-
Name
DIBUTYL CARBONATE
- EINECS 208-816-0
- CAS No. 542-52-9
- Article Data100
- CAS DataBase
- Density 0.932 g/cm3
- Solubility
- Melting Point < 25 °C
- Formula C9H18O3
- Boiling Point 207.2 °C at 760 mmHg
- Molecular Weight 174.24
- Flash Point 76 °C
- Transport Information
- Appearance liquid
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Di-n-butylcarbonate;Dibutyl carbonate;NSC 8462;
- PSA 35.53000
- LogP 2.73980
Synthetic route

| Conditions | Yield |
|---|---|
| 1,1,3,3-tetra-n-butyl-1,3-di(n-butyloxy)-distanoxane; di-n-butyl-di(n-butyloxy)tin at 120℃; for 4h; Industry scale; Autoclave; | 99% |
| Stage #1: butan-1-ol; 1,1,3,3-tetra-n-butyl-1,3-di(n-butyloxy)-distanoxane; di-n-butyl-di(n-butyloxy)tin In water at 160℃; under 1125.11 Torr; Industry scale; Stage #2: carbon dioxide; 1,1,3,3-tetra-n-butyl-1,3-di(n-butyloxy)-distanoxane; di-n-butyl-di(n-butyloxy)tin In water at 120℃; under 30003 Torr; for 600h; Autoclave; Industry scale; | 99% |
| With 2-Cyanopyridine; cerium(IV) oxide at 119.84℃; under 37503.8 Torr; for 24h; Autoclave; | 87% |

| Conditions | Yield |
|---|---|
| at 120℃; for 4h; Autoclave; | 99% |
| at 180℃; under 90009 Torr; for 1h; Autoclave; | 55.2% |
| at 120℃; for 4h; Autoclave; Industry scale; | |
| 1,1,3,3-tetra-n-butyl-1,3-di(n-butyloxy)-distanoxane at 120℃; under 30003 Torr; for 4h; Industry scale; Autoclave; | |
| In neat (no solvent) at 180℃; under 90009 Torr; for 1h; Pressure; | 62 %Chromat. |

| Conditions | Yield |
|---|---|
| Heating; | A 96% B n/a |


| Conditions | Yield |
|---|---|
| Heating; | 96% |

| Conditions | Yield |
|---|---|
| With nickel diacetate; triphenylphosphine at 110 - 170℃; for 20h; | 95% |
| With zinc(II) oxide at 223.84℃; for 7h; Catalytic behavior; Reagent/catalyst; Temperature; Autoclave; | 36.2 %Chromat. |
-
-
2171-74-6
1,3-benzodioxol-2-one

-
-
71-36-3
butan-1-ol

-
A
-
542-52-9
Dibutyl carbonate

-
B
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With sodium methylate In neat (no solvent) at 60℃; for 1h; Catalytic behavior; Temperature; Reagent/catalyst; Inert atmosphere; | A 58% B 90% |
| With sodium methylate at 60℃; for 1h; Reagent/catalyst; Temperature; Inert atmosphere; | A 71 %Chromat. B n/a |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 190℃; under 1125.11 Torr; for 70h; Temperature; Pressure; Solvent; | 89.4% |

| Conditions | Yield |
|---|---|
| With MgZn1.7Al hydrotalcite calcined at 450°C at 230℃; under 15001.5 Torr; for 4h; Catalytic behavior; Autoclave; | A 9% B 85% |
| With MgZn1.7Al hydrotalcite calcined at 450°C at 200℃; under 15001.5 Torr; for 4h; Catalytic behavior; Time; Autoclave; | A 63% B 22% |
| calcined Yb(NO3)3.6H2O at 180℃; under 6460.43 Torr; for 6h; Inert atmosphere; Autoclave; | A 12.8% B 43.1% |
| With di(n-butyl)tin oxide In diphenylether at 170 - 200℃; for 8h; |

-
-
1351865-61-6
tert-butyl 3-bromodiazirine-3-carboxylate

-
-
2372-45-4
sodium butanolate

-
A
-
2568-90-3
di-n-butyloxymethane

-
B
-
542-52-9
Dibutyl carbonate

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide; butan-1-ol at 0 - 20℃; for 0.333333h; | A 31% B 82% |

-
-
2372-45-4
sodium butanolate

-
-
792950-89-1
butyl 3-bromo-3H-diazirine-3-carboxylate

-
A
-
2568-90-3
di-n-butyloxymethane

-
B
-
542-52-9
Dibutyl carbonate

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide; butan-1-ol at 0 - 20℃; for 0.333333h; | A 35% B 75% |


| Conditions | Yield |
|---|---|
| With tetraethylammonium L-prolinate at 110℃; for 4h; Reagent/catalyst; Temperature; Time; | 72% |
| With C5H8NO2(1-)*C8H20N(1+) at 110℃; for 4h; Kinetics; Reagent/catalyst; Temperature; Time; | |
| With Ru2O/SrO/HgO/SiO2(K2O/MgO/ZnO/SiO2) catalyst at 130 - 140℃; under 1125.11 - 1500.15 Torr; Temperature; Reagent/catalyst; Pressure; Inert atmosphere; Green chemistry; |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate under 8 Torr; Heating; | A 71% B n/a |

| Conditions | Yield |
|---|---|
| With auphen; potassium iodide In butan-1-ol at 80℃; under 22502.3 Torr; for 5h; Autoclave; | 70.3% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; potassium carbonate In N,N-dimethyl-formamide at 60℃; | 70% |
| With N,N,N',N'-tetramethylguanidine In neat liquid at 60℃; for 6h; Mechanism; Reagent/catalyst; Temperature; |


| Conditions | Yield |
|---|---|
| With mercury(II) diacetate at 210℃; for 2h; | 68% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 180℃; under 90009 Torr; for 1h; Pressure; | 67% |

| Conditions | Yield |
|---|---|
| di(n-butyl)tin oxide In diphenylether at 130 - 210℃; for 4h; | 66.7% |
| di(n-butyl)tin oxide In diphenylether at 5 - 235℃; for 4h; Heating / reflux; | 66.7% |
| With urea | 62.2% |

| Conditions | Yield |
|---|---|
| In hexane Heating; | 64% |

| Conditions | Yield |
|---|---|
| 62.2% |


| Conditions | Yield |
|---|---|
| In hexane Heating; | 60% |
-
-
1351865-61-6
tert-butyl 3-bromodiazirine-3-carboxylate

-
-
2372-45-4
sodium butanolate

-
B
-
542-52-9
Dibutyl carbonate

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at -15℃; | A 27% B 52% |


| Conditions | Yield |
|---|---|
| bis(tri-n-butyltin)oxide In N,N-dimethyl-formamide at 100℃; for 10h; | 51% |

| Conditions | Yield |
|---|---|
| Stage #1: carbon dioxide With 1-butyl-3-methyl-1H-imidazol-3-iumhydrogencarbonate at 25℃; under 760.051 Torr; for 6h; Stage #2: 1-iodo-butane; butan-1-ol at 25℃; under 760.051 Torr; for 12h; | 44.3% |

-
-
201230-82-2
carbon monoxide

-
-
2372-45-4
sodium butanolate

-
A
-
542-52-9
Dibutyl carbonate

-
B
-
2050-60-4
di-n-butyl oxalate

| Conditions | Yield |
|---|---|
| With [nickel(II)(pyridine)4(chloride)2] In tetrahydrofuran under 30002.4 Torr; for 20h; Ambient temperature; | A 11% B 41% |
| With dibromo(N,N,N',N'-tetramethylethane-1,2-diamine)nickel(II) In tetrahydrofuran under 30002.4 Torr; for 20h; Ambient temperature; | A 35% B 12% |


-
-
14076-99-4, 20154-73-8, 36829-43-3
[nickel(II)(pyridine)4(chloride)2]

-
A
-
542-52-9
Dibutyl carbonate

-
B
-
2050-60-4
di-n-butyl oxalate

-
C
-
13463-39-3, 71564-36-8
tetracarbonyl nickel

| Conditions | Yield |
|---|---|
| With carbon monoxide In tetrahydrofuran byproducts: NaCl; addn. of n-C4H9ONa to suspn. of Ni compd., CO (40 bar), room temp., 20h, hydrolysis with H2SO4, sepn. of organic phase, dried; yield of organic products detected by chromy.; | A 11% B 41% C n/a |
| With carbon monoxide In tetrahydrofuran byproducts: NaCl; addn. of n-C4H9ONa to suspn. of Ni compd., CO (40 bar), 75°C, 20h, hydrolysis with H2SO4, sepn. of organic phase, dried; yield of organic products detected by chromy.; | A 22% B 28% C n/a |


| Conditions | Yield |
|---|---|
| at 180℃; under 90009 Torr; for 1h; Autoclave; | 39.5% |
| In neat (no solvent) at 180℃; under 90009 Torr; for 1h; Pressure; | 42 %Chromat. |
-
-
787624-20-8, 14057-03-5
bis(triphenylphosphine)nickel(II) diiodide

-
A
-
542-52-9
Dibutyl carbonate

-
B
-
2050-60-4
di-n-butyl oxalate

-
C
-
13463-39-3, 71564-36-8
tetracarbonyl nickel

| Conditions | Yield |
|---|---|
| With carbon monoxide In tetrahydrofuran byproducts: NaI; addn. of n-C4H9ONa to suspn. of Ni compd., CO (40 bar), room temp., 20h, hydrolysis with H2SO4, sepn. of organic phase, dried; yield of organic products detected by chromy.; | A 9% B 38% C n/a |


-
-
14878-48-9
dibromo(N,N,N',N'-tetramethylethane-1,2-diamine)nickel(II)

-
A
-
542-52-9
Dibutyl carbonate

-
B
-
2050-60-4
di-n-butyl oxalate

-
C
-
13463-39-3, 71564-36-8
tetracarbonyl nickel

| Conditions | Yield |
|---|---|
| With carbon monoxide In tetrahydrofuran byproducts: NaI; addn. of n-C4H9ONa to suspn. of Ni compd., CO (40 bar), room temp., 20h, hydrolysis with H2SO4, sepn. of organic phase, dried; yield of organic products detected by chromy.; | A 35% B 12% C n/a |


-
A
-
542-52-9
Dibutyl carbonate

-
B
-
13463-39-3, 71564-36-8
tetracarbonyl nickel

| Conditions | Yield |
|---|---|
| With carbon monoxide In tetrahydrofuran byproducts: NaI; addn. of n-C4H9ONa to suspn. of Ni compd., CO (40 bar), room temp., 20h, hydrolysis with H2SO4, sepn. of organic phase, dried; yield of organic product detected by chromy.; | A 31% B n/a |

-
-
542-52-9
Dibutyl carbonate

-
-
95-80-7
4-methylbenzene-1,3-diamine

-
-
7469-49-0
1-methyl-2,4-bis-(butoxycarbonylamino)-benzene

| Conditions | Yield |
|---|---|
| With sodium butanolate at 120℃; for 0.5h; Inert atmosphere; | 99% |
-
-
50-00-0
formaldehyd

-
-
542-52-9
Dibutyl carbonate

-
-
616-34-2
methoxycarbonylmethylamine

-
-
1487424-91-8
N,N-bis(dibutoxyphosphinoylmethyl)glycine methyl ester

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 100℃; for 1h; Kabachnik-Fields Reaction; Microwave irradiation; | 93% |

-
-
50-00-0
formaldehyd

-
-
542-52-9
Dibutyl carbonate

-
-
459-73-4
GlyOEt*HCl

-
-
1487424-92-9
N,N-bis(dibutoxyphosphinoylmethyl)glycine ethyl ester

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 100℃; for 1h; Kabachnik-Fields Reaction; Microwave irradiation; | 93% |

-
-
124-09-4
1,6-Hexanediamine

-
-
542-52-9
Dibutyl carbonate

-
-
3066-67-9
1,6-bis-(n-butoxycarbonyl-amino)-hexane

| Conditions | Yield |
|---|---|
| In butan-1-ol Reflux; Large scale; | 92% |
| With sodium methylate In methanol at 80℃; Product distribution / selectivity; Inert atmosphere; | |
| With sodium methylate In methanol at 80℃; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In N,N,N,N,N,N-hexamethylphosphoric triamide at 140℃; for 5h; | 90% |

| Conditions | Yield |
|---|---|
| With silica gel-supported lanthanum(III) oxide at 150℃; for 12h; Autoclave; Inert atmosphere; | 87% |

| Conditions | Yield |
|---|---|
| With 5percent K-silica gel at 150℃; for 8h; | 80% |
| 5% K-silica gel at 150℃; for 8h; Conversion of starting material; | |
| 5% K-silica gel at 150℃; for 8h; Conversion of starting material; |

| Conditions | Yield |
|---|---|
| With silica gel-supported lanthanum(III) oxide at 150℃; for 6h; Autoclave; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| With sodium butanolate Reagent/catalyst; | 80% |

| Conditions | Yield |
|---|---|
| With silica gel-supported lanthanum(III) oxide at 150℃; for 12h; Autoclave; Inert atmosphere; | 75% |
-
-
124-09-4
1,6-Hexanediamine

-
-
542-52-9
Dibutyl carbonate

-
A
-
3066-67-9
1,6-bis-(n-butoxycarbonyl-amino)-hexane

| Conditions | Yield |
|---|---|
| In butan-1-ol Reflux; Large scale; | A 74% B 21% |


| Conditions | Yield |
|---|---|
| heating for 1-1.5 h at 140°C; | 69% |
n-Butyl carbonate Specification
The Carbonic acid, dibutyl ester, with the CAS registry number 542-52-9, is also known as Di-n-butylcarbonate. It belongs to the product category of Carbonates. Its EINECS number is 208-816-0. This chemical's molecular formula is C9H18O3 and molecular weight is 174.24. What's more, its systematic name is dibutyl carbonate.
Physical properties of Carbonic acid, dibutyl ester are: (1)ACD/LogP: 3.34; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 3.34; (4)ACD/LogD (pH 7.4): 3.34; (5)ACD/BCF (pH 5.5): 201.79; (6)ACD/BCF (pH 7.4): 201.79; (7)ACD/KOC (pH 5.5): 1554.03; (8)ACD/KOC (pH 7.4): 1554.03; (9)#H bond acceptors: 3; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 8; (12)Polar Surface Area: 35.53 Å2; (13)Index of Refraction: 1.419; (14)Molar Refractivity: 47.26 cm3; (15)Molar Volume: 186.9 cm3; (16)Polarizability: 18.73×10-24cm3; (17)Surface Tension: 29.2 dyne/cm; (18)Density: 0.932 g/cm3; (19)Flash Point: 76 °C; (20)Enthalpy of Vaporization: 44.34 kJ/mol; (21)Boiling Point: 207.2 °C at 760 mmHg; (22)Vapour Pressure: 0.228 mmHg at 25°C.
Preparation: this chemical can be prepared by 1-bromo-butane and Potassium carbonate at the temperature of 100 °C. This reaction will need solvent dimethylformamide and catalyst 5 mol percent hexabutyldistannoxane ((Bu3Sn)2O) with the reaction time of 10 hours. The yield is about 51%.
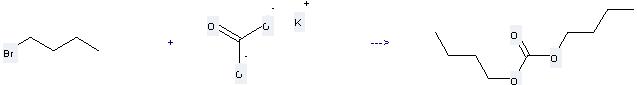
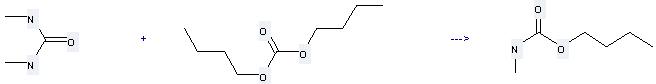
Uses of Carbonic acid, dibutyl ester: it can be used to produce O-Butyl-N-methyl-carbamat at the temperature of 150 °C. It will need reagent 5 percent K-silica gel with the reaction time of 8 hours. The yield is about 80%.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(OCCCC)OCCCC
(2)Std. InChI: InChI=1S/C9H18O3/c1-3-5-7-11-9(10)12-8-6-4-2/h3-8H2,1-2H3
(3)Std. InChIKey: QLVWOKQMDLQXNN-UHFFFAOYSA-N
Related Products
- n-BUTYL AMIDO SULFURYL AZIDE
- n-Butyl carbamate
- n-Butyl carbonate
- N-Butyl Cinnamate
- n-Butyl cyanoacetate
- N-Butyl cyclohexyl amine
- n-Butyl glycidyl ether
- N-Butyl Mesityl Oxide Oxalate
- n-Butyl methyl sulphide
- n-Butyl thiourea
- 54253-49-5
- 54253-62-2
- 5425-44-5
- 542-55-2
- 54255-79-7
- 542-56-3
- 54256-33-6
- 54256-45-0
- 5425-78-5
- 5425-81-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View