-
Name
Troglitazone
- EINECS
- CAS No. 97322-87-7
- Article Data6
- CAS DataBase
- Density 1.266 g/cm3
- Solubility
- Melting Point 184-186 °C
- Formula C24H27NO5S
- Boiling Point 657 °C at 760 mmHg
- Molecular Weight 441.548
- Flash Point 351.2 °C
- Transport Information
- Appearance crystalline solid
- Safety 22-24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms CI 991;CS045;Depotox;GR 92132X;Noscal;Rezulin;Romglizone;
- PSA 110.16000
- LogP 4.70300
Synthetic route

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; dimethylaminoacetic acid; sodium hydroxide In tetrahydrofuran; water at 20℃; | 84% |
| With hydrogen; acetic acid In water; palladium |
-
-
199441-78-6
5-[4-(6-benzyloxy-2,5,7,8-tetramethyl-chroman-2-ylmethoxy)-benzyl]-thiazolidine-2,4-dione

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid at 70 - 80℃; debenzylation; | 70% |
-
-
17356-08-0
thiourea

-
-
97322-68-4
Ethyl 3-<4-<(6-acetoxy-2,5,7,8-tetramethylchroman-2-yl)methoxy>phenyl>-2-chloropropionate

-
A
-
97322-87-7
Troglitazone

-
B
-
97323-08-5
5-[4-(6-hydroxy-2,5,7,8-tetramethylchroman-2-ylmethoxy)benzyl]-2-iminothiazolidin-4-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid 1.) sulfolane, 115 to 120 deg C, 80 min, 2.) H2O, 85 to 90 deg c, 12 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
127040-01-1
Troglitazone glucuronide

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| enzymatic treatment using glusulase; |
-
-
459-57-4
4-fluorobenzaldehyde

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium tert-butoxide / dimethylformamide / 8 h / 80 °C 2: AcOH; piperidine / toluene / 4 h / Heating 3: HCl / methanol / 0.25 h 4: 84 percent / CoCl2; DMG; NaBH4 / NaOH / tetrahydrofuran; H2O / 20 °C View Scheme |
-
-
56305-04-5, 53188-07-1
trolox

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: imidazole / dimethylformamide 2: LAH / 3 h / 20 °C 3: potassium tert-butoxide / dimethylformamide / 8 h / 80 °C 4: AcOH; piperidine / toluene / 4 h / Heating 5: HCl / methanol / 0.25 h 6: 84 percent / CoCl2; DMG; NaBH4 / NaOH / tetrahydrofuran; H2O / 20 °C View Scheme | |
| Multi-step reaction with 8 steps 1: toluene-4-sulfonic acid / 12 h / Reflux 2: dmap / dichloromethane / 2 h / 20 °C / Inert atmosphere 3: lithium aluminium tetrahydride / tetrahydrofuran / 0 - 20 °C / Inert atmosphere 4: pyridine / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 5: potassium carbonate / N,N-dimethyl-formamide / 48 h / 2 °C / Inert atmosphere 6: piperidine / ethanol / 12 h / Inert atmosphere; Reflux 7: palladium 10% on activated carbon; hydrogen / 1,4-dioxane / 20 h / 20 °C / 3620.13 Torr 8: hydrogenchloride; water / acetonitrile / 0.75 h / Reflux View Scheme |
-
-
228114-44-1
(6-t-butyldimethylsilyloxy-2,5,7,8-tetramethyl-3,4-dihydro-2H-1-benzo[1,2-b]pyran-2yl)methanol

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium tert-butoxide / dimethylformamide / 8 h / 80 °C 2: AcOH; piperidine / toluene / 4 h / Heating 3: HCl / methanol / 0.25 h 4: 84 percent / CoCl2; DMG; NaBH4 / NaOH / tetrahydrofuran; H2O / 20 °C View Scheme |
-
-
716320-66-0
6-tert-butyl(dimethyl)silyloxy-2,5,7,8-tetramethyl-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: LAH / 3 h / 20 °C 2: potassium tert-butoxide / dimethylformamide / 8 h / 80 °C 3: AcOH; piperidine / toluene / 4 h / Heating 4: HCl / methanol / 0.25 h 5: 84 percent / CoCl2; DMG; NaBH4 / NaOH / tetrahydrofuran; H2O / 20 °C View Scheme |
-
-
716320-67-1
4-[6-(tert-butyl-dimethyl-silanyloxy)-2,5,7,8-tetramethyl-chroman-2-ylmethoxy]-benzaldehyde

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: AcOH; piperidine / toluene / 4 h / Heating 2: HCl / methanol / 0.25 h 3: 84 percent / CoCl2; DMG; NaBH4 / NaOH / tetrahydrofuran; H2O / 20 °C View Scheme |

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: HCl / methanol / 0.25 h 2: 84 percent / CoCl2; DMG; NaBH4 / NaOH / tetrahydrofuran; H2O / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: t-BuOK / dimethylformamide / 1 h / 25 °C 1.2: 60 percent / dimethylformamide / 15 h / 25 °C 2.1: benzoic acid; piperidine / toluene / 4 h / 120 °C 3.1: 54 percent / Mg; MeOH / 8 h / 45 °C 4.1: 70 percent / HCl; AcOH / 70 - 80 °C View Scheme |
-
-
171270-07-8
(6-Benzyloxy-3,4-dihydro-2,5,7,8-tetramethyl-2H-1-benzopyran-2-yl)methanol

-
A
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: t-BuOK / dimethylformamide / 1 h / 25 °C 1.2: 60 percent / dimethylformamide / 15 h / 25 °C 2.1: benzoic acid; piperidine / toluene / 4 h / 120 °C 3.1: 54 percent / Mg; MeOH / 8 h / 45 °C 4.1: 70 percent / HCl; AcOH / 70 - 80 °C View Scheme |
-
-
138564-69-9
4-[6-benzyloxy-2,5,7,8-tetramethylchroman-2-ylmethoxy]benzaldehyde

-
A
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: benzoic acid; piperidine / toluene / 4 h / 120 °C 2: 54 percent / Mg; MeOH / 8 h / 45 °C 3: 70 percent / HCl; AcOH / 70 - 80 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 54 percent / Mg; MeOH / 8 h / 45 °C 2: 70 percent / HCl; AcOH / 70 - 80 °C View Scheme |
-
-
79907-49-6
2-hydroxymethyl-2,5,7,8-tetramethyl-chroman-6-ol

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 1.) NaH / 1.) DMF, rt, 1 h, 2.) benzene, rt, 1 h 2: 1.) NaH / 1.) DMSO, benzene, 60 deg C, 20 min, 2.) 60 deg C, 1 h 3: 94 percent / AcOH, 10percent H2SO4 / 0.17 h / 60 °C 4: 94 percent / pyridine / 1.) 10 deg C to rt, 2.) 30 deg C, 1 h 5: 98 percent / H2 / 10percent Pd-C / methanol; benzene / 3 h / 2325.2 - 2850.2 Torr 6: 1.) aq. NaNO2, conc. HCl, 2.) Cu2O / 1.) acetone, 0 deg C, 2.) 40 to 43 deg C, 30 min 7: 2.) AcOH, conc. HCl / 1.) sulfolane, 115 to 120 deg C, 80 min, 2.) H2O, 85 to 90 deg c, 12 h View Scheme |
-
-
107188-55-6
2-hydroxymethyl-6-methoxymethoxy-2,5,7,8-tetramethylchroman

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 1.) NaH / 1.) DMSO, benzene, 60 deg C, 20 min, 2.) 60 deg C, 1 h 2: 94 percent / AcOH, 10percent H2SO4 / 0.17 h / 60 °C 3: 94 percent / pyridine / 1.) 10 deg C to rt, 2.) 30 deg C, 1 h 4: 98 percent / H2 / 10percent Pd-C / methanol; benzene / 3 h / 2325.2 - 2850.2 Torr 5: 1.) aq. NaNO2, conc. HCl, 2.) Cu2O / 1.) acetone, 0 deg C, 2.) 40 to 43 deg C, 30 min 6: 2.) AcOH, conc. HCl / 1.) sulfolane, 115 to 120 deg C, 80 min, 2.) H2O, 85 to 90 deg c, 12 h View Scheme |
-
-
107188-58-9
6-Hydroxy-2,5,7,8-tetramethyl-2-<(4-nitrophenoxy)methyl>chroman

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 94 percent / pyridine / 1.) 10 deg C to rt, 2.) 30 deg C, 1 h 2: 98 percent / H2 / 10percent Pd-C / methanol; benzene / 3 h / 2325.2 - 2850.2 Torr 3: 1.) aq. NaNO2, conc. HCl, 2.) Cu2O / 1.) acetone, 0 deg C, 2.) 40 to 43 deg C, 30 min 4: 2.) AcOH, conc. HCl / 1.) sulfolane, 115 to 120 deg C, 80 min, 2.) H2O, 85 to 90 deg c, 12 h View Scheme |
-
-
107188-37-4
6-acetoxy-2-[(4-aminophenoxy)methyl]-2,5,7,8-tetramethylchromane

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) aq. NaNO2, conc. HCl, 2.) Cu2O / 1.) acetone, 0 deg C, 2.) 40 to 43 deg C, 30 min 2: 2.) AcOH, conc. HCl / 1.) sulfolane, 115 to 120 deg C, 80 min, 2.) H2O, 85 to 90 deg c, 12 h View Scheme |
-
-
118070-83-0
2,5,7,8-tetramethyl-6-acetoxy-2-(4-nitrophenyloxy)methylchroman

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 98 percent / H2 / 10percent Pd-C / methanol; benzene / 3 h / 2325.2 - 2850.2 Torr 2: 1.) aq. NaNO2, conc. HCl, 2.) Cu2O / 1.) acetone, 0 deg C, 2.) 40 to 43 deg C, 30 min 3: 2.) AcOH, conc. HCl / 1.) sulfolane, 115 to 120 deg C, 80 min, 2.) H2O, 85 to 90 deg c, 12 h View Scheme |
-
-
118070-84-1
6-(Methoxymethoxy)-2,5,7,8-tetramethyl-2-<(4-nitrophenoxy)methyl>chroman

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 94 percent / AcOH, 10percent H2SO4 / 0.17 h / 60 °C 2: 94 percent / pyridine / 1.) 10 deg C to rt, 2.) 30 deg C, 1 h 3: 98 percent / H2 / 10percent Pd-C / methanol; benzene / 3 h / 2325.2 - 2850.2 Torr 4: 1.) aq. NaNO2, conc. HCl, 2.) Cu2O / 1.) acetone, 0 deg C, 2.) 40 to 43 deg C, 30 min 5: 2.) AcOH, conc. HCl / 1.) sulfolane, 115 to 120 deg C, 80 min, 2.) H2O, 85 to 90 deg c, 12 h View Scheme |
-
-
107187-56-4
(+/-)-5-<4-(6-hydroxy-2,5,7,8-tetramethyl-2H-chromen-2-ylmethoxy)benzyl>thiazolidine-2,4-dione

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| In methanol; palladium |
-
-
109-86-4
2-methoxy-ethanol

-
-
97322-68-4
Ethyl 3-<4-<(6-acetoxy-2,5,7,8-tetramethylchroman-2-yl)methoxy>phenyl>-2-chloropropionate

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; thiourea In sulfolane |
-
-
1375483-15-0
carbonic acid tert-butyl ester 2-{4-[2,4-dioxo-thiazolidin-(5Z)-ylidenemethyl]phenoxymethyl}-2,5,7,8-tetramethyl-chroman-6-yl ester

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: palladium 10% on activated carbon; hydrogen / 1,4-dioxane / 20 h / 20 °C / 3620.13 Torr 2: hydrogenchloride; water / acetonitrile / 0.75 h / Reflux View Scheme |
-
-
1375483-24-1
carbonic acid tert-butyl ester 2-{4-[2,4-dioxothiazolidin-5-ylmethyl]phenoxymethyl}-2,5,7,8-tetramethylchroman-6-yl ester

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water In acetonitrile for 0.75h; Reflux; |

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium carbonate / N,N-dimethyl-formamide / 48 h / 2 °C / Inert atmosphere 2: piperidine / ethanol / 12 h / Inert atmosphere; Reflux 3: palladium 10% on activated carbon; hydrogen / 1,4-dioxane / 20 h / 20 °C / 3620.13 Torr 4: hydrogenchloride; water / acetonitrile / 0.75 h / Reflux View Scheme |
-
-
1375483-09-2
6-tert-butoxycarbonyloxy-2,5,7,8-tetramethyl-chroman-2-carboxylic acid ethyl ester

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: lithium aluminium tetrahydride / tetrahydrofuran / 0 - 20 °C / Inert atmosphere 2: pyridine / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 3: potassium carbonate / N,N-dimethyl-formamide / 48 h / 2 °C / Inert atmosphere 4: piperidine / ethanol / 12 h / Inert atmosphere; Reflux 5: palladium 10% on activated carbon; hydrogen / 1,4-dioxane / 20 h / 20 °C / 3620.13 Torr 6: hydrogenchloride; water / acetonitrile / 0.75 h / Reflux View Scheme |
-
-
1375483-11-6
carbonic acid tert-butyl ester 2-hydroxymethyl-2,5,7,8-tetramethyl-chroman-6-yl ester

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: pyridine / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 2: potassium carbonate / N,N-dimethyl-formamide / 48 h / 2 °C / Inert atmosphere 3: piperidine / ethanol / 12 h / Inert atmosphere; Reflux 4: palladium 10% on activated carbon; hydrogen / 1,4-dioxane / 20 h / 20 °C / 3620.13 Torr 5: hydrogenchloride; water / acetonitrile / 0.75 h / Reflux View Scheme |
-
-
1375483-13-8
carbonic acid tert-butyl ester 2-(4-formylphenoxymethyl)-2,5,7,8-tetramethyl-chroman-6-yl ester

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: piperidine / ethanol / 12 h / Inert atmosphere; Reflux 2: palladium 10% on activated carbon; hydrogen / 1,4-dioxane / 20 h / 20 °C / 3620.13 Torr 3: hydrogenchloride; water / acetonitrile / 0.75 h / Reflux View Scheme |
-
-
53174-07-5
ethyl 6-hydroxy-2,5,7,8-tetramethyl-3,4-dihydro-2H-chromene-2-carboxylate

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: dmap / dichloromethane / 2 h / 20 °C / Inert atmosphere 2: lithium aluminium tetrahydride / tetrahydrofuran / 0 - 20 °C / Inert atmosphere 3: pyridine / dichloromethane / 0.5 h / 0 °C / Inert atmosphere 4: potassium carbonate / N,N-dimethyl-formamide / 48 h / 2 °C / Inert atmosphere 5: piperidine / ethanol / 12 h / Inert atmosphere; Reflux 6: palladium 10% on activated carbon; hydrogen / 1,4-dioxane / 20 h / 20 °C / 3620.13 Torr 7: hydrogenchloride; water / acetonitrile / 0.75 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| With pyridine for 48h; Ambient temperature; | 80% |

-
-
97322-87-7
Troglitazone

-
-
97323-02-9
5-[4-(2,5,7,8-tetramethyl-6-nicotinoyloxychroman-2-ylmethoxy)benzyl]thiazolidine-2,4-dione

| Conditions | Yield |
|---|---|
| 75% |
-
-
108-30-5
succinic acid anhydride

-
-
97322-87-7
Troglitazone

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane for 48h; Reflux; | 63% |
| With pyridine at 85℃; for 6h; | |
| With pyridine In cyclohexane |
-
-
97322-87-7
Troglitazone

-
-
97323-01-8
5-[4-(6-benzoyloxy-2,5,7,8-tetramethylchroman-2-ylmethoxy)benzyl]thiazolidine-2,4-dione

| Conditions | Yield |
|---|---|
| 62% |
-
-
97322-87-7
Troglitazone

-
-
97323-00-7
5-[4-(6-butyryloxy-2,5,7,8-tetramethylchroman-2-ylmethoxy)benzyl]thiazolidine-2,4-dione

| Conditions | Yield |
|---|---|
| 56% |
-
-
97322-87-7
Troglitazone

-
-
7355-18-2
(2S,3S,4S,5R,6S)-3,4,5,6-Tetraacetoxy-tetrahydro-pyran-2-carboxylic acid methyl ester

-
-
127040-00-0
methyl 2,3,4-tri-O-acetyl-1-O-<2-<4-<2,4-dioxothiazolidin-5-ylmethyl>phenoxymethyl>-2,5,7,8-tetramethylchroman-6-yl>-β-D-glucopyranuronate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In nitrobenzene at 85℃; under 20 Torr; for 4h; | 23% |
-
-
97322-87-7
Troglitazone

-
-
127040-01-1
Troglitazone glucuronide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 23 percent / p-toluenesulfonic acid monohydrate / nitrobenzene / 4 h / 85 °C / 20 Torr 2: 100 percent / 2.4percent aq.NaOH / methanol / 1 h / Ambient temperature View Scheme |
-
-
5292-43-3
bromoacetic acid tert-butyl ester

-
-
97322-87-7
Troglitazone

-
-
107187-69-9
Di-t-butyl α,α'-{5-[4-(6-t-butoxycarbonylmethoxy-2,5,7,8-tetramethylchroman-2-ylmethoxy)benzyl ]-2,4-dioxothiazolidine-3,5-diyl}diacetate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone |
Troglitazone Chemical Properties
Product Name: Troglitazone (CAS NO.97322-87-7)
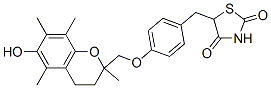
Molecular Formula: C24H27NO5S
Molecular Weight: 441.54g/mol
Mol File: 97322-87-7.mol
Appearance: Crystalline Solid
Melting Point: 184-186°C
Boiling point: 657 °C at 760 mmHg
Flash Point: 351.2 °C
Density: 1.266 g/cm3
Surface Tension: 51 dyne/cm
Enthalpy of Vaporization: 100.24 kJ/mol
Vapour Pressure: 7.24E-18 mmHg at 25°C
XLogP3-AA: 5.1
H-Bond Donor: 2
H-Bond Acceptor: 5
Structure Descriptors of Troglitazone (CAS NO.97322-87-7):
IUPAC Name: 5-[[4-[(6-hydroxy-2,5,7,8-tetramethyl-3,
4-dihydrochromen-2-yl)methoxy]phenyl]methyl]-1,3-thiazolidine-2,4-dione
Canonical SMILES: CC1=C(C(=C2CCC(OC2=C1C)(C)COC3=CC=C(C=C3)CC4C(=O)NC(=O)S4)C)O
InChI: InChI=1S/C24H27NO5S/c1-13-14(2)21-18(15(3)20(13)26)9-10-24(4,
30-21)12-29-17-7-5-16(6-8-17)11-19-22(27)25-23(28)31-19/h5-8,19,26H,
9-12H2,1-4H3,(H,25,27,28)
InChIKey: GXPHKUHSUJUWKP-UHFFFAOYSA-N
Product Categories: Active Pharmaceutical Ingredients; Intermediates & Fine Chemicals; Pharmaceuticals; Intracellular receptor
Troglitazone Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| monkey | LD50 | oral | > 2gm/kg (2000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 38, Pg. 1121, 1996. | |
| mouse | LD | oral | > 300mg/kg (300mg/kg) | European Patent Application. Vol. #439321, | |
| mouse | LD50 | intraperitoneal | > 5gm/kg (5000mg/kg) | Iyakuhin Kenkyu. Study of Medical Supplies. Vol. 26, Pg. 953, 1995. | |
| mouse | LD50 | intravenous | 42mg/kg (42mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 9, Pg. 759, 1967. | |
| rat | LD50 | intraperitoneal | > 4gm/kg (4000mg/kg) | Iyakuhin Kenkyu. Study of Medical Supplies. Vol. 26, Pg. 953, 1995. | |
| rat | LD50 | oral | > 5gm/kg (5000mg/kg) | Iyakuhin Kenkyu. Study of Medical Supplies. Vol. 26, Pg. 953, 1995. | |
| women | TDLo | oral | 840mg/kg/15W- (840mg/kg) | BRAIN AND COVERINGS: ENCEPHALITIS LIVER: "HEPATITIS (HEPATOCELLULAR NECROSIS), DIFFUSE" LIVER: "JAUNDICE, OTHER OR UNCLASSIFIED" | Annals of Internal Medicine. Vol. 129, Pg. 98, 1998. |
Troglitazone Safety Profile
Safety Information of Troglitazone (CAS NO.97322-87-7):
Safety Statements: 22-24/25
22: Do not breathe dust
24: Avoid contact with skin
25: Avoid contact with eyes
Troglitazone Specification
Troglitazone ,its CAS NO. is 97322-87-7,the synonyms is (+-)-all-rac-5-(p-((6-Hydroxy-2,5,7,8-tetramethyl-2-chromanyl)methoxy)benzyl)-2,4-thiazolidinedione ; 2,4-Thiazolidinedione, 5-((4-((3,4-dihydro-6-hydroxy-2,5,7,8-tetramethyl-2H-1-benzopyran-2-yl)methoxy)phenyl)methyl)- ; 5-(4-(6-Hydroxy-2,5,7,8-tetramethylchroman-2-ylmethoxy)benzyl)thiazolidine-2,4-dione ; BRN 4338399 ; CCRIS 8969 ; CI 991 ; CS 045 ; GR 92132X ; Prelay ; Rezulin ; Romglizone ; UNII-I66ZZ0ZN0E .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View