-
Name
THYMOQUINONE
- EINECS 207-721-1
- CAS No. 490-91-5
- Article Data96
- CAS DataBase
- Density 1.065 g/cm3
- Solubility
- Melting Point 45-47 ºC(lit.)
- Formula C10H12 O2
- Boiling Point 232 ºC at 760 mmHg
- Molecular Weight 164.204
- Flash Point 103.9 ºC
- Transport Information
- Appearance orange to brown crystals
- Safety Poison by intraperitoneal route. When heated to decomposition it emits acrid smoke and fumes.
- Risk Codes 22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms p-Mentha-3,6-diene-2,5-dione(6CI,7CI,8CI); 2-Isopropyl-5-methyl-1,4-benzoquinone; 2-Isopropyl-5-methyl-p-benzoquinone;2-Isopropyl-5-methylbenzoquinone; 2-Methyl-5-isopropyl-1,4-benzoquinone;2-Methyl-5-isopropyl-p-benzoquinone; 5-Isopropyl-2-methyl-1,4-benzoquinone;5-Isopropyl-2-methyl-p-benzoquinone; NSC 2228; Thymoquinon; Thymoquinone;p-Cymene-2,5-dione
- PSA 34.14000
- LogP 1.66690
Synthetic route
-
-
145674-57-3
3-isopropyl-4,4-dimethoxy-6-methyl-cyclohexa-2,5-dienone

-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride | 100% |

| Conditions | Yield |
|---|---|
| With [N,N'-bis(salicylidene)ethane-1,2-diaminato]cobalt(II); oxygen In N,N-dimethyl-formamide | 93% |
| With oxygen; salcomine In N,N-dimethyl-formamide for 9h; Ambient temperature; | 79% |
| With perchloric acid; lead dioxide In acetic acid at 25℃; | 60% |

| Conditions | Yield |
|---|---|
| With oxygen; salcomine In N,N-dimethyl-formamide Ambient temperature; | 90% |
| With Oxone In methanol; water at 20℃; for 0.5h; Catalytic behavior; Reagent/catalyst; | 32% |
| With sulfuric acid; water Oxydation mit Braunstein; |

| Conditions | Yield |
|---|---|
| With benzeneseleninic acid In dichloromethane for 2h; Ambient temperature; | A 82% B 7% C n/a |


| Conditions | Yield |
|---|---|
| With benzeneseleninic acid In dichloromethane for 2h; Ambient temperature; | A 75% B 19% C n/a |

-
-
89-83-8
thymol

-
A
-
490-91-5
thymoquinone

-
B
-
98268-32-7
2-(4-hydroxy-5-isopropyl-2-methylphenyl)-6-isopropyl-3-methyl-1,4-benzoquinone

-
C
-
98268-34-9
3-hydroxy-5,5'-diisopropyl-2,2'-dimethyl-4,4'-diphenoquinone

| Conditions | Yield |
|---|---|
| With oxygen; copper dichloride In ethanol at 60℃; for 48h; Product distribution; var. solvents; | A 25% B 65% C 10% |
| With oxygen; copper dichloride In ethanol at 60℃; for 48h; | A 25% B 65% C 10% |


| Conditions | Yield |
|---|---|
| With phosphomolybdic acid; dihydrogen peroxide In acetic acid at 30℃; for 5h; | A 64% B 13.5 % Chromat. |
| With phosphomolybdic acid; dihydrogen peroxide In acetic acid at 30℃; for 5h; Further byproducts given; | A 64 % Chromat. B 13.2% |
| With dihydrogen peroxide; methyltrioxorhenium(VII) In acetic acid at 40℃; for 2h; | A 4% B 8% |

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In chloroform for 3h; Ambient temperature; | A 50.2% B 25.5% |

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In chloroform for 3h; Ambient temperature; | A 47% B 30% |
| With 3-chloro-benzenecarboperoxoic acid In chloroform for 3h; Ambient temperature; | A 47% B 30% |
-
-
499-75-2
carvacrol

-
A
-
490-91-5
thymoquinone

-
-
95475-28-8, 106623-23-8, 113159-72-1
3,10-dihydroxy-6,12-di-iso-propyl-3,10-dimethyltricyclo[6.2.2.02,7]dodeca-5,11-diene-4,9-dione

| Conditions | Yield |
|---|---|
| With o-iodophenylacetic acid; 3-chloro-benzenecarboperoxoic acid In dichloromethane at 20℃; for 3h; Inert atmosphere; | A 37% B 10% C 32% |

-
-
89-83-8
thymol

-
A
-
490-91-5
thymoquinone

-
B
-
98268-34-9
3-hydroxy-5,5'-diisopropyl-2,2'-dimethyl-4,4'-diphenoquinone

| Conditions | Yield |
|---|---|
| With oxygen; copper dichloride In acetonitrile at 60℃; for 48h; | A 32% B 10% |
| With oxygen; copper dichloride In N,N-dimethyl-formamide at 60℃; for 48h; | A 25% B 30% |
-
-
89-83-8
thymol

-
A
-
490-91-5
thymoquinone

-
B
-
4586-58-7
3-hydroxy-5-isopropyl-2-methyl-[1,4]benzoquinone

-
C
-
138525-31-2
2-hydroxy-2-isopropyl-5-methylcyclohex-5-ene-1,4-dione

-
-
10476-80-9, 138525-30-1
trans-2-hydroxy-6-isopropyl-3-methylcyclohex-5-ene-1,4-dione

| Conditions | Yield |
|---|---|
| With 3,3-dimethyldioxirane; sodium hydrogencarbonate In acetone Further byproducts given; | A 23% B 9% C 18% D 12% |

-
-
89-83-8
thymol

-
A
-
490-91-5
thymoquinone

-
B
-
78617-27-3
3-methyl-6-(2-propyl)-1,2-benzoquinone

-
C
-
138525-31-2
2-hydroxy-2-isopropyl-5-methylcyclohex-5-ene-1,4-dione

-
-
10476-80-9, 138525-30-1
trans-2-hydroxy-6-isopropyl-3-methylcyclohex-5-ene-1,4-dione

| Conditions | Yield |
|---|---|
| With 3,3-dimethyldioxirane; sodium hydrogencarbonate In acetone Further byproducts given; | A 23% B 9% C 18% D 12% |


| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In chloroform for 3h; Ambient temperature; | 20.8% |
-
-
89-83-8
thymol

-
A
-
490-91-5
thymoquinone

-
B
-
490-06-2
3-isopropyl-6-methyl-1,2-benzenediol

-
C
-
138525-31-2
2-hydroxy-2-isopropyl-5-methylcyclohex-5-ene-1,4-dione

-
-
10476-80-9, 138525-30-1
trans-2-hydroxy-6-isopropyl-3-methylcyclohex-5-ene-1,4-dione

| Conditions | Yield |
|---|---|
| With 3,3-dimethyldioxirane In acetone | A 10% B 15 % Spectr. C 5% D 5% |


| Conditions | Yield |
|---|---|
| at 130 - 140℃; |

| Conditions | Yield |
|---|---|
| With nitric acid | |
| With iron(III) chloride | |
| With p-benzoquinone | |
| durch eine Oxydase, die in den Blaettern von Monarda fistulosa vorkommt; | |
| With 3,3-dimethyldioxirane; sodium hydrogencarbonate In acetone | 81 % Chromat. |
-
-
74317-48-9
2-isopropyl-5-methyl-p-benzoquinone dioxime

-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With copper(I) oxide; anhydrous methoxyethanol; acetone weiteres Reagens: wss. HCl; |
-
-
117886-49-4
4-aminocarvacrol

-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| bei der Oxydation; |

| Conditions | Yield |
|---|---|
| With potassium dichromate; sulfuric acid | |
| With Nitrite Diazotization; | |
| With hydrogenchloride; copper(II) sulfate | |
| With chromium(III) oxide; sulfuric acid |

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 20℃; |
-
-
120108-97-6
1,1-bis(5-isopropyl-4-hydroxy-2-methylphenyl)ethane

-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; sulfuric acid |

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With chromium(VI) oxide; sulfuric acid; water weiteres Reagens: Essigsaeure; |

| Conditions | Yield |
|---|---|
| at 130 - 140℃; |

-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate 1.) o-xylene, 100 deg C, 20 min, 2.) CH3CN, H2O, 0.5 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| In acetonitrile Rate constant; 0.1 M Bu4NPF6; |

-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide In benzene |
-
-
145674-58-4
2-isopropyl-4,4-dimethoxy-5-methyl-cyclohexa-2,5-dienone

-
A
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| In water at 22℃; Rate constant; |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In toluene at 25℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 3h; Diels-Alder Cycloaddition; Reflux; | 96% |
| With iron(III) chloride In neat (no solvent, solid phase) at 20℃; for 0.333333h; Diels-Alder Cycloaddition; | 93% |
| In dichloromethane at 20℃; for 120h; Time; Diels-Alder Cycloaddition; diastereoselective reaction; | 80% |
-
-
490-91-5
thymoquinone

-
-
17302-61-3
2-isopropyl-5-methyl-[1,4]benzoquinone-4-oxime

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride In ethanol for 24h; Heating; | 94% |
| With hydroxylamine hydrochloride; sodium acetate In ethanol for 1.5h; Reflux; | 81% |
| With hydrogenchloride; ethanol; hydroxylamine hydrochloride |

| Conditions | Yield |
|---|---|
| With triethylamine In isopropyl alcohol at 70℃; for 2h; Inert atmosphere; regioselective reaction; | 93% |
-
-
490-91-5
thymoquinone

-
-
85873-18-3
triphenylphosphonium trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 24h; Inert atmosphere; regioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| In ethanol for 24h; Heating; | 87% |

| Conditions | Yield |
|---|---|
| With acetic acid; zinc at 20℃; for 4h; Inert atmosphere; | 83% |
| With sodium dithionite In methanol; water at 25℃; for 2h; Inert atmosphere; | 49% |
| With hydrogenchloride; tin(ll) chloride |
-
-
490-91-5
thymoquinone

-
-
142572-85-8
triphenylphosphonium hydrotrifluoroacetate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 24h; Inert atmosphere; regioselective reaction; | 81% |
-
-
490-91-5
thymoquinone

-
-
147722-36-9
(3-Cyclopropylidenprop-1-enyl)-ethyl-ether

| Conditions | Yield |
|---|---|
| In 1,2-dimethoxyethane at 50℃; for 24h; | A 2.8% B 80.4% |

-
-
490-91-5
thymoquinone

-
-
914796-82-0
(4E)-2-isopropyl-5-methylbenzo-1,4-quinone 4-oxime

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium acetate In ethanol for 1.5h; Heating; | 80% |
-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol at 90℃; for 2h; | 78% |

| Conditions | Yield |
|---|---|
| In melt at 118℃; for 120h; | 76% |

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol at 70℃; for 12h; | 75% |
-
-
490-91-5
thymoquinone

-
-
1000-70-0
bis(trimethylsilyl)carbodi-imide

-
-
110375-15-0
(E,E)-N,N'-Dicyan-2-isopropyl-5-methyl-1,4-benzochinondiimin

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane for 3h; Ambient temperature; | 74% |

| Conditions | Yield |
|---|---|
| With (R)-2-(diphenyl(trimethylsilyloxy)methyl)pyrrolidine; o-fluoro-benzoic acid In chloroform-d1 at 40℃; for 24h; enantioselective reaction; | 74% |

| Conditions | Yield |
|---|---|
| With oxygen In methanol at 20℃; for 72h; | 73.6% |

-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With oxygen In methanol at 20℃; for 48h; | 68.3% |

| Conditions | Yield |
|---|---|
| With oxygen In methanol at 20℃; for 36h; | 66.7% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic anhydride In tetrahydrofuran at 100℃; Sealed tube; Inert atmosphere; | 66% |

| Conditions | Yield |
|---|---|
| With oxygen In methanol at 20℃; for 24h; | 61.1% |
-
-
16251-77-7
3-Phenylbutyraldehyde

-
-
490-91-5
thymoquinone

| Conditions | Yield |
|---|---|
| With α,α-diphenyl-2-pyrrolidinemethanol trimethylsilyl ether; oxygen; palladium diacetate; copper(II) bis(trifluoromethanesulfonate); o-fluoro-benzoic acid In dimethyl sulfoxide at 40℃; for 36h; Schlenk technique; Molecular sieve; enantioselective reaction; | 54% |
-
-
490-91-5
thymoquinone

-
B
-
2217-60-9
thymohydroquinone

-
C
-
17414-17-4
3-azido-2-methyl-5-isopropyl-1,4-benzohydroquinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium azide In methanol; water for 5h; Ambient temperature; | A 14% B 20% C 52% |

-
-
110-15-6
succinic acid

-
-
490-91-5
thymoquinone

-
-
1240724-54-2
3-(2-methyl-5-(1-methylethyl)-3,6-dioxocyclohexa-1,4-dienyl)propanoic acid

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate; silver nitrate In water; acetonitrile at 100℃; for 12h; | 52% |
| With ammonium peroxydisulfate; silver nitrate In water; acetonitrile | |
| With ammonium persulfate; silver nitrate In water; acetonitrile at 100℃; | |
| With ammonium peroxydisulfate; silver nitrate In water; acetonitrile at 100℃; for 13h; | 0.22 g |
Thymoquinone Chemical Properties
Product Name: Thymoquinone (CAS NO.490-91-5)
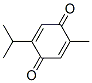
Molecular Formula: C10H12O2
Molecular Weight: 164.2g/mol
Mol File: 490-91-5.mol
Einecs: 207-721-1
Appearance: Orange to brown crystals
Melting Point: 45-47 °C(lit.)
Boiling point: 232 °C at 760 mmHg
Flash Point: 103.9 °C
Density: 1.065 g/cm3
Surface Tension: 35.8 dyne/cm
Enthalpy of Vaporization: 46.87 kJ/mol
Vapour Pressure: 0.0603 mmHg at 25°C
XLogP3-AA: 2
H-Bond Donor: 0
H-Bond Acceptor: 2
Product Categories: Anthraquinones, Hydroquinones and Quinones; Benzoquinones
Thymoquinone Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | intraperitoneal | 10mg/kg (10mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 15, Pg. 1227, 1965. |
Thymoquinone Consensus Reports
Reported in EPA TSCA Inventory.
Thymoquinone Safety Profile
Poison by intraperitoneal route. When heated to decomposition it emits acrid smoke and fumes.
Safety Information of Thymoquinone (CAS NO.490-91-5):
Hazard Codes: Xn
Risk Statements: 22-36/37/38
22: Harmful if swallowed
36: Irritating to the eyes
37: Irritating to the respiratory system
38: Irritating to the skin
Safety Statements: 26-36
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
Thymoquinone Specification
Thymoquinone ,its CAS NO. is 490-91-5,the synonyms is 2,5-Cyclohexadiene-1,4-dione, 5-isopropyl-2-methyl- ; 2-Isopropyl-5-methylbenzo-1,4-quinone ; 2-Isopropyl-5-methylbenzoquinone ; 2-Methyl-5-(1-methylethyl)-2,5-cyclohexadiene-1,4-dione ; 2-Methyl-5-isopropyl-1,4-benzoquinone ; 2-Methyl-5-isopropyl-p-benzoquinone ; 4-dione,2-methyl-5-(1-methylethyl)-5-cyclohexadiene-1 ; 4-dione,5-isopropyl-2-methyl-5-cyclohexadiene-1 .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View