-
Name
alpha,alpha-Diphenyl-4-piperidinomethanol
- EINECS 204-092-5
- CAS No. 115-46-8
- Article Data15
- CAS DataBase
- Density 1.103 g/cm3
- Solubility
- Melting Point 160-163 °C
- Formula C18H21NO
- Boiling Point 445.5 °C at 760 mmHg
- Molecular Weight 267.371
- Flash Point 142 °C
- Transport Information
- Appearance white to light beige crystalline powder
- Safety 24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 4-(Diphenylhydroxymethyl)piperidine;4-(Hydroxydiphenylmethyl)piperidine;Ataractan;Azacyclonol;Calmeran;Diphenyl-4-piperidylmethanol;Frenoton;MDL 4829;MER 17;Psychosan;a-(4-Piperidyl)benzhydrol;g-Pipradol;
- PSA 32.26000
- LogP 3.25090
Synthetic route
-
-
114399-88-1
α,α-diphenyl-1-(phenylmethyl)-4-piperidinemethanol

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol at 70℃; under 2585.7 Torr; | 99% |
| palladium In ethanol |
-
-
58113-28-3
N-acetyl-α,α-diphenyl-4-piperidinemethanol

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water at 65 - 97℃; for 12h; | 95% |

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With hydrogen; palladium(II) hydroxide In methanol at 10 - 20℃; under 258.581 Torr; for 5h; Reagent/catalyst; Autoclave; | 95% |
-
-
96067-93-5
benzyl 4-(hydroxydiphenylmethyl)-piperidine-1-carboxylate

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With hydrogen; acetic acid; palladium on activated charcoal In ethanol at 25℃; under 3102.89 Torr; for 2.5h; Hydrogenolysis; | 88% |
-
-
112818-77-6
ethyl 4-(hydroxydiphenylmethyl)piperidine-1-carboxylate

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol for 24h; Heating; | 75% |
| In ethanol for 24h; Reflux; Inert atmosphere; Alkaline conditions; | 75% |

| Conditions | Yield |
|---|---|
| With acetic acid; platinum Hydrogenation; | |
| Multi-step reaction with 5 steps 1: acetone / 20 °C 2: sodium tetrahydroborate / methanol / 4 h / 20 °C 3: cyclohexanol / 2 h / Reflux 4: potassium carbonate / chloroform / 20 °C 5: ethanol / 24 h / Reflux; Inert atmosphere; Alkaline conditions View Scheme | |
| Multi-step reaction with 3 steps 1: acetonitrile / Reflux 2: potassium borohydride / methanol / 3 h / 10 - 20 °C 3: hydrogen; palladium(II) hydroxide / methanol / 5 h / 10 - 20 °C / 258.58 Torr / Autoclave View Scheme | |
| Multi-step reaction with 3 steps 1: acetonitrile / Reflux 2: lithium borohydride / methanol / 3 h / 10 - 20 °C 3: hydrogen; palladium(II) hydroxide / methanol / 5 h / 10 - 20 °C / 258.58 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 99 percent / triethylamine / tetrahydrofuran; H2O / 2 h / 25 °C 2: 73 percent / tetrahydrofuran / 0.83 h / -20 - 20 °C 3: 88 percent / acetic acid; hydrogen / Pd/C / ethanol / 2.5 h / 25 °C / 3102.89 Torr View Scheme |
-
-
160809-38-1
1-benzyl 4-ethyl piperidine-1,4-dicarboxylate

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 73 percent / tetrahydrofuran / 0.83 h / -20 - 20 °C 2: 88 percent / acetic acid; hydrogen / Pd/C / ethanol / 2.5 h / 25 °C / 3102.89 Torr View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 70 percent / K2CO3 / CH2Cl2 / Ambient temperature 2: 75 percent / 50percent KOH / ethanol / 24 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / chloroform / 20 °C 2: ethanol / 24 h / Reflux; Inert atmosphere; Alkaline conditions View Scheme |
-
-
20735-04-0
(1,2,3,6-tetrahydro-1-methylpyridin-4-yl)diphenylmethanol

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 85 percent / Ra-Ni / various solvent(s) / 2 h / Heating 2: 70 percent / K2CO3 / CH2Cl2 / Ambient temperature 3: 75 percent / 50percent KOH / ethanol / 24 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: cyclohexanol / 2 h / Reflux 2: potassium carbonate / chloroform / 20 °C 3: ethanol / 24 h / Reflux; Inert atmosphere; Alkaline conditions View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1) Mg / 1) THF, reflux; 2) THF, room temperature, overnight 2: 99 percent / H2 / 5percent Pd/C / ethanol / 70 °C / 2585.7 Torr View Scheme |
-
-
24228-40-8
N-benzyl isonipecotic acid ethyl ester

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1) Mg / 1) THF, reflux; 2) THF, room temperature, overnight 2: 99 percent / H2 / 5percent Pd/C / ethanol / 70 °C / 2585.7 Torr View Scheme |
-
-
114399-88-1
α,α-diphenyl-1-(phenylmethyl)-4-piperidinemethanol

-
-
108-20-3
di-isopropyl ether

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| palladium-carbon In ethanol | |
| palladium In ethanol |
-
-
113012-26-3
4-(hydroxy-diphenyl-methyl)-1-methyl-pyridinium iodide

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: sodium tetrahydroborate / methanol / 4 h / 20 °C 2: cyclohexanol / 2 h / Reflux 3: potassium carbonate / chloroform / 20 °C 4: ethanol / 24 h / Reflux; Inert atmosphere; Alkaline conditions View Scheme |
-
-
14548-46-0
4-Benzoylpyridine

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: tetrahydrofuran / 20 °C / Inert atmosphere 2: acetone / 20 °C 3: sodium tetrahydroborate / methanol / 4 h / 20 °C 4: cyclohexanol / 2 h / Reflux 5: potassium carbonate / chloroform / 20 °C 6: ethanol / 24 h / Reflux; Inert atmosphere; Alkaline conditions View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: tetrahydrofuran / 20 °C / Inert atmosphere 2: acetone / 20 °C 3: sodium tetrahydroborate / methanol / 4 h / 20 °C 4: cyclohexanol / 2 h / Reflux 5: potassium carbonate / chloroform / 20 °C 6: ethanol / 24 h / Reflux; Inert atmosphere; Alkaline conditions View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: 4 h / Reflux 2.1: thionyl chloride / toluene / 6 h / 40 °C 3.1: aluminum (III) chloride / 10 h / 70 °C 4.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 4.2: 10 - 30 °C / Inert atmosphere 5.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 4 h / Reflux 2.1: thionyl chloride / toluene / 6 h / 40 °C 3.1: aluminum (III) chloride / 10 h / 70 °C 4.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 4.2: 10 - 30 °C / Inert atmosphere 5.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 6 h / Reflux 2.1: thionyl chloride / toluene / 6 h / 40 °C 3.1: aluminum (III) chloride / 10 h / 70 °C 4.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 4.2: 10 - 30 °C / Inert atmosphere 5.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 6 h / Reflux 2.1: thionyl chloride / toluene / 6 h / 40 °C 3.1: aluminum (III) chloride / 10 h / 70 °C 4.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 4.2: 10 - 30 °C / Inert atmosphere 5.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme |
-
-
25503-90-6
1-acetyl-piperidine-4-carboxylic acid

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: thionyl chloride / toluene / 6 h / 40 °C 2.1: aluminum (III) chloride / 10 h / 70 °C 3.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 3.2: 10 - 30 °C / Inert atmosphere 4.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: thionyl chloride / toluene / 6 h / 40 °C 2.1: aluminum (III) chloride / 10 h / 70 °C 3.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 3.2: 10 - 30 °C / Inert atmosphere 4.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme |
-
-
25519-79-3
1-(4-benzoylpiperidin-1-yl)ethanone

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 1.2: 10 - 30 °C / Inert atmosphere 2.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme |
-
-
59084-16-1
1-acetylpiperidine-4-carbonyl chloride

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: aluminum (III) chloride / 10 h / 70 °C 2.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 2.2: 10 - 30 °C / Inert atmosphere 3.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: aluminum (III) chloride / 10 h / 70 °C 2.1: magnesium; iodine / tetrahydrofuran / Inert atmosphere; Reflux 2.2: 10 - 30 °C / Inert atmosphere 3.1: hydrogenchloride / water; ethanol / 12 h / 65 - 97 °C View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: bromobenzene With magnesium In tetrahydrofuran Reflux; Stage #2: isonipecotic acid In tetrahydrofuran; toluene at 50 - 120℃; under 304.02 - 1140.08 Torr; for 11h; Inert atmosphere; | 415 g |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: lithium / 5,5-dimethyl-1,3-cyclohexadiene / Inert atmosphere; Reflux 2: acetonitrile / Reflux 3: potassium borohydride / methanol / 3 h / 10 - 20 °C 4: hydrogen; palladium(II) hydroxide / methanol / 5 h / 10 - 20 °C / 258.58 Torr / Autoclave View Scheme | |
| Multi-step reaction with 4 steps 1: lithium / 5,5-dimethyl-1,3-cyclohexadiene / Inert atmosphere; Reflux 2: acetonitrile / Reflux 3: lithium borohydride / methanol / 3 h / 10 - 20 °C 4: hydrogen; palladium(II) hydroxide / methanol / 5 h / 10 - 20 °C / 258.58 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: lithium / 5,5-dimethyl-1,3-cyclohexadiene / Inert atmosphere; Reflux 2: acetonitrile / Reflux 3: potassium borohydride / methanol / 3 h / 10 - 20 °C 4: hydrogen; palladium(II) hydroxide / methanol / 5 h / 10 - 20 °C / 258.58 Torr / Autoclave View Scheme | |
| Multi-step reaction with 4 steps 1: lithium / 5,5-dimethyl-1,3-cyclohexadiene / Inert atmosphere; Reflux 2: acetonitrile / Reflux 3: lithium borohydride / methanol / 3 h / 10 - 20 °C 4: hydrogen; palladium(II) hydroxide / methanol / 5 h / 10 - 20 °C / 258.58 Torr / Autoclave View Scheme |

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: potassium borohydride / methanol / 3 h / 10 - 20 °C 2: hydrogen; palladium(II) hydroxide / methanol / 5 h / 10 - 20 °C / 258.58 Torr / Autoclave View Scheme |
-
-
115-46-8
C18H21NO

-
-
50706-57-5
4-(diphenylmethylene)piperidine

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; | 100% |
| With trifluoroacetic acid In dichloromethane for 7h; Inert atmosphere; | 96% |
| With trifluoroacetic acid In dichloromethane at 20℃; for 9h; | 95% |
-
-
254453-61-7
[4-(4-chloro-1-oxobutyl)phenyl]methyl-propanedioic acid diethyl ester

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; toluene for 168h; Heating / reflux; | 99% |
| With potassium carbonate In water; toluene for 168h; Reflux; | 99% |
-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With [(4-acetamidophenyl)(fluorosulfonyl)amino]sulfonyl fluoride; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran at 20℃; for 0.166667h; Reagent/catalyst; Solvent; | 98% |
| With [(4-acetamidophenyl)(fluorosulfonyl)amino]sulfonyl fluoride; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran at 20℃; for 0.166667h; | 96% |
| With dmap; fluorosulfonyl fluoride; magnesium oxide In dichloromethane; water at 20℃; | 94% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 68℃; for 20h; Heating / reflux; | A n/a B 96.1% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 68℃; for 20h; Heating / reflux; | A n/a B 95.5% |

-
-
115-46-8
C18H21NO

-
-
5332-06-9
4-bromobutanenitrile

-
-
118419-86-6
1-(3-Cyanopropyl)-4-(hydroxydiphenylmethyl)piperidine

| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In butanone for 5h; Heating; | 95% |
-
-
115-46-8
C18H21NO

-
-
19841-73-7
4-(Diphenylmethyl)piperidine

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; trifluoroacetic acid at 0℃; for 0.75h; | 95% |
| With sodium tetrahydroborate; trifluoroacetic acid In dichloromethane at 0℃; for 0.833333h; | 88% |
| With sodium tetrahydroborate In trifluoroacetic acid | |
| Multi-step reaction with 2 steps 1: trifluoroacetic acid / dichloromethane / 6 h / 20 °C 2: trimethylsilan / dichloromethane / 72 h / 20 °C View Scheme |
-
-
28547-33-3
1-(4-(tert-butyl)phenyl)-3-chloropropan-1-one

-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water; butanone at 85℃; for 16h; | 95% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 70℃; for 4h; | 95% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 70℃; for 4h; | 95% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate | 94% |
-
-
16686-11-6
2-(3-chloropropyl)-1,3-dioxolane

-
-
115-46-8
C18H21NO

-
-
185454-47-1
{1-[3-(1,3-dioxolan-2-yl)prop-1-yl]piperidin-4-yl}diphenylmethanol

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; potassium iodide In N,N-dimethyl-formamide at 50 - 68℃; Alkylation; | 94% |
-
-
115-46-8
C18H21NO

-
-
7693-46-1
4-Nitrophenyl chloroformate

-
-
1209468-67-6
4-nitrophenyl 4-(hydroxydiphenylmethyl)piperidine-1-carboxylate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 1h; | 94% |
| With triethylamine In dichloromethane for 2h; Inert atmosphere; | 32% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 70℃; for 4h; | 94% |
| With triethylamine In acetonitrile at 85℃; for 19h; | 94% |
-
-
115-46-8
C18H21NO

-
-
16004-15-2
1-bromomethyl-4-iodobenzene

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 70℃; for 4h; | 94% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 70℃; for 4h; | 92% |
-
-
115-46-8
C18H21NO

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 70℃; for 4h; | 92% |
| With triethylamine In acetonitrile at 50℃; for 20h; | 92% |
-
-
115-46-8
C18H21NO

-
-
1444336-77-9
3-(benzyloxy)-6-bromo-2,4,5-trimethylpyridine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate In toluene for 3h; Reflux; | 92% |
-
-
2386-60-9
perfluoro-1-butanesulfonyl fluoride

-
-
115-46-8
C18H21NO

-
-
1164112-42-8
[1-(butane-1-sulfonyl)piperidin-4-yl]diphenylmethanol

| Conditions | Yield |
|---|---|
| Stage #1: C18H21NO With triethylamine In dichloromethane at 0 - 5℃; for 0.166667h; Stage #2: perfluoro-1-butanesulfonyl fluoride In dichloromethane at 0 - 20℃; | 91% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 70℃; for 4h; | 91% |
-
-
169280-06-2
2-(4-cyclopropanecarbonyl-phenyl)-2-methyl-propionitrile

-
-
115-46-8
C18H21NO

-
-
394222-36-7
2-(4-(4-(4-(hydroxydiphenylmethyl)piperidin-1-yl)butanoyl)phenyl)-2-methylpropanenitrile

| Conditions | Yield |
|---|---|
| With lithium perchlorate In toluene at 150℃; for 4h; Reagent/catalyst; Temperature; Concentration; Solvent; | 91% |
-
-
115-46-8
C18H21NO

-
-
1694-92-4
2-Nitrobenzenesulfonyl chloride

-
-
1152785-34-6
[1-(2-nitrobenzenesulfonyl)piperidin-4-yl](diphenyl)methanol

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 5h; | 90% |
| Stage #1: C18H21NO With triethylamine In dichloromethane at 0 - 5℃; for 0.166667h; Stage #2: 2-Nitrobenzenesulfonyl chloride In dichloromethane at 0 - 20℃; | 87% |
-
-
115-46-8
C18H21NO

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
852860-46-9
diphenyl[1-(toluene-4-sulfonyl)piperidin-4-yl]methanol

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 90% |
| Stage #1: C18H21NO With triethylamine In dichloromethane at 0 - 5℃; for 0.166667h; Stage #2: p-toluenesulfonyl chloride In dichloromethane at 0 - 20℃; | 90% |
-
-
115-46-8
C18H21NO

-
-
98-09-9
benzenesulfonyl chloride

-
-
950054-08-7
(1-benzenesulfonylpiperidin-4-yl)diphenylmethanol

| Conditions | Yield |
|---|---|
| Stage #1: C18H21NO With triethylamine In dichloromethane at 0 - 5℃; for 0.166667h; Stage #2: benzenesulfonyl chloride In dichloromethane at 0 - 20℃; | 90% |
-
-
1347739-33-6
2-chloro-3-((4-chlorobenzyl)thio)naphthalene-1,4-dione

-
-
115-46-8
C18H21NO

-
-
1404074-76-5
2-((4-chlorobenzyl)thio)-3-(4-(hydroxydiphenylmethyl)piperidin-1-yl)naphthalene-1,4-dione

| Conditions | Yield |
|---|---|
| With sodium carbonate In dichloromethane for 4 - 6h; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: C18H21NO With triethylamine In dichloromethane for 0.166667h; Stage #2: 4-chlorobenzenesulfonyl chloride In dichloromethane at 20℃; for 5h; Further stages.; | 89% |
| Stage #1: C18H21NO With triethylamine In dichloromethane at 0 - 5℃; for 0.166667h; Stage #2: 4-chlorobenzenesulfonyl chloride In dichloromethane at 0 - 20℃; | 83% |
-
-
80466-79-1
isoxazole-4-sulfonyl chloride

-
-
115-46-8
C18H21NO

-
-
1164112-38-2
(1-(3,5-dimethyl-2,3-dihydro-isoxazole-4-sulfonyl)-piperidin-4-yl)-diphenyl-methanol

| Conditions | Yield |
|---|---|
| Stage #1: C18H21NO With triethylamine In dichloromethane at 0 - 5℃; for 0.166667h; Stage #2: isoxazole-4-sulfonyl chloride In dichloromethane at 0 - 20℃; | 89% |
alpha,alpha-Diphenyl-4-piperidinomethanol Specification
The alpha,alpha-Diphenyl-4-piperidinomethanol, with the CAS registry number 115-46-8, is also known as Azacyclonol. It belongs to the product categories of Pharmaceutical Intermediates; Chemical intermediate for Fexofenadine HCl; Intermediatesoffexofenadine. Its EINECS number is 204-092-5. This chemical's molecular formula is C18H21NO and molecular weight is 267.37. What's more, its systematic name is Diphenyl(4-piperidinyl)methanol. Its classification code is Drug / Therapeutic Agent. This chemical is the intermediates of Fexofenadine hydrochloride.
Physical properties of alpha,alpha-Diphenyl-4-piperidinomethanol are: (1)ACD/LogP: 2.736; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.36; (4)ACD/LogD (pH 7.4): 0.07; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.00; (8)ACD/KOC (pH 7.4): 1.59; (9)#H bond acceptors: 2; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 32.26 Å2; (13)Index of Refraction: 1.584; (14)Molar Refractivity: 81.078 cm3; (15)Molar Volume: 242.242 cm3; (16)Polarizability: 32.142×10-24cm3; (17)Surface Tension: 44.87 dyne/cm; (18)Density: 1.104 g/cm3; (19)Flash Point: 142.033 °C; (20)Enthalpy of Vaporization: 74.14 kJ/mol; (21)Boiling Point: 445.491 °C at 760 mmHg; (22)Vapour Pressure: 0 mmHg at 25°C.
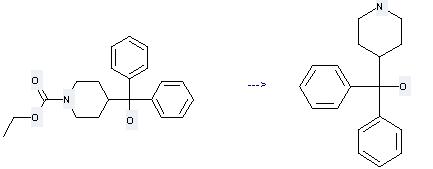
Preparation of alpha,alpha-Diphenyl-4-piperidinomethanol: this chemical can be prepared by 4-(Hydroxy-diphenyl-methyl)-1-ethoxycarbonyl-piperidin by heating. This reaction will need reagent 50% KOH and solvent ethanol with the reaction time of 24 hours. The yield is about 75%.
Uses of alpha,alpha-Diphenyl-4-piperidinomethanol: it can be used to produce (1-Methyl-[4]piperidyl)-diphenyl-methanol. It will need reagent NaBH4. The yield is about 94%.
![alpha,alpha-Diphenyl-4-piperidinomethanol can be used to produce (1-Methyl-[4]piperidyl)-diphenyl-methanol](/UserFilesUpload/Uses of alpha,alpha-Diphenyl-4-piperidinomethanol.jpeg)
When you are using this chemical, please be cautious about it as the following:
This chemical is irritating to eyes, respiratory system and skin. When using it, you must avoid contact with skin and eyes.
You can still convert the following datas into molecular structure:
(1)SMILES: OC(c1ccccc1)(c2ccccc2)C3CCNCC3
(2)Std. InChI: InChI=1S/C18H21NO/c20-18(15-7-3-1-4-8-15,16-9-5-2-6-10-16)17-11-13-19-14-12-17/h1-10,17,19-20H,11-14H2
(3)Std. InChIKey: ZMISODWVFHHWNR-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 220mg/kg (220mg/kg) | Psychopharmacology Service Center, Bulletin. Vol. 2, Pg. 17, 1963. | |
| mouse | LD50 | intravenous | 177mg/kg (177mg/kg) | Psychopharmacology Service Center, Bulletin. Vol. 2, Pg. 17, 1963. | |
| mouse | LD50 | oral | 650mg/kg (650mg/kg) | Psychopharmacology Service Center, Bulletin. Vol. 2, Pg. 17, 1963. | |
| mouse | LD50 | subcutaneous | 350mg/kg (350mg/kg) | Psychopharmacology Service Center, Bulletin. Vol. 2, Pg. 17, 1963. |
Related Products
- alpha-((2-Methoxyethoxy)methyl)-omega-hydroxypoly(oxy-1,2-ethanediyl) coco alkyl ethers
- alpha-((2-Methylpropoxy)methyl)pyrrolidine-1-ethanol
- alpha-(1,2,3,6-Tetrahydrophthalimido)glutarimide
- alpha-(1-Aminoethyl)-2,4-dimethoxybenzyl alcohol hydrochloride
- alpha-(1-Aminoethyl)-4-methoxybenzyl alcohol hydrochloride
- alpha-(2,3-Dimethylphenyl)-1-(trityl)-1H-imidazole-4-methanol
- alpha-(2,4-Dichlorophenyl)-1H-imidazole-1-ethanol
- alpha-(2-Chloroehtyl)-2-thiophenemethanol
- alpha-(3-(1,1-Dimethylethyl)-2-hydroxyphenyl)-1-methyl-5-nitro-1H-imidazole-2-methanol
- alpha-(4-Biphenylyloxy)propionic acid
- 115473-15-9
- 1154-78-5
- 1154-82-1
- 1154870-59-3
- 1154870-85-5
- 1154871-02-9
- 1155-00-6
- 1155058-96-0
- 115509-13-2
- 115514-66-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View