Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem’s R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantiti
Hangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service an
Hangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance Colorless or light yellow liquid Colorless or light yellow liquid Purity of GC
Cas:104-87-0
Min.Order:1 Kilogram
FOB Price: $15.0 / 50.0
Type:Manufacturers
inquiryJinan Finer Chemical Co., Ltd
Product description: Product name p-Tolualdehyde CAS number 104-87-0 Assay ≥99% Appearance Colorless to yellowish liquid Capacity 200mt/year Application Perfume/pharmaceutica
Cas:104-87-0
Min.Order:100 Gram
FOB Price: $12.0
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:104-87-0
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryCOLORCOM LTD.
Colorcom is a global leader in industrial chemical manufacturing and is continuously innovating and transforming to exceed client expectations and industry standards. Colorcom prides itself on superior customer and technical focus, while focusing on
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new ap
Hebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developm
Cas:104-87-0
Min.Order:1 Metric Ton
FOB Price: $1.0 / 3.0
Type:Trading Company
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hubei DiBo chemical co., LTD
Name:p-Tolualdehyde CAS No. : 104-87-0 Grade: It is the main flavor of Hawthorn flower flavor. It can also be used in flower fragrance and new flower mowing, fragrance, fragrance and other non fragrance essence such as lilac, orchid, sunflower, aca
Cas:104-87-0
Min.Order:25 Kilogram
FOB Price: $1.0 / 2.0
Type:Other
inquiryHenan Tianfu Chemical Co., Ltd.
Product Name: p-Tolualdehyde Synonyms: 4-methyl-benzaldehyd;benzaldehyde,-methyl-;para-methylbenzaldehyde;Paratolualdehyde;para-tolualdehyde;para-toluylaldehyde;p-Formyltoluene;FEMA 3068 CAS: 104-87-0 MF: C8H8O MW: 120.15 EINECS: 203-246-9 Pr
Cas:104-87-0
Min.Order:1 Kilogram
FOB Price: $300.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufactu
Cas:104-87-0
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:104-87-0
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
p-Tolualdehyde Synonyms 4-Methylbenzaldehyde CAS NO.: 104-87-0 EINECS 203-246-9
Hangzhou Sartort Biopharma Co., Ltd
Appearance:Clear colorless to yellow liquid Storage:Store below +30°C. Package:200kg/drum Application:p-Tolualdehyde is made by the Vilsmeier reaction employing toluene and dimethylformamide or the Guttermann–Koch reaction employing toluene and car
Cas:104-87-0
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:104-87-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXi'an Faithful Biotech Co., Ltd.
We are the manufacturers and suppliers of API in China, and warehouse in Germany and USA of California, which can quickly and safely deliver to your address 1.High quality and competitive price. 2.Free sample for your evaluation. 3.Promptly delivery
Cas:104-87-0
Min.Order:10 Gram
FOB Price: $3.5
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
p-Tolualdehyde CAS:104-87-0 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediat
Cas:104-87-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Changchun Artel lmport and Export trade company
Minimum Order Qty. 10 Gram Supply Ability 500 Kilograms/Month Storage store in cool, dry, ventilated place 20℃ Delivery Time 3 business days after payment Payment Term TT,western union,Paypal,MoneyGram Package 10g,20g,50g,100g,500g,1KGS,
Cas:104-87-0
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryHubei Jiutian Bio-medical Technology Co., Ltd
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highly qualifie
Cas:104-87-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei Mojin Biotechnology Co.,Ltd
Company information: Hebei Mojin Biotechnology Co., Ltd, Our company is a professional in chemical raw materials and chemical reagents research and development production enterprises. Our business covers more than 30 countries, most of the big cust
Cas:104-87-0
Min.Order:1 Kilogram
FOB Price: $7.0 / 10.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:104-87-0
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Afine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Chemlyte Solutions
Chemlyte Solutions believe that customers and suppliers deserve much more than what traditional distributors can offer. To grow in today s fast-paced and increasingly competitive market it is essential to be able to quickly adapt to market forces eff
Synthetic route

| Conditions | Yield |
|---|---|
| With dinitrogen tetraoxide; ferric nitrate In dichloromethane Ambient temperature; further oxidizing agent, further conditions and solvents; | 100% |
| In ethanol at 40℃; for 9h; | 100% |
| With TGSE; sodium hydrogencarbonate; sodium carbonate In water for 1.66667h; Electrochemical reaction; | 100% |

| Conditions | Yield |
|---|---|
| With oxygen; 10-methyl-9-phenylacridin-10-ium perchlorate In chloroform at 24.84℃; for 10h; Oxidation; Pyrolysis; visible light; | 100% |
| With sulfuric acid; 9-mesityl-2,7,10-trimethylacridinium perchlorate; water; oxygen In acetonitrile at 24.84℃; for 1.33333h; Quantum yield; Reagent/catalyst; Irradiation; | 100% |
| With N-hydroxyphthalimide; oxygen; cobalt(II) acetate; acetic acid at 20℃; under 760.051 Torr; chemoselective reaction; | 100% |
-
-
120445-48-9, 3030-95-3
4-methylbenzaldehyde semicarbazone

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride; Tonsil In ethyl acetate for 2h; Heating; | 100% |
| With copper(II) sulfate In tetrahydrofuran; methanol; water for 48h; Heating; | 95% |
| With potassium permanganate; montmorillonite K-10 for 0.25h; | 94% |

| Conditions | Yield |
|---|---|
| With [NO(1+)*18-crown-6*H(NO3)2(1-)]; silica gel In dichloromethane at 20℃; for 0.0833333h; | 100% |
| With sulphated zirconia In acetonitrile at 60℃; for 0.3h; Microwave irradiation; | 100% |
| With Montmorillonite K10 In dichloromethane for 0.333333h; Heating; | 99% |
-
-
40739-81-9
p-tolualdehyde p-toluenesulfonylhydrazone

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In tetrahydrofuran; methanol; water for 48h; Heating; | 100% |
| With Cr-MCM-41 zeolite on silica gel for 0.15h; microwave irradiation; | 98% |
| Amberlyst 15 In water; acetone at 80℃; for 24h; | 86% |
| With polymer-supported phenyliodine(III) diacetate; water In dichloromethane at 20℃; for 2h; Oxidation; | 78% |

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In tetrahydrofuran; methanol; water for 6.5h; Heating; | 100% |
| With benzyltriphenylphosphonium peroxodisulfate In acetonitrile for 0.133333h; Heating; | 95% |
| With CuCl*Kieselghur; oxygen In dichloromethane at 20℃; for 0.5h; | 94% |
-
-
160594-72-9
N,N-diethyl-4-methylselenobenzamide

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0℃; for 3h; | 100% |
-
-
89200-92-0
trimethyl(4-methylbenzyloxy)silane

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With nitrogen dioxide at 20℃; for 0.0833333h; | 100% |
| With phosphomolybdic acid In toluene for 0.166667h; Heating; | 97% |
| With allyltriphenylphopsphonium peroxodisulfate In acetonitrile for 0.25h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| With sodium periodate; C31H29Br2N3Ru*CH2Cl2 In water; ethyl acetate; acetonitrile at 25℃; for 0.5h; Inert atmosphere; Schlenk technique; | 100% |
| With tert.-butylhydroperoxide; manganese(II,III) oxide In acetonitrile at 70℃; for 3h; | 98% |
| With iron(III) trifluoromethanesulfonate; 2-((4R,5R)-1-((4-(tert-butyl)phenyl)sulfonyl)-4,5-diphenylimidazolidin-2-yl)-6-((4R,5R)-1-((4-(tert-butyl)phenyl)sulfonyl)-4,5-diphenylimidazolidin-2-yl)pyridine; oxygen In 1,2-dichloro-ethane at 70℃; under 760.051 Torr; for 6h; Reagent/catalyst; Solvent; Green chemistry; chemoselective reaction; | 96% |
-
-
1203650-44-5
N-(2-hydroxyl-1-(2-pyridyl)-2-(4-methylphenyl)ethyl)benzenecarbothioamide

-
A
-
1203650-52-5
bis(3-phenyl-1-imidazo[1,5-a]pyridyl)-4-methylphenylmethane

-
B
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With pyridine; iodine In tetrahydrofuran at 20℃; for 0.5h; | A 100% B n/a |

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With α,α-diphenyl-2-pyrrolidinemethanol trimethylsilyl ether In chloroform at 20℃; for 24h; regioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 24.85℃; under 56886.3 Torr; for 0.5h; Product distribution; Further Variations:; Pressures; Temperatures; | 99.1% |
| With 1,10-Phenanthroline; triphenylphosphine; 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate; scandium tris(trifluoromethanesulfonate); 1-n-butyl-3-methylimidazolium hexafluoroantimonate; cerium triflate at 50℃; under 15001.5 Torr; for 5h; Reagent/catalyst; Autoclave; Inert atmosphere; | 76.8% |
| With trifluorormethanesulfonic acid under 95000.1 Torr; for 4h; Ambient temperature; | 72 % Chromat. |

| Conditions | Yield |
|---|---|
| Stage #1: 4-methylbenzoic acid ethyl ester With morpholine; diisobutylaluminium hydride In tetrahydrofuran; hexane at 0℃; for 3.16667h; Inert atmosphere; Stage #2: With diisobutylaluminium hydride In tetrahydrofuran; hexane at 0℃; for 0.166667h; Inert atmosphere; | 99% |
| With sodium tris(diethylamino)aluminum hydride In tetrahydrofuran; dodecane at 0℃; for 24h; | 93% |
| With phenylsilane; cobalt(II) diacetate tetrahydrate; sodium triethylborohydride In 1,2-dimethoxyethane; toluene at 25℃; for 15h; Inert atmosphere; Schlenk technique; | 92% |
-
-
51490-06-3
1,2-di-p-tolyl-ethanone

-
-
62-53-3
aniline

-
A
-
6833-18-7
4-methyl-N-phenylbenzamide

-
B
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; copper(II) chloride dihydrate; oxygen In acetonitrile under 760.051 Torr; for 24h; Reflux; | A 99% B n/a |

-
-
106-42-3
para-xylene

-
A
-
100-21-0
terephthalic acid

-
B
-
104-87-0
4-methyl-benzaldehyde

-
C
-
619-66-9
4-Carboxybenzaldehyde

-
D
-
99-94-5
p-Toluic acid

| Conditions | Yield |
|---|---|
| With hydrogen bromide; oxygen; acetic acid; cobalt(II) acetate; manganese(II) acetate In water at 190℃; under 16501.7 Torr; for 1h; Product distribution / selectivity; | A 98.1% B 0.2% C 0.4% D 0.4% |
| With oxygen; acetic acid; palladium diacetate; antimony(III) acetate In water at 182 - 195℃; under 16501.7 - 20929.4 Torr; for 1 - 1.5h; Product distribution / selectivity; | A 50.3% B 7.2% C 6.4% D 6.2% |
| With hydrogen bromide; oxygen; acetic acid; zirconium oxyacetate; cobalt(II) acetate In water at 190℃; under 16501.7 Torr; for 1h; Product distribution / selectivity; | A 4.9% B 3% C 1.9% D 36.9% |


| Conditions | Yield |
|---|---|
| With water; sodium hydroxide at 20℃; for 0.05h; Microwave irradiation; | 98% |
| With 4-methylmorpholine N-oxide; 1-ethyl-3-methyl-1H-imidazol-3-ium chloride; potassium iodide at 100℃; for 0.0333333h; Microwave irradiation; Ionic liquid; | 94% |
| With 1-dodecyl-3-methylimidazolium iron chloride; periodic acid at 30℃; for 1.5h; | 93% |
-
-
3235-02-7, 3717-15-5, 3717-16-6, 140461-26-3, 140461-27-4
p-methylbenzaldehyde oxime

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With CuCl*Kieselghur; oxygen In dichloromethane at 20℃; for 0.333333h; | 98% |
| Stage #1: p-methylbenzaldehyde oxime With hexachlorodisilane; silica gel In toluene at 110℃; for 0.5h; Stage #2: With water In toluene for 0.5h; | 97% |
| In dichloromethane at 20℃; for 4h; | 96% |
-
-
18484-04-3
2-((4-methylbenzyl)oxy)tetrahydro-2H-pyran

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With zeofen (zeolite HZSM-5, Fe(NO3)3*9H2O) In dichloromethane for 6h; Heating; | 98% |
| With chromium(VI) oxide; HZSM-5 zeolite for 0.0333333h; microwave irradiation; | 95% |
| With NTPPPODS In acetonitrile for 0.166667h; Reflux; | 94% |
-
-
79439-44-4
C16H18OSe

-
A
-
13527-05-4
4-methylbenzyl nitrate

-
B
-
66361-14-6
p-methylbenzeneseleninic acid

-
C
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With nitric acid In dichloromethane at -5 - 0℃; for 1h; | A 98% B n/a C n/a D n/a E n/a |
| With nitric acid In dichloromethane at -5 - 0℃; for 1h; | A n/a B 98% C n/a D n/a E n/a |


| Conditions | Yield |
|---|---|
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; N-methyl-N-[3-(4-diacetoxyiodo)phenoxy-1-propyl]pyrrolidinium 4-methylbenzenesulfonate In dichloromethane at 20℃; for 1.5h; Inert atmosphere; | 98% |

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 0.0416667h; Product distribution; Ambient temperature; pH = 4-6, regeneration of aldehyde; | 97.1% |

| Conditions | Yield |
|---|---|
| With lithium diisobutylmorpholinoaluminum hydride In tetrahydrofuran; hexane at 0℃; for 0.5h; | 97% |
| With calcium bis(hypophosphite); calcium acetate; nickel(II) acetate tetrahydrate In ethanol; water at 100℃; for 7h; Sealed tube; | 94% |
| With C13H26B(1-)*K(1+) In tetrahydrofuran for 24h; Ambient temperature; | 83% |

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide at 20℃; for 0.0666667h; Microwave irradiation; | 97% |
| With dihydrogen peroxide In ethanol for 4h; Reflux; Green chemistry; | 95% |
| With 1-dodecyl-3-methylimidazolium iron chloride; periodic acid at 30℃; for 1.5h; | 93% |
-
-
71778-40-0
1-((phenylthio)(p-tolyl)methylthio)benzene

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With sodium perborate In acetic acid at 25℃; for 2h; | 97% |
| With oxygen; 2,4,6-tris(p-chlorophenyl)pyrylium perchlorate In dichloromethane for 0.2h; Irradiation; | 83% |

| Conditions | Yield |
|---|---|
| With potassium permanganate; Amberlite IR-120; water In dichloromethane at 20℃; for 0.333333h; | 97% |
| With sulfuric acid; quinolinium dichromate(VI) In acetic acid at 29.9℃; Rate constant; Thermodynamic data; ΔHact, ΔSact, ΔGact; var. concentration of reagents; | |
| Multi-step reaction with 2 steps 1: magnesium chloride; ATP / 20 h / pH 7.5 / Enzymatic reaction 2: water / 14 h / 30 °C / Enzymatic reaction View Scheme |
-
-
122334-36-5
N-methoxy-N,4-dimethylbenzamide

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With lithium diisobutyl-tert-butoxyaluminum hydride In tetrahydrofuran; hexane at 0℃; Reagent/catalyst; Inert atmosphere; chemoselective reaction; | 97% |
| With benzoic acid ethyl ester; copper diisobutyl-t-butoxyaluminum hydride In tetrahydrofuran at 20℃; for 12h; Inert atmosphere; chemoselective reaction; | 96 %Chromat. |

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; tetrakis(pyridine)silver(II) peroxodisulfate; oxygen In N,N-dimethyl-formamide at 23℃; under 760.051 Torr; Irradiation; | 97% |

| Conditions | Yield |
|---|---|
| With polyaniline-sulfate salt; water for 0.5h; Heating; | 96% |
| With silica-OSO3H; silica gel In toluene at 60 - 70℃; for 1h; | 94% |
| In(OSO2CF3)3 In acetone at 20℃; for 4h; | 92% |
-
-
23229-29-0
2-(4-methylphenyl)-1,3-dithiolane

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With tetrachlorosilane; dimethyl sulfoxide In dichloromethane at 20℃; for 0.583333h; | 96% |
| With silica gel In neat (no solvent) at 20℃; for 0.05h; | 96% |
| With sodium nitrate; sulfuric acid; silica gel; 4-nitrobenzylidene diacetate In dichloromethane at 20℃; for 0.25h; | 93% |

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In acetonitrile at 40℃; for 0.5h; Irradiation; | A 96% B n/a |
| With ammonium cerium(IV) nitrate In acetonitrile at 40℃; for 0.5h; Mechanism; Irradiation; | A 96% B n/a |
| ammonium cerium(IV) nitrate; sodium dodecyl-sulfate In water at 25℃; for 3h; | A 3.95 mmol B 3.10 mmol |
| With ammonium cerium(IV) nitrate; sodium dodecyl-sulfate In water at 25℃; for 3h; Title compound not separated from byproducts; | A 3.95 mmol B 3.10 mmol |
| With sodium dodecyl-sulfate; ammonium cerium(IV) nitrate In water at 25℃; for 3h; Product distribution; other reagents, solvents, reac. time; | A 3.95 mmol B 3.1 mmol |
-
-
79-24-3
Nitroethane

-
-
104-87-0
4-methyl-benzaldehyde

-
-
52287-56-6, 29816-55-5
1-(p-tolyl)-2-nitropropene

| Conditions | Yield |
|---|---|
| With ammonium acetate at 110℃; for 16h; | 100% |
| With (2-hydroxyethyl)ammonium formate at 20℃; for 4.1h; Knoevenagel condensation; Ionic liquid; | 95% |
| With ammonium acetate at 120℃; for 2h; | 95% |

| Conditions | Yield |
|---|---|
| With C62H54N2O2; copper diacetate In ethanol at 10℃; for 48h; Henry Nitro Aldol Condensation; enantioselective reaction; | 100% |
| With C62H48CuN2O2; copper diacetate; benzaldehyde In ethanol at 10℃; for 48h; Reagent/catalyst; Henry Nitro Aldol Condensation; | 100% |
| With (2-hydroxyethyl)ammonium formate at 20℃; for 0.8h; Knoevenagel condensation; Ionic liquid; | 95% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
540-63-6
ethane-1,2-dithiol

-
-
23229-29-0
2-(4-methylphenyl)-1,3-dithiolane

| Conditions | Yield |
|---|---|
| With silica gel; toluene-4-sulfonic acid In dichloromethane for 1.5h; Heating; | 100% |
| With Cu(OTf)2-SiO2 for 0.5h; Ambient temperature; | 99% |
| With PPA; silica gel In 1,2-dichloro-ethane at 20℃; for 0.5h; | 99% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
100-63-0
phenylhydrazine

-
-
2829-25-6
4-methylbenzaldehyde phenylhydrazone

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane | 100% |
| In neat (no solvent) at 20℃; for 0.00277778h; | 97% |
| With oxidized single-walled carbon nanotubes(SWCNs-COOH) In ethanol at 80℃; for 1.25h; | 96% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
74-89-5
methylamine

-
-
17972-13-3, 29086-13-3, 53699-34-6
p-methylbenzylidene-methylamine

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |
| In methanol; dichloromethane for 18h; Molecular sieve; | 86% |

| Conditions | Yield |
|---|---|
| In chloroform at 20℃; for 1h; | 100% |
| for 6h; Kinetics; Molecular sieve; Reflux; | 100% |
| With magnesium sulfate In dichloromethane at 20℃; Inert atmosphere; | 65% |

| Conditions | Yield |
|---|---|
| With diisopropoxytitanium(III) tetrahydroborate In dichloromethane at -20℃; for 0.133333h; | 100% |
| With sodium tetrahydroborate In methanol at 20℃; for 1h; | 100% |
| With hydrogen at 30℃; under 22502.3 Torr; for 10h; Temperature; Autoclave; Sealed tube; Ionic liquid; chemoselective reaction; | 100% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
5173-28-4, 5173-29-5, 24133-59-3, 66749-58-4, 66768-19-2
1,2-di-p-tolylethane-1,2-diol

| Conditions | Yield |
|---|---|
| With aluminium; potassium hydroxide In methanol at 0℃; for 1h; Inert atmosphere; | 100% |
| With (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; acetic acid In dichloromethane at 25℃; for 16h; Michael Addition; Inert atmosphere; Irradiation; | 99% |
| With naphthalene; lithium; manganese(ll) chloride In tetrahydrofuran at 22 - 26℃; for 1h; sonication; | 98% |

| Conditions | Yield |
|---|---|
| With phosphate-buffered silica gel supported KMnO4 In cyclohexane at 65℃; | 100% |
| With periodic acid; tripropylammonium fluorochromate (VI) In acetonitrile at 0℃; for 1.5h; | 100% |
| With [Cu2C6H4(CHNCH2CH2N(CH2C5H4N)2)2](2+)*2ClO4(1-)=C36H38Cu2N8(ClO4)2; oxygen In acetone at -90.16℃; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; antimony In water for 16h; | 100% |
| With titanium(III) chloride; tin(ll) chloride In water at 20℃; for 8h; Barbier reaction; | 100% |
| With tin(ll) chloride In water at 20℃; for 24h; | 99% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2017-88-1
ethyl (E)-2-cyano-3-p-tolylacrylate

| Conditions | Yield |
|---|---|
| With L-proline for 0.0833333h; Knoevenagel condensation; microwave irradiation; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane In neat liquid at 20℃; for 0.0166667h; Knoevenagel Condensation; Green chemistry; | 100% |
| With ASCPEI In ethanol at 43℃; for 3h; Knoevenagel condensation; | 99% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
109-77-3
malononitrile

-
-
2826-25-7
2-(4-methylbenzylidene)malononitrile

| Conditions | Yield |
|---|---|
| With L-arginine at 20℃; for 0.0833333h; Knoevenagel condensation; Ionic liquid; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane In water at 20℃; for 0.0333333h; Knoevenagel Condensation; Green chemistry; | 100% |
| With 2C9H3O6(3-)*2C27H18N6*3Ni(2+)*15H2O*5C2H6O In dichloromethane at 60℃; for 2h; Knoevenagel Condensation; Inert atmosphere; | 100% |
-
-
3528-17-4
2,3-dihydro-4H-[1]benzothiopyran-4-one

-
-
104-87-0
4-methyl-benzaldehyde

-
-
69964-53-0, 135521-92-5, 101001-08-5
(Z)-3-(4-methylbenzylidene)thiochroman-4-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 3h; | 100% |
| With sulfuric acid In acetic acid at 20℃; for 0.333333h; | 89% |
| With hydrogenchloride In ethanol for 0.0833333h; Ambient temperature; | 64.1% |
| With piperidine In chloroform for 5h; Reflux; | |
| With sodium hydroxide In ethanol; water at 0℃; for 0.5h; |
-
-
27353-29-3
diethyl (morpholinomethyl)phosphonate

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at 0℃; for 1.25h; | 100% |
-
-
109-80-8
1.3-propanedithiol

-
-
104-87-0
4-methyl-benzaldehyde

-
-
56637-44-6
2-(4-methylphenyl)-1,3-dithiane

| Conditions | Yield |
|---|---|
| With lithium tetrafluoroborate at 25℃; for 1h; | 100% |
| With amberlyst-15 In acetonitrile for 1h; | 99.97% |
| With nickel dichloride In dichloromethane | 99% |
-
-
563-80-4
3-methyl-butan-2-one

-
-
104-87-0
4-methyl-benzaldehyde

-
-
67962-11-2
4-methyl-1-p-tolyl-pent-1-en-3-one

| Conditions | Yield |
|---|---|
| barium dihydroxide In ethanol for 1h; Heating; | 100% |
| With sodium hydroxide In water Heating; |
-
-
35161-71-8
N-methylpropargylamine

-
-
104-87-0
4-methyl-benzaldehyde

-
-
709-86-4
N-methyl-N-(4-methylbenzyl)prop-2-yn-1-amine

| Conditions | Yield |
|---|---|
| Stage #1: 4-methyl-benzaldehyde With acetic acid In dichloromethane at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: methyl(propargyl)amine In dichloromethane at 20℃; for 1h; Inert atmosphere; Stage #3: With sodium tris(acetoxy)borohydride In dichloromethane at 0℃; Inert atmosphere; | 100% |
| With hydrogenchloride; sodium cyanoborohydride 1.) methanol, 15 min, 2.) MeOH, 24 h; Multistep reaction; |
-
-
107-10-8
propylamine

-
-
104-87-0
4-methyl-benzaldehyde

-
-
99484-07-8
N-((4-methylphenyl)methylene)-1-propanamine

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |
| With 1-hydrosilatrane In neat (no solvent) at 70℃; for 23h; Sealed tube; Green chemistry; | 99% |
| In methanol at 25℃; Mechanism; Rate constant; Thermodynamic data; ΔH(excit.), ΔS(excit.); |
-
-
558-13-4
carbon tetrabromide

-
-
104-87-0
4-methyl-benzaldehyde

-
-
60512-56-3
1-(2,2-dibromovinyl)-4-methylbenzene

| Conditions | Yield |
|---|---|
| Stage #1: carbon tetrabromide With triphenylphosphine In dichloromethane at 0℃; for 0.5h; Inert atmosphere; Stage #2: 4-methyl-benzaldehyde In dichloromethane at 0℃; for 2h; Inert atmosphere; | 100% |
| Stage #1: carbon tetrabromide With triphenylphosphine In dichloromethane at 0℃; for 0.5h; Inert atmosphere; Stage #2: 4-methyl-benzaldehyde In dichloromethane at 0℃; for 2h; Inert atmosphere; | 100% |
| With triphenylphosphine In dichloromethane at 0℃; for 0.5h; | 98% |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
104-87-0
4-methyl-benzaldehyde

-
-
66985-49-7
2-(4-methylphenyl)-2-(trimethylsilyloxy)acetonitrile

| Conditions | Yield |
|---|---|
| With Na(1+)*C7H3NO4(2-)*Cu(2+)*C7H4NO4(1-) at 20℃; for 3h; | 100% |
| Stage #1: 4-methyl-benzaldehyde With scandium tris(trifluoromethanesulfonate); 1-n-butyl-3-methylimidazolium hexafluoroantimonate at 20℃; for 0.166667h; Stage #2: trimethylsilyl cyanide at 20℃; for 0.0833333h; Inert atmosphere; | 99% |
| With C7H3O8S2(3-)*5H2O*Pr(3+) In neat (no solvent) at 40℃; for 0.5h; Inert atmosphere; | 99% |
-
-
2942-58-7
diethyl cyanophosphonate

-
-
104-87-0
4-methyl-benzaldehyde

-
-
123346-48-5
Phosphoric acid cyano-p-tolyl-methyl ester diethyl ester

| Conditions | Yield |
|---|---|
| With lithium cyanide In tetrahydrofuran Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol at 0℃; | 100% |
-
-
557-20-0
diethylzinc

-
-
104-87-0
4-methyl-benzaldehyde

-
-
25574-04-3, 112777-65-8, 138809-38-8, 73854-03-2
(S)-1-(4-methylphenyl)propan-1-ol

| Conditions | Yield |
|---|---|
| (1S)-1-(9-piperidylfluoren-9-yl)ethanol In hexane; toluene at 0℃; for 4h; Addition; | 100% |
| Stage #1: diethylzinc In toluene at 0℃; for 0.5h; Stage #2: 4-methyl-benzaldehyde In toluene at 0 - 20℃; for 48h; Reagent/catalyst; enantioselective reaction; | 99% |
| Stage #1: diethylzinc; 4-methyl-benzaldehyde With diphenyl-((S)-1-((S)-1-phenylethyl)aziridin-2-yl)-methanol In hexane; toluene at 0 - 25℃; for 48.5h; Inert atmosphere; Stage #2: With ammonium chloride In hexane; water; toluene Saturated solution; optical yield given as %ee; enantioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| In diethyl ether under 7500600 Torr; for 168h; Product distribution; Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| barium dihydroxide In ethanol for 1h; Heating; | 100% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View




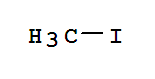











 Xn
Xn