Simagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:107-87-9
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis
Cas:107-87-9
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
LIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc. We are specialized in chemical synthesis, process development of
Cas:107-87-9
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:107-87-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China and Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED intermediat
Cas:107-87-9
Min.Order:1 Kilogram
FOB Price: $4.0 / 5.0
Type:Lab/Research institutions
inquiryHebei yanxi chemical co.,LTD.
Hebei Yanxi Chemical Co., Ltd. is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low car
Shanghai Minstar Chemical Co., Ltd
Synonyms: METHYL PROPYL KETONE;METHYL N-PROPYL KETONE;ETHYLACETONE;FEMA 2842;2-PENTANONE;PENTAN-2-ONE;2-Oxopentane;2-Pentanon CAS: 107-87-9 MF: C5H10O MW: 86.13 EINECS: 203-528-1 Product Categories: Organics;ketone;ketone Flavor;ACS and Reagen
Cas:107-87-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
Minimum Order Qty. 10 Gram Supply Ability 500 Kilograms/Month Storage store in cool, dry, ventilated place 20℃ Delivery Time 3 business days after payment Payment Term TT,western union,Paypal,MoneyGram Package 10g,20g,50g,100g,500g,1KGS,
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:107-87-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colorless liquid Storage:sealed in a cool place Package:25kg/drum,or as per your request. Application:Used as a solvent, organic synthesis intermediate. Transportation:by air or by s
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Wuhan Fortuna Chemical Co.,Ltd
good quality and cheap price Appearance:colorless liquid Package:170kg/Steel drum Transportation:by sea Port:Shanghai
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:107-87-9
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Wuhan ZeShanCheng Biomedical Technology Co., Ltd.
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highl
Hangzhou ZeErRui Chemical Co., Ltd.
Hangzhou ZeErRui Chemical Co., Ltd. located in Lingang industrial areas, our plant covers an area of 6000 square meters.ZeErRui dedicated to the development, production and marketing of chemicals. We have earned ourselves a good reputation at home an
Hunan Longxianng Runhui Trading Co.,Ltd
107-87-9 2-PentanoneAppearance:white powder Storage:room tempurature Package:As required Application:medical Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small amount. By air(airport to airport) or by sea LCL/FCL for large amo
Synthetic route

| Conditions | Yield |
|---|---|
| With limonene.; palladium on activated charcoal for 0.25h; Heating; | 100% |
| With potassium bis(trimethylsilyl)amide In tetrahydrofuran at 0℃; for 6h; | 94.2% |
| With hydrogen; 1,5-hexadienerhodium(I)-chloride dimer In hexane for 3h; Ambient temperature; pH=7.6; | 71% |

| Conditions | Yield |
|---|---|
| With sodium dichromate; sulfuric acid; silica gel In dichloromethane at 20℃; for 0.5h; | 99% |
| With dihydrogen peroxide In water at 89.84℃; for 5h; | 98% |
| With pyridinium chlorochromate In chloroform at 16℃; for 168h; or 1-methyl imidazolium chlorochromate or imidazolium chlorochromate; | 97% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride; di(rhodium)tetracarbonyl dichloride In dichloromethane | 98% |
| With [Ru(η3:η3-C10H16)Cl2(benzimidazole)] In glycerol at 75℃; for 8h; Sealed tube; Inert atmosphere; Green chemistry; | 95% |
| With 2C25H28N2OP(1-)*2Cl(1-)*2Ru(2+) In tetrahydrofuran at 80℃; for 1h; Inert atmosphere; Schlenk technique; Sealed tube; | 88% |

| Conditions | Yield |
|---|---|
| With disodium chloro[1,3-bis(2,6-diisopropyl-4-sodiumsulfonatophenyl)imidazol-2-ylidene]gold(I); water at 100℃; for 1.17h; | 97% |
| With 1,3-bis{2,6-bis[bis(4-tert-butylphenyl)methyl]-4-methylphenyl}-1H-imidazol-2-ylidenegold(I) chloride; water; silver(I) triflimide In methanol at 80℃; for 0.25h; Temperature; | 88% |
| With hexafluoroantimonic acid; AuOH(1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene); water at 120℃; for 24h; | 40% |
-
-
111957-98-3, 625-31-0
Pent-4-en-2-ol

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With Grotjahn’s catalyst In [(2)H6]acetone at 70℃; for 1h; Reagent/catalyst; Solvent; Inert atmosphere; Glovebox; | 97% |
| With copper at 300℃; | |
| {CpRu[P(i-Pr)2(1-Me-4-tBu-imidazol-2-yl)]MeCN}*PF6 In acetone at 70℃; for 1h; | 97 % Spectr. |
-
-
14848-64-7
2-pentanol(d O-H)

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With pyridinium chlorochromate In chloroform at 16℃; for 2.8h; Rate constant; Product distribution; Mechanism; | 96.4% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; [RuCl2(η6-p-cymene){κ1-(P)-PPh2(OCH2CH2NMe2)}] In tetrahydrofuran at 75℃; for 0.0833333h; | A n/a B 95% |
-
-
57230-59-8
2-methyl-2-propyl-1,3-dithiolane

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide In acetic acid for 1.16667h; Ambient temperature; | 94% |
-
-
147050-27-9
2-Methyl-1-morpholino-1-pentene

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With potassium permanganate on Y-Zeolite In 1,2-dichloro-ethane for 6h; Ambient temperature; | 93% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; dimethyl sulfoxide at 70℃; for 9.25h; | 92.03% |
| With boron trifluoride diethyl etherate In diethyl ether for 4h; Ambient temperature; | 91% |
-
-
98-01-1
furfural

-
A
-
534-22-5
2-methylfuran

-
B
-
6032-29-7
(+/-)-2-pentanol

-
C
-
71-41-0
pentan-1-ol

-
D
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With Cu/SiO2; hydrogen at 220℃; under 760.051 Torr; for 0.5h; Catalytic behavior; Reagent/catalyst; Time; Temperature; Green chemistry; | A 89.5% B n/a C n/a D n/a |

-
-
98-01-1
furfural

-
A
-
96-47-9
2-methyltetrahydrofuran

-
B
-
534-22-5
2-methylfuran

-
C
-
6032-29-7
(+/-)-2-pentanol

-
D
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With hydrogen; Cu-based catalyst at 212℃; Product distribution; Further Variations:; Temperatures; reaction in vapour phase, fixed bed reactor; | A 3.3% B 88.6% C 4.8% D 2.7% |


-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With sulfuric acid In benzene for 4h; Heating; | 87% |

| Conditions | Yield |
|---|---|
| With caro's acid; silica gel In acetonitrile at 20℃; for 0.416667h; | 86% |
| With tellurium; sodium tetrahydroborate; water 1.) EtOH, 25 deg C, 30 min; Multistep reaction; |
-
-
26990-57-8
2-methyl-2-n-propyl-1,3-oxathiolane

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With Montmorillonite K10 In benzene for 2.5h; Heating; | 86% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; [RuCl2(η6-p-cymene){κ1-(P)-PPh2(OCH2CH2NMe2)}] In tetrahydrofuran at 75℃; for 5.41667h; | A n/a B 85% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; β‐cyclodextrin In water; acetone at 20℃; for 0.333333h; | 84% |
| With sodium bromate; ion exchange resin at 25 - 30℃; for 0.25h; ultrasonic irradiation; | 97 % Chromat. |

| Conditions | Yield |
|---|---|
| With oxygen; Cu-X zeolite In 1,2-dichloro-ethane; acetonitrile at 50℃; for 6h; Oxidation; Oxidative cleavage; | A 83% B 80% |

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With calcium hypochlorite; montmorillonite K-10 In chloroform at 20℃; for 2.75h; | 82% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid; zinc for 2h; Heating; | A 79% B 21% |
| With hydrogenchloride; acetic acid; zinc for 2h; Product distribution; Mechanism; Heating; further co-reagents: LiCl, TFA; also amalgamated zinc; further β-dicarbonal substrates; | A 79% B 21% |
-
-
123-54-6
acetylacetone

-
A
-
6032-29-7
(+/-)-2-pentanol

-
B
-
107-87-9
2-Pentanone

-
C
-
1825-14-5, 3950-21-8, 36402-52-5, 42075-32-1, 72345-23-4, 625-69-4
pentane-1,3-diol

| Conditions | Yield |
|---|---|
| With samarium diiodide; water In tetrahydrofuran at 20℃; for 0.141667h; | A 78% B 6% C 6% |


| Conditions | Yield |
|---|---|
| With hydrogen; In diethylene glycol dimethyl ether; water at 30℃; under 735.5 Torr; for 5h; | A 7.8% B 77.3% |
| With nickel kieselguhr at 100℃; under 88260.9 Torr; Hydrogenation; | |
| With nickel kieselguhr at 100 - 160℃; under 88260.9 Torr; Hydrogenation.Hydrieren des Reaktionsprodukts an Kupferchromit; (+-)-pentanol-(2); |
-
-
106-31-0
butanoic acid anhydride

-
A
-
99668-71-0
Co(OCO-n-C3H7)

-
B
-
187737-37-7
propene

-
C
-
34557-54-5
methane

-
D
-
74-98-6
propane

-
E
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran in THF at -40-20°C; | A n/a B 7% C 6% D 2% E 76% |


| Conditions | Yield |
|---|---|
| With lead(IV) acetate; potassium bromide In benzene at 80℃; | A 75% B 3% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; Dess-Martin periodane In water; acetonitrile at 50℃; Wacker-Tsuji Olefin Oxidation; Inert atmosphere; | 72% |
| With dihydrogen peroxide In water; acetonitrile at 55℃; for 12h; Wacker Oxidation; | 71% |
| With oxygen; cetyltrimethylammonim bromide; copper dichloride; palladium dichloride In water; benzene at 80℃; under 2327.2 Torr; Product distribution; | 48% |

| Conditions | Yield |
|---|---|
| With tetraethylammonium bromate In ethanol for 5h; Heating; | 72% |
| With potassium carbonate In water at 9.9℃; Kinetics; Mechanism; Thermodynamic data; E(activ.), ΔG(excit.), ΔH(excit.), ΔS(excit.); other temperatures; |
-
-
105-30-6
2-methylpentan-1-ol

-
A
-
6032-29-7
(+/-)-2-pentanol

-
B
-
97-61-0, 22160-39-0
2-Methylpentanoic acid

-
C
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; methyltrioctylammonium tetrakis(oxodiperoxotungsto)phosphate at 90℃; for 6h; | A n/a B 70% C n/a |

-
-
123-15-9
2-methylvaleraldehyde

-
A
-
6032-29-7
(+/-)-2-pentanol

-
B
-
97-61-0, 22160-39-0
2-Methylpentanoic acid

-
C
-
58368-66-4
1-methylbutyl formate

-
D
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; methyltrioctylammonium tetrakis(oxodiperoxotungsto)phosphate at 90℃; for 0.5h; | A n/a B 70% C 7% D n/a |


| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In diethyl ether for 2h; -78 deg C; | 69% |

| Conditions | Yield |
|---|---|
| With water; palladium dichloride at 25℃; under 1300 Torr; for 22h; in microemulsion system, closed reactor; | 66% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 0℃; | 100% |
| With tetra(n-butyl)ammonium hydroxide In water; dimethyl sulfoxide at 5℃; for 1h; Favorskii-Babayan Synthesis; | 90% |
| With diethyl ether; potassium 2-methylbutan-2-olate |

| Conditions | Yield |
|---|---|
| barium dihydroxide In ethanol for 1h; Heating; | 100% |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
107-87-9
2-Pentanone

-
-
91390-80-6
2-methyl-2-(trimethylsiloxy)pentanenitrile

| Conditions | Yield |
|---|---|
| With trans-{(iBu)2ATIGeiPr}2Pt(CN)2 In chloroform-d1 at 50℃; for 6h; Catalytic behavior; Schlenk technique; Glovebox; | 100% |
| With C29H38AlN4O2(1+)*CF3O3S(1-) In neat (no solvent) at 20℃; for 0.0833333h; Catalytic behavior; Inert atmosphere; Schlenk technique; | 99% |
| With gold(III) chloride In dichloromethane at 20℃; for 0.5h; | 92% |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In diethyl ether for 3h; Heating; | 100% |
| sulfur dioxide In benzene Heating; | 72% |
| With toluene-4-sulfonic acid In benzene Heating; |
-
-
501-94-0
p-hydroxyphenethyl alcohol

-
-
107-87-9
2-Pentanone

-
-
386263-87-2
2-(4-hydroxylphenyl)ethyl butyrate

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 45℃; for 24h; Enzymatic reaction; | 100% |
-
-
621-87-4
3-phenoxy-2-propanone

-
-
107-87-9
2-Pentanone

-
A
-
625-30-9, 33985-20-5, 54542-13-1, 63493-28-7
(R)‐2‐aminopentane

-
B
-
45972-74-5
R-(-)-1-phenoxy-2-propanamine

| Conditions | Yield |
|---|---|
| With Candida boidinii formate dehydrogenase; pyridoxal 5'-phosphate; Aspergillus terreus ω-trans aminase; Lysinibacillus fusiformis leucine dehydrogenase; ammonium formate; nicotinamide adenine dinucleotide In aq. buffer at 30℃; for 24h; pH=8.8; Catalytic behavior; Green chemistry; Enzymatic reaction; | A 99.2% B 99.8% |

-
-
459-03-0
4-fluorophenyl acetone

-
-
107-87-9
2-Pentanone

-
A
-
625-30-9, 33985-20-5, 54542-13-1, 63493-28-7
(R)‐2‐aminopentane

-
B
-
72522-20-4
(R)-(-)-α-methyl-β-(4-fluorophenyl)ethylamine

| Conditions | Yield |
|---|---|
| With Candida boidinii formate dehydrogenase; pyridoxal 5'-phosphate; Aspergillus terreus ω-trans aminase; Lysinibacillus fusiformis leucine dehydrogenase; ammonium formate; nicotinamide adenine dinucleotide In aq. buffer at 30℃; for 24h; pH=8.8; Catalytic behavior; Green chemistry; Enzymatic reaction; | A 99.7% B 99.4% |

-
-
122-84-9
4-methoxybenzyl methyl ketone

-
-
107-87-9
2-Pentanone

-
A
-
625-30-9, 33985-20-5, 54542-13-1, 63493-28-7
(R)‐2‐aminopentane

-
B
-
58993-79-6
(R)-(-)-p-methoxyamphetamine

| Conditions | Yield |
|---|---|
| With Candida boidinii formate dehydrogenase; pyridoxal 5'-phosphate; Aspergillus terreus ω-trans aminase; Lysinibacillus fusiformis leucine dehydrogenase; ammonium formate; nicotinamide adenine dinucleotide In aq. buffer at 30℃; for 24h; pH=8.8; Catalytic behavior; Green chemistry; Enzymatic reaction; | A 99.7% B 99.4% |

-
-
107-87-9
2-Pentanone

-
-
5586-88-9
1-(4-chlorophenyl)propan-2-one

-
A
-
625-30-9, 33985-20-5, 54542-13-1, 63493-28-7
(R)‐2‐aminopentane

| Conditions | Yield |
|---|---|
| With Candida boidinii formate dehydrogenase; pyridoxal 5'-phosphate; Aspergillus terreus ω-trans aminase; Lysinibacillus fusiformis leucine dehydrogenase; ammonium formate; nicotinamide adenine dinucleotide In aq. buffer at 30℃; for 24h; pH=8.8; Catalytic behavior; Green chemistry; Enzymatic reaction; | A 99.3% B 99.1% |

-
-
2550-26-7
4-Phenyl-2-butanone

-
-
107-87-9
2-Pentanone

-
A
-
625-30-9, 33985-20-5, 54542-13-1, 63493-28-7
(R)‐2‐aminopentane

-
B
-
937-52-0
(R)-1-methyl-3-phenylpropylamine

| Conditions | Yield |
|---|---|
| With Candida boidinii formate dehydrogenase; pyridoxal 5'-phosphate; Aspergillus terreus ω-trans aminase; Lysinibacillus fusiformis leucine dehydrogenase; ammonium formate; nicotinamide adenine dinucleotide In aq. buffer at 30℃; for 24h; pH=8.8; Catalytic behavior; Reagent/catalyst; pH-value; Green chemistry; Enzymatic reaction; | A 99.1% B 96.4% |


| Conditions | Yield |
|---|---|
| With sodium isopropylate; acetonitrile In isopropyl alcohol at 80℃; for 1h; Catalytic behavior; | 99% |
| With formic acid; sodium formate; (η5-C4Ph4COHOC4Ph4-η5)(μ-H)(CO)4Ru2 In water at 100℃; for 3h; | 98% |
| With C40H37ClN2PRuS(1+)*C24H20B(1-); isopropyl alcohol; potassium hydroxide at 82℃; for 2h; Catalytic behavior; | 97.7% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Pentanone; benzylamine With formic acid; chlorido(8-quinolinolato-k2N,O)(η5-pentamethylcyclo-pentadienyl)rhodium(III) In ethyl acetate at 0 - 40℃; Inert atmosphere; Cooling with ice; Schlenk tube; Stage #2: With sodium hydrogencarbonate In water; ethyl acetate Product distribution / selectivity; | 99% |
| With 5-ethyl-2-methylpyridine borane In methanol at 20℃; | 84% |
| With 4 A molecular sieve; borane pyridine complex In methanol for 16h; | 80% |
| With 2-picoline borane complex; acetic acid In methanol at 20℃; for 6h; | 79% |

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| In water byproducts: water; addn. of 10% water to suspn. of Ni-complex in PrCOMe (dissoln.), refluxing for 10 min; evapn. (reduced pressure), recrystn. (Me2CO/Et2O); elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| With titanium(IV) tetrabutoxide; triethylsilyl chloride In dichloromethane at 20℃; | A n/a B 99% |

-
-
1260828-48-5
C8H4BrClF3NO

-
-
107-87-9
2-Pentanone

-
-
1448544-33-9
8-bromo-6-chloro-2-propyl-4-(trifluoromethyl)quinoline

| Conditions | Yield |
|---|---|
| With L-proline potassium salt In dimethyl sulfoxide at 20℃; for 0.416667h; Friedlaender Quinoline Synthesis; regioselective reaction; | 99% |
-
-
107-87-9
2-Pentanone

-
-
93-03-8
(3,4-dimethoxyphenyl)methanol

-
-
39728-57-9
1-(3,4-dimethoxyphenyl)hexan-3-one

| Conditions | Yield |
|---|---|
| With potassium phosphate; 5%-palladium/activated carbon In toluene at 100℃; for 24h; Reagent/catalyst; Temperature; Inert atmosphere; | 99% |
| With potassium phosphate In toluene at 80℃; for 24h; Inert atmosphere; | 79% |
-
-
77086-38-5
ketene t-butyldimethylsilyl methyl acetal

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| Stage #1: 2-Pentanone With bis(trifluoromethanesulfonyl)amide In diethyl ether at -78 - 23℃; Mukaiyama Aldol Addition; Schlenk technique; Inert atmosphere; Stage #2: ketene t-butyldimethylsilyl methyl acetal In diethyl ether at -20℃; for 0.5h; Mukaiyama Aldol Addition; Schlenk technique; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With Nafion-H resin In 1,2-dimethoxyethane at 70℃; for 8h; | 98% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In ethanol at 20℃; for 1.66667h; | 98% |

-
-
864847-09-6
2-allyl-2,3,3-tris-ethoxycarbonyl-hept-5-ynoic acid ethyl ester

-
-
107-87-9
2-Pentanone

-
-
1017603-41-6
1,3-dimethyl-3-propyl-3,4,4a,5-tetrahydro-8H-isochromene-6,6,7,7-tetracarboxylic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| With 3 A molecular sieve; 1,3-bis-(2,6-diisopropylphenyl)-imidazol-2-ylidene; bis(1,5-cyclooctadiene)nickel (0) In toluene at 20℃; | 98% |
-
-
107-87-9
2-Pentanone

-
-
154598-53-5
1-(2-amino-5-chloro-phenyl)-2,2,2-trifluoro-ethanone

-
-
1042738-31-7
6-chloro-2-propyl-4-(trifluoromethyl)quinoline

| Conditions | Yield |
|---|---|
| With L-proline potassium salt In dimethyl sulfoxide at 20℃; for 0.583333h; Reagent/catalyst; Time; Temperature; Friedlaender Quinoline Synthesis; regioselective reaction; | 98% |
| With L-proline In dimethyl sulfoxide at 50℃; for 48h; Friedlaender synthesis; regioselective reaction; | 93% |

-
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With L-proline In dimethyl sulfoxide at 20℃; for 24h; Mannich Aminomethylation; enantioselective reaction; | 97% |
-
-
107-87-9
2-Pentanone

-
-
26184-62-3
(S)-2-pentanol

| Conditions | Yield |
|---|---|
| With dried cells of Geotrichum candidum; NAD; isopropyl alcohol In various solvent(s) at 30℃; for 9h; pH=7.0; Enzymatic reaction; | 96% |
| With glucose dehydrogenase; ketoreductase Kred-108; glucose; NADPH at 37℃; for 24h; pH=6.9; aq. phosphate buffer; Enzymatic reaction; optical yield given as %ee; stereoselective reaction; | 92% |
| Daucas carota root; extract of In water at 35 - 40℃; for 88h; pH=7.5; Conversion of starting material; Enzymatic reaction; Aqueous phosphate buffer; | 49% |
-
-
107-87-9
2-Pentanone

-
-
1099-45-2
ethyl (triphenylphosphoranylidene)acetate

-
-
15677-00-6
ethyl 3-methylhex-2-enoate

| Conditions | Yield |
|---|---|
| In xylene at 50℃; under 6750540 Torr; for 30h; | 96% |
| With benzoic acid |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View




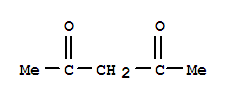











 F,
F, Xn
Xn