Amadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
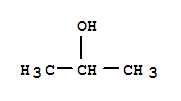
Cas:75-85-4
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryQINGDAO ON-BILLION INDUSTRAIL CO.,LTD
CASNO.75-85-4 EINECSNO.200-908-9 FORMULAC5H12O MOLWT.88.15 TOXICITYOralratLD50:1000mg/kg SYNONYMS t-AmylAlcohol;2-… Appearance:Colorless transparent liquid Storage:Flammables area Package:200L drum Application:synthetic material of perfume a
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen; copper-palladium; silica gel In ethanol at 25℃; under 760 Torr; Kinetics; | A n/a B 98% |
| With hydrogen; palladium dichloride In N,N-dimethyl-formamide under 18751.5 Torr; for 0.3h; Product distribution; Ambient temperature; various time; | A 2% B 91% |
| With hydrogen; nickel dihydroxide; Ni(C17H35COO)2 In toluene at 40℃; Product distribution; Kinetics; other catalyst. Object of study: selectivity; | A 41.6% B 85.5% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; hydrogen; nickel dichloride In isopropyl alcohol at 60℃; under 760.051 Torr; for 7h; | 90% |
| With hydrogen; palladium membrane at 29.9℃; Kinetics; electrochemical reduction; | |
| With alkali durch elektrolytische Reduktion an Platinkathoden; |

| Conditions | Yield |
|---|---|
| With nano magnetic sulfated zirconia (Fe3O4 at ZrO2/SO42-) In neat (no solvent) at 20℃; for 0.416667h; Green chemistry; | 80% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide In tetrahydrofuran; N,N-dimethyl-formamide for 7h; Ambient temperature; electrolysis with Pt/Cu electrodes; | 76% |
-
-
78-78-4
methylbutane

-
A
-
563-80-4
3-methyl-butan-2-one

-
B
-
75-85-4
tert-Amyl alcohol

-
C
-
96-17-3, 57456-98-1
2-Methylbutyraldehyde

-
D
-
598-75-4
3-methyl-2-butanol

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; Ba; trifluoroacetic acid In dichloromethane at 20℃; for 0.0333333h; | A 11% B 70% C 4.5% D n/a |
| With [2,2]bipyridinyl; Ba; trifluoroacetic acid In dichloromethane at 20℃; for 0.0333333h; Product distribution; | A 11% B 70% C 4.5% D n/a |

-
-
563-46-2
2-Methyl-1-butene

-
-
64-19-7
acetic acid

-
A
-
75-85-4
tert-Amyl alcohol

-
B
-
661-53-0
2-fluoro-2-methylbutane

-
C
-
625-16-1
tert-amyl acetate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water at 25℃; Kinetics; | A 70% B 25% C n/a |

-
-
54269-84-0
1,1-dimethyl-1-(2-propenyloxy)-propane

-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; titanium tetrachloride In tetrahydrofuran for 8h; Ambient temperature; | 49% |

| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; air; cobalt(II) acetate In benzonitrile at 100℃; under 7600 Torr; for 8h; Oxidation; | A 21% B 15% C 32% |


| Conditions | Yield |
|---|---|
| With acid-washed bentonite In acetone at 40 - 50℃; | 10% |

| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride |

| Conditions | Yield |
|---|---|
| at 100℃; |


| Conditions | Yield |
|---|---|
| With sulfuric acid at 50℃; | |
| With sulfuric acid at 120℃; under 7355.08 - 11032.6 Torr; | |
| With ammonium sulfate; sulfuric acid at 0℃; |
-
-
60-29-7
diethyl ether

-
-
28276-08-6
1,1-dimethylpropylmagnesium chloride

-
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| ausser Dimethyl-aethyl-essigsaeure; |


| Conditions | Yield |
|---|---|
| With glyceride alkali salt; water Hydrolysis; | |
| With sodium hydroxide Hydrolysis.in Gegenwart von fettsauren Alkalisalzen; |

| Conditions | Yield |
|---|---|
| With alkali durch elektrolytische Reduktion an Platinkathoden; | |
| With hydrogen; palladium at 20℃; under 760 Torr; Rate constant; effect of solvent (methanol, cyclohexane) on hydrogenation; | |
| With hydrogen; palladium at 20℃; under 735.5 Torr; Rate constant; effects of solvent on hydrogenation of title compound; |

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With hydrogen bromide at 165℃; | |
| With 3,3-dimethyldioxirane In acetone Rate constant; Ambient temperature; |
-
-
507-36-8
2-bromo-2-methylbutane

-
-
623-11-0
1-methyl-4-nitrosobenzene

-
A
-
75-85-4
tert-Amyl alcohol

-
B
-
513-35-9
2-methyl-but-2-ene

| Conditions | Yield |
|---|---|
| at 35℃; Hydrolysis; | |
| at 45℃; Hydrolysis; |


| Conditions | Yield |
|---|---|
| Hydrolysis; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
19781-24-9
3,3-dimethyl-2-pentanol

-
A
-
75-85-4
tert-Amyl alcohol

-
B
-
20669-04-9
3,3-dimethyl-2-pentanone

| Conditions | Yield |
|---|---|
| With chromium(VI) oxide; acetic acid |
-
-
66793-92-8
isopropyl-tert-pentyl-carbinol

-
A
-
75-85-4
tert-Amyl alcohol

-
B
-
34575-34-3
2,4,4-trimethyl-hexan-3-one

| Conditions | Yield |
|---|---|
| With chromium(VI) oxide; acetic acid |
-
-
4111-08-4
2-hydroxy-2-methyl-butyronitrile

-
A
-
75-85-4
tert-Amyl alcohol

-
B
-
109916-61-2, 133645-41-7, 560-24-7
3-hydroxy-3-methyl-pentan-2-one

-
C
-
78-93-3
butanone

-
-
24675-20-5
2-methyl-butan-2-ol; zirconium (IV)-compound

-
A
-
75-85-4
tert-Amyl alcohol

-
B
-
591-95-7
penta-1,2-diene

| Conditions | Yield |
|---|---|
| at 250 - 320℃; under 175 - 760 Torr; Pyrolysis; |
-
-
28276-08-6
1,1-dimethylpropylmagnesium chloride

-
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
-
75-85-4
tert-Amyl alcohol
-
-
28276-08-6
1,1-dimethylpropylmagnesium chloride

-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| With oxygen Zersetzung des Reaktionsproduktes mit Wasser; |

| Conditions | Yield |
|---|---|
| With bismuth(III) chloride In tetrachloromethane at 25℃; for 0.0833333h; | 100% |
| With hydrogenchloride In water at 20℃; for 0.05h; | 98% |
| With chloro-trimethyl-silane; dimethyl sulfoxide for 0.166667h; | 88% |
-
-
75-85-4
tert-Amyl alcohol

-
-
32115-55-2
N,N-Dimethylcarbamidsaeure-trimethylsilylester

-
-
6689-16-3
2-methyl-2-trimethylsiloxybutane

| Conditions | Yield |
|---|---|
| at 100℃; for 15h; | 100% |
| at 69.9℃; Mechanism; Rate constant; Kinetics; var. time and temp.; |

| Conditions | Yield |
|---|---|
| With pyridine In diethyl ether at 0℃; | 100% |
-
-
75-85-4
tert-Amyl alcohol

-
-
52177-63-6
4-methylthiophenyl chloroformate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane | 100% |

| Conditions | Yield |
|---|---|
| In tert-Amyl alcohol byproducts: isopropanol; anhydrous conditions; refluxing (48 h, distn. off of i-PrOH); solvent removal (vac.); elem. anal.; | 99.5% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride; iron(III) chloride at 0 - 5℃; for 6h; Large scale; | 99.4% |
| With AlCl2 | |
| With iron(III) chloride | |
| With aluminium trichloride |
-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| under N2; elem. anal.; | 99.3% |

| Conditions | Yield |
|---|---|
| With erbium(III) chloride at 50℃; for 3.5h; | 99% |
| cerium triflate In acetonitrile at 20℃; for 24h; | 98% |
| With Methylenediphosphonic acid at 20℃; for 3.5h; neat (no solvent); | 97% |
-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| With bis(tri-t-butylphosphine)palladium(0); N,N-diethyl-1,1,1-trimethylsilanamine at 80℃; for 24h; Inert atmosphere; Schlenk technique; | 99% |
-
-
75-85-4
tert-Amyl alcohol

-
-
94370-87-3
tert-amyl hypobromite

| Conditions | Yield |
|---|---|
| With bromine; mercury(II) oxide In pentane for 1h; Product distribution; Ambient temperature; other tertiary and secondary alcohols; | 98.5% |
-
-
43112-38-5
3-(trimethylsilyl)-2-oxazolidinone

-
-
75-85-4
tert-Amyl alcohol

-
-
6689-16-3
2-methyl-2-trimethylsiloxybutane

| Conditions | Yield |
|---|---|
| chloro-trimethyl-silane at 0℃; for 0.0333333h; | 98% |
-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| In benzene byproducts: isopropanol; moisture excluded; excess alcohol added to soln. of nickel tantalum isopropoxide; refluxed over a fractionating column; isopropanol liberated was continuously fractionated out with benzene between 72-80°C; excess of solvent removed under reduced pressure and compd. dried; elem. anal.; | 98% |
-
-
5765-44-6
5-methylisoxazole

-
-
75-85-4
tert-Amyl alcohol

-
-
4878-36-8
3,5-diamino-6-chloro-pyrazine-2-carboxylic acid

-
-
1431937-50-6
1-(2-methyl-2-butyl-carbamoyl)prop-1-en-2-yl 3,5-diamino-6-chloropyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: 5-methylisoxazole; tert-Amyl alcohol With trifluorormethanesulfonic acid In N,N-dimethyl-formamide for 1h; Cooling with ice; Stage #2: 3,5-diamino-6-chloro-pyrazine-2-carboxylic acid With triethylamine In N,N-dimethyl-formamide at 20℃; for 2h; Cooling with ice; | 98% |
| Stage #1: 5-methylisoxazole; tert-Amyl alcohol With trifluorormethanesulfonic acid Cooling with ice; Stage #2: 3,5-diamino-6-chloro-pyrazine-2-carboxylic acid With triethylamine In N,N-dimethyl-formamide at 25℃; for 2h; Cooling with ice; | 98% |

-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| In benzene byproducts: isopropanol; Excess of 2,2'-dimethylpentanol is added to a benzene soln. of the starting complex, the mixt. is refluxed for 5 h (N2).; The liberated isopropanol is removed azeotropically (72-80°C) till no isopropanol is detected in the destillate, the volatiles are stripped off in vacuo, the resulting complex is recrystallized from n-hexane (-10 to -15°C), elem. anal.; | 97.5% |

| Conditions | Yield |
|---|---|
| With oxygen In isopropyl alcohol at 20℃; under 51.7162 - 103.432 Torr; Schlenk technique; Cooling with ice; | 97.3% |
| Glovebox; Inert atmosphere; Sealed tube; | 97.3% |
| With 2,2',2''-triaminotriethylamine In pentane for 2h; Schlenk technique; Cooling with ice; Glovebox; | 435 g |
-
-
75-85-4
tert-Amyl alcohol

-
-
6343-16-4, 24425-58-9, 38447-89-1, 51252-31-4
bicyclo<2.2.1>hept-5,6-ene-2α,3β-dicarbonitrile

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid for 12h; Ritter reaction; | 97% |
-
-
75-85-4
tert-Amyl alcohol

-
-
463934-08-9
methanesulfonic acid 3-(naphthalen-2-oxy)-1-propyl ester

-
A
-
894085-83-7
2-[3-(2-methyl-2-butoxy)propoxy]naphthalene

-
B
-
398-53-8
2-(3-fluoro-n-propoxy)naphthalene

| Conditions | Yield |
|---|---|
| With rubidium fluoride; polystyrene-supported 1-n-hexyl-3-methylimidazolium BF4 at 90℃; for 1.5h; | A 2 % Spectr. B 97% |
| With cesium fluoride at 80℃; for 2.5 - 6h; Product distribution / selectivity; | A 4% B 93% |
| With cesium fluoride at 80℃; for 6h; | A 5 % Spectr. B 93% |


-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| With cesium fluoride; polystyrene-supported 1-n-hexyl-3-methylimidazolium BF4 at 80℃; for 1h; | A n/a B 97% |

-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| In benzene byproducts: isopropanol; moisture excluded; excess alcohol added to soln. of nickel niobium isopropoxide; refluxed over a fractionating column; isopropanol liberated was continuously fractionated out with benzene between 72-80°C; excess of solvent removed under reduced pressure and compd. dried; elem. anal.; | 97% |
-
-
75-85-4
tert-Amyl alcohol

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at 0 - 22℃; for 1h; Inert atmosphere; | 97% |
| With lithium In hexane Inert atmosphere; Schlenk technique; Reflux; | 77.8% |
-
-
75-85-4
tert-Amyl alcohol

-
-
88477-42-3
RbOC(Me)2Et

| Conditions | Yield |
|---|---|
| With rubidium In n-heptane Inert atmosphere; Schlenk technique; Reflux; | 96.7% |
| With rubidium In tetrahydrofuran for 48h; Inert atmosphere; Reflux; | 92.5% |

| Conditions | Yield |
|---|---|
| With hydrogen bromide at 50℃; for 0.25h; | 96.5% |
| With hydrogen bromide; lithium bromide at -10℃; for 2h; | 91% |
| With 1,2-dibromo-1,1,2,2-tetrachloroethane; triphenylphosphine In dichloromethane at 20℃; for 0.15h; Appel Halogenation; | 85% |
-
-
75-85-4
tert-Amyl alcohol

-
-
54237-96-6
2-Chloromethyl-1,3-dioxepane

-
-
1634-04-4
tert-butyl methyl ether

-
-
107-21-1
ethylene glycol

-
-
69814-56-8
2-methylene-1,3-dioxepane

| Conditions | Yield |
|---|---|
| With potassium hydroxide | 96.3% |


| Conditions | Yield |
|---|---|
| at 120℃; for 0.5h; Mechanism; | 96% |
| at 120℃; for 0.5h; | 96% |
| copper(II) 2-ethylhexanoate In benzene Heating; |
-
-
75-85-4
tert-Amyl alcohol

-
-
7677-24-9
trimethylsilyl cyanide

-
-
6689-16-3
2-methyl-2-trimethylsiloxybutane

| Conditions | Yield |
|---|---|
| at 25℃; for 0.0833333h; | 96% |
-
-
75-85-4
tert-Amyl alcohol

-
-
939-26-4
2-bromomethylnaphthyl bromide

-
B
-
55831-11-3
2-(fluoromethyl)naphthalene

| Conditions | Yield |
|---|---|
| With cesium fluoride; polystyrene-supported 1-n-hexyl-3-methylimidazolium BF4 at 80℃; for 0.5h; | A n/a B 96% |


| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: HN(CH3)2; under Ar, stirring at room temp. for 10 min;; layering with pentane, cooling to -25°C, standing overnight, elem. anal.;; | 96% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 24h; | 95% |
| With sodium carbonate; potassium carbonate; N-benzyl-N,N,N-triethylammonium chloride In dichloromethane for 3h; Heating; | 80% |
| With diethyl ether; magnesium |
-
-
75-85-4
tert-Amyl alcohol

-
-
999-97-3
1,1,1,3,3,3-hexamethyl-disilazane

-
-
6689-16-3
2-methyl-2-trimethylsiloxybutane

| Conditions | Yield |
|---|---|
| With aluminium(III) triflate at 20℃; for 1.11667h; | 95% |
| With copper(II) bis(trifluoromethanesulfonate) In acetonitrile at 20℃; for 2.5h; | 93% |
| With nano magnetic sulfated zirconia (Fe3O4 at ZrO2/SO42-) In neat (no solvent) at 20℃; for 0.5h; Green chemistry; | 82% |
Related products
Raw Materials
Downstream Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View















 F,
F, Xn
Xn