dinitrophenate Specification
The Sodium 2,4-dinitrophenate is an organic compound with the formula C6H3N2NaO5. The IUPAC name of this chemical is sodium 2,4-dinitrophenolate. With the CAS registry number 1011-73-0, it is also named as phenol, 2,4-dinitro-, sodium salt (1:1). The product's category is Intermediates of Dyes and Pigments.
Physical properties about Sodium 2,4-dinitrophenate are: (1)ACD/LogP: 1.74; (2)ACD/LogD (pH 5.5): 0.27; (3)ACD/LogD (pH 7.4): -1.21; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 7.19; (7)ACD/KOC (pH 7.4): 1; (8)#H bond acceptors: 7; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 3; (11)Polar Surface Area: 111.87 Å2; (12)Flash Point: 142.8 °C; (13)Enthalpy of Vaporization: 57.51 kJ/mol; (14)Boiling Point: 312.1 °C at 760 mmHg; (15)Vapour Pressure: 0.000294 mmHg at 25°C.
Preparation: this chemical can be prepared by 1-fluoro-2,4-dinitro-benzene. This reaction will need reagent NaOH and solvent H2O.
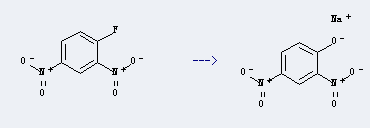
Uses of Sodium 2,4-dinitrophenate: it can be used to produce benzoic acid-(2,4-dinitro-phenyl ester) at temperature of 25 °C. It will need reagent tricaprylmethylammonium chloride and solvent benzene.

You can still convert the following datas into molecular structure:
(1)SMILES: [Na+].O=[N+]([O-])c1cc(ccc1[O-])[N+]([O-])=O
(2)InChI: InChI=1/C6H4N2O5.Na/c9-6-2-1-4(7(10)11)3-5(6)8(12)13;/h1-3,9H;/q;+1/p-1
(3)InChIKey: JMOHQJVXBQAVNW-REWHXWOFAB
(4)Std. InChI: InChI=1S/C6H4N2O5.Na/c9-6-2-1-4(7(10)11)3-5(6)8(12)13;/h1-3,9H;/q;+1/p-1
(5)Std. InChIKey: JMOHQJVXBQAVNW-UHFFFAOYSA-M
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| cat | LDLo | intramuscular | 25mg/kg (25mg/kg) | VASCULAR: BP ELEVATION NOT CHARACTERIZED IN AUTONOMIC SECTION | Journal of Pharmacology and Experimental Therapeutics. Vol. 48, Pg. 410, 1933. |
| cat | LDLo | subcutaneous | 25mg/kg (25mg/kg) | VASCULAR: BP ELEVATION NOT CHARACTERIZED IN AUTONOMIC SECTION | Journal of Pharmacology and Experimental Therapeutics. Vol. 48, Pg. 410, 1933. |
| dog | LDLo | intramuscular | 25mg/kg (25mg/kg) | LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION | Journal of Pharmacology and Experimental Therapeutics. Vol. 48, Pg. 410, 1933. |
| dog | LDLo | intravenous | 20mg/kg (20mg/kg) | LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 50, Pg. 20, 1935. |
| dog | LDLo | oral | 30mg/kg (30mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 49, Pg. 187, 1933. | |
| dog | LDLo | subcutaneous | 25mg/kg (25mg/kg) | VASCULAR: BP ELEVATION NOT CHARACTERIZED IN AUTONOMIC SECTION | Journal of Pharmacology and Experimental Therapeutics. Vol. 48, Pg. 410, 1933. |
| mouse | LD50 | subcutaneous | 50mg/kg (50mg/kg) | PERIPHERAL NERVE AND SENSATION: SPASTIC PARALYSIS WITH OR WITHOUT SENSORY CHANGE LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION SKIN AND APPENDAGES (SKIN): HAIR: OTHER | Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. Vol. 56, Pg. 23, 1960. |
| pigeon | LDLo | intramuscular | 15mg/kg (15mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 49, Pg. 187, 1933. | |
| pigeon | LDLo | intravenous | 15mg/kg (15mg/kg) | BEHAVIORAL: EXCITEMENT LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 50, Pg. 20, 1935. |
| rabbit | LDLo | intramuscular | 20mg/kg (20mg/kg) | LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION | Journal of Pharmacology and Experimental Therapeutics. Vol. 48, Pg. 410, 1933. |
| rabbit | LDLo | subcutaneous | 60mg/kg (60mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 49, Pg. 187, 1933. | |
| rat | LDLo | subcutaneous | 15mg/kg (15mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 48, Pg. 410, 1933. |
Related Products
- Sodium
- Sodium 2,4-dimethylbenzenesulfonate
- SODIUM γ-FLUORO-β-HYDROXYBUTYRATE
- Sodium ((3-methoxy-1-methyl-3-oxo-1-propenyl)amino)phenylacetate
- Sodium (+)-10-camphorsulfonate
- Sodium (2-carbamoylphenoxy)acetate
- Sodium (2-methyl-4-chlorophenoxy)acetate
- Sodium (6R,7R)-3-[(5-methyl-1,3,4-thiadiazol-2-yl)sulfanylmethyl]-8-oxo-7-[[2-(tetrazol-1-yl)acetyl]amino]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate pentahydrate
- Sodium (C10-16)alkylbenzenesulfonate
- Sodium (R,R)-5-(2-((2-(3-chlorophenyl)-2-hydroxyethyl)amino)propyl)-1,3-benzodioxole-2,2-
- 10117-38-1
- 101-17-7
- 1011-84-3
- 101184-73-0
- 101-18-8
- 10118-90-8
- 101191-83-7
- 101200-48-0
- 101200-51-5
- 1012-01-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View