Shanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
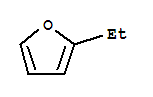
Cas:1487-18-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
2-ethenylfuran CAS:1487-18-9 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermed
Cas:1487-18-9
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:1487-18-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:1487-18-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHebei Mojin Biotechnology Co.,Ltd
1, High quality with competitive price:2, Fast and safe delivery3.Excellent pre-sales and after-sales service4. Well-trained and professional technologist and sales with rich experience in the field for 5-10 yearsAppearance:see detailed specification
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM is one of China's leading providers of integrated fine chemical services including offering, research and development, Custom manufacturing business, as well as other Value-added customer services, for diversified range products of chemicals
Xi`an Eastling Biotech Co., Ltd.
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:Beijing or Guangzhou
Henan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Henan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Shanghai Chinqesen Biotechnology Co., Ltd.
Good Quality Package:1kg/bag Application:Medical or chemical Transportation:Air/Train/Sea Port:Shenzhen
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Chungking Joyinchem Co., Ltd.
Joyinchem have been committed to chemical supply for several years and have built good cooperation records with multinational chemical corporations and importers from all over the world. Our services include:-Spot goods-Contract manufacturing-Custom
Shanghai Run-Biotech Co., Ltd.
Shanghai, Run-Biotech Co., Ltd is a leading domestic pharmaceutical, biopharmaceutical, and health care products R & D outsourcing services company. As an innovation-driven and customer-focused company, Run Biotech provides a broad and integrated por
Nanjing Raymon Biotech Co., Ltd.
Furan, 2-ethenyl-Appearance:Off white to slight yellow solid Storage:keep in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/pharma intermediate Transportation:By Sea,by Air,By courier like DHL or Fe
Hebei Ruishun Trade Co.,Ltd
Supply top quality products with a reasonable price Application:api
Chemical Co.Ltd
Furan, 2-ethenyl-Appearance:Off white to slight yellow solid Storage:Stored in shaded, cool and dry places Package:1L 5L 10L 25L bottle Application:pharma intermediate Transportation:Handle with cares to avoid damaging the packages. Protect them from
Synthetic route

| Conditions | Yield |
|---|---|
| With thiourea In water at 90℃; | 98% |

| Conditions | Yield |
|---|---|
| With copper(I) oxide In various solvent(s) at 200℃; for 3h; | 73% |
| at 270 - 320℃; | |
| With quinoline; copper(II) sulfate | |
| With Camellia sinensis at 25℃; for 120h; pH=6.4; Decarboxylation; | |
| With propane at 350℃; |

| Conditions | Yield |
|---|---|
| With barium dihydroxide; water In 1,4-dioxane at 70℃; for 0.25h; Product distribution; other aldehydes; var. times and reagents; | 55% |
-
-
113348-42-8
1,2-epithio-(2'-furyl)-ethane

-
-
1487-18-9
(furan-2-yl)ethylene

| Conditions | Yield |
|---|---|
| With triphenylphosphine | 54% |

| Conditions | Yield |
|---|---|
| With scandium tris(trifluoromethanesulfonate) In acetonitrile Wittig Olefination; Inert atmosphere; Glovebox; Electrochemical reaction; | 46% |

| Conditions | Yield |
|---|---|
| With SAPO-34 at 320 - 520℃; for 3h; Reagent/catalyst; Temperature; Inert atmosphere; Molecular sieve; | 44% |

| Conditions | Yield |
|---|---|
| With bromine In N,N-dimethyl-formamide at 20℃; for 1h; Inert atmosphere; | A 9% B 29% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl-triphenylphosphonium iodide With n-butyllithium In tetrahydrofuran at -78 - 0℃; for 0.25h; Inert atmosphere; Stage #2: furfural In tetrahydrofuran at 23℃; Inert atmosphere; | 21% |
| Stage #1: methyl-triphenylphosphonium iodide With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 0.25h; Wittig Olefination; Inert atmosphere; Stage #2: furfural In tetrahydrofuran; hexane at 0 - 20℃; for 2.25h; Wittig Olefination; |

| Conditions | Yield |
|---|---|
| With quinoline at 200℃; Erhitzen des Reaktionsgemisches mit Kupfer(II)-sulfat auf 150gradC; |

| Conditions | Yield |
|---|---|
| With potassium acetate-silica gel at 350℃; |
-
-
13129-26-5
1-(3'-furyl)-ethanol

-
-
1487-18-9
(furan-2-yl)ethylene

| Conditions | Yield |
|---|---|
| With aluminum oxide at 350℃; |

| Conditions | Yield |
|---|---|
| With copper at 250℃; | |
| With potassium carbonate at 300℃; |

| Conditions | Yield |
|---|---|
| at 350℃; beim Leiten durch ein Rohr; |

| Conditions | Yield |
|---|---|
| With I Heating; | |
| With iodine distillation; | |
| With iodine distillation; ethylfurylcarbinol; |

-
-
1487-18-9
(furan-2-yl)ethylene

| Conditions | Yield |
|---|---|

| Conditions | Yield |
|---|---|


| Conditions | Yield |
|---|---|
| With acetic anhydride |


| Conditions | Yield |
|---|---|
| at 380℃; |


| Conditions | Yield |
|---|---|
| Grignard reaction; | |
| Stage #1: furfural; (trimethylsilyl)methylmagnesium chloride In tetrahydrofuran at 0 - 20℃; Peterson Olefination; Inert atmosphere; Stage #2: With bis(trifluoromethanesulfonyl)amide In chloroform-d1 at 20℃; for 0.25h; Peterson Olefination; | 75 %Spectr. |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether for 1h; Elimination; | |
| With hydrogenchloride In diethyl ether for 1.25h; | |
| With hydrogenchloride In diethyl ether for 1h; |

| Conditions | Yield |
|---|---|
| copper(I) oxide In quinoline Heating; | |
| With copper(II) sulfate Inert atmosphere; | |
| With ferulic acid decarboxylase from Aspergillus niger In aq. phosphate buffer; dimethyl sulfoxide at 30℃; for 18h; pH=7.5; Catalytic behavior; Reagent/catalyst; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: NaI; triethylamine / acetonitrile 2: CH2Cl2 / 1 h / 20 °C 3: 56 percent / NaBH4 / diethyl ether; methanol / 1 h / 10 - 20 °C 4: 54 percent / triphenylphosphine View Scheme | |
| With C18H21BrMnN3O3; potassium tert-butylate; isopropyl alcohol at 40℃; for 24h; | 92 %Chromat. |
-
-
62889-08-1
1-(2-furyl)-1-(trimethylsilyloxy)ethylene

-
-
1487-18-9
(furan-2-yl)ethylene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: CH2Cl2 / 1 h / 20 °C 2: 56 percent / NaBH4 / diethyl ether; methanol / 1 h / 10 - 20 °C 3: 54 percent / triphenylphosphine View Scheme |
-
-
1026377-65-0
2-<(5,5-Dimethyl-2-oxido-1,3,2-dioxaphosphinan-2-yl)sulfanyl>-1-furan-2-ylethanone

-
-
1487-18-9
(furan-2-yl)ethylene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 56 percent / NaBH4 / diethyl ether; methanol / 1 h / 10 - 20 °C 2: 54 percent / triphenylphosphine View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: Mg; dibromoethane / diethyl ether / 18 h / Heating 1.2: diethyl ether / 18 h / 0 - 23 °C 2.1: aq. HCl / diethyl ether / 1.25 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: Mg / diethyl ether / 1 h / Heating 1.2: 90 percent / diethyl ether / 12 h 2.1: aq. HCl / diethyl ether / 1 h View Scheme | |
| Multi-step reaction with 2 steps 1: 90 percent / magnesium / diethyl ether / 12 h / 0 - 20 °C 2: 1M HCl / diethyl ether / 1 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: acetic acid anhydride / 150 °C 2: 270 - 320 °C View Scheme | |
| Multi-step reaction with 2 steps 1: in Gegenwart basischer Katalysatoren 2: potassium carbonate / 300 °C View Scheme | |
| Multi-step reaction with 2 steps 1: acetic acid anhydride 2: 270 - 320 °C View Scheme | |
| Multi-step reaction with 2 steps 1: diethyl ether / 10 h / 0 °C 2: hydrogenchloride / water; diethyl ether / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With n-butyllithium In toluene |
-
-
1487-18-9
(furan-2-yl)ethylene

-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
103-71-9
phenyl isocyanate

-
-
2622-14-2
tricyclohexylphosphine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran exclusion of air; addn. of org. compds. to soln. opf Ni-compd. in THF (-78°C), warming with stirring (-10°C, 2 d); partial evapn., addn. of pentane, filtration (-10°C), washing (cold pentane), drying (vac.); elem. anal.; | 99.9% |


| Conditions | Yield |
|---|---|
| With 2-benzyloxymethylphenyldiphenylphosphine; bi(allylnickel bromide); sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate In dichloromethane at 0 - 23℃; under 760.051 Torr; for 2h; Schlenk technique; | 99% |

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; oxygen; palladium diacetate; [(NH4)5H6PMo4V7.8O40]*xH2O; sodium chloride In methanol; water at 20℃; for 3.5h; | 98.2% |
| With tert.-butylhydroperoxide; C21H19N5Pd(2+)*2BF4(1-) In decane; acetonitrile at 45℃; for 12h; Wacker Oxidation; | 90% |
| With dihydrogen peroxide In water; acetonitrile at 55℃; for 12h; Wacker Oxidation; | 80% |
-
-
1487-18-9
(furan-2-yl)ethylene

-
-
19377-75-4
1-(furan-2-yl)ethane-1,2-diol

| Conditions | Yield |
|---|---|
| With osmium(VIII) oxide; N-methyl-2-indolinone In tetrahydrofuran at 20℃; for 48h; Oxidation; Dihydroxylation; | 91% |
| With AD-mix-β; water In diethyl ether; tert-butyl alcohol at 0℃; Sharpless dihydroxylation; |
-
-
1487-18-9
(furan-2-yl)ethylene

-
-
93017-30-2
bis(2-phenethyl)phosphine

-
-
1351415-42-3
1-(2-furyl)ethyl bis(2-phenylethyl)diselenophosphinate

| Conditions | Yield |
|---|---|
| With selenium In 1,4-dioxane at 85℃; for 3h; Inert atmosphere; regioselective reaction; | 91% |
-
-
1487-18-9
(furan-2-yl)ethylene

-
-
1351415-45-6
1-(2-furyl)ethyl bis[2-(2-furyl)ethyl]diselenophosphinate

| Conditions | Yield |
|---|---|
| With selenium In 1,4-dioxane at 85℃; for 3h; Inert atmosphere; regioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With iodine; dimethyl sulfoxide at 110℃; for 12h; | 87% |
-
-
110-89-4
piperidine

-
-
1487-18-9
(furan-2-yl)ethylene

-
-
201230-82-2
carbon monoxide

-
-
129762-31-8
N-<2-(2-Furyl)propyl>piperidine

| Conditions | Yield |
|---|---|
| With hydrogen; carbonylhydridetris(triphenylphosphine)rhodium(I) at 100℃; under 76000.1 Torr; for 4h; | 85% |

-
-
1487-18-9
(furan-2-yl)ethylene

-
-
617-89-0
furan-2-ylmethanamine

-
-
201230-82-2
carbon monoxide

-
-
129762-35-2
N-<2-(2-Furyl)propyl>-N-urfurylimine

| Conditions | Yield |
|---|---|
| With hydrogen; carbonylhydridetris(triphenylphosphine)rhodium(I) at 100℃; under 76000.1 Torr; for 4h; | 85% |

-
-
1487-18-9
(furan-2-yl)ethylene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether for 3h; Cooling with ice; Inert atmosphere; | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: (furan-2-yl)ethylene With tert.-butylhydroperoxide; water; iodine at 120℃; for 1h; Sealed tube; Stage #2: 1,2-diamino-benzene With dimethyl sulfoxide at 120℃; for 2h; Sealed tube; | 84% |

| Conditions | Yield |
|---|---|
| With pyridine; lanthanum tris(N,N-dimethylbenzylamine) at 80℃; for 16h; Glovebox; regioselective reaction; | 84% |

| Conditions | Yield |
|---|---|
| With sodium 2,2,2-trifluoroethanolate In 2,2,2-trifluoroethanol at 20℃; for 2h; Product distribution; Further Variations:; Solvents; Reagents; Cycloaddition; | 83% |

| Conditions | Yield |
|---|---|
| With manganese(II) acetate; copper dichloride In 1-methyl-pyrrolidin-2-one at 60℃; Inert atmosphere; | 82% |

-
-
1487-18-9
(furan-2-yl)ethylene

-
-
28321-79-1
diiminosuccinonitrile

-
-
37494-43-2
5-(2-furyl)-1,4,5,6-tetrahydropyrazine-2,3-dicarbonitrile

| Conditions | Yield |
|---|---|
| In acetonitrile for 20h; Ambient temperature; | 81% |
| In acetonitrile |

| Conditions | Yield |
|---|---|
| In benzene boiling; elem. anal.; | 80% |

| Conditions | Yield |
|---|---|
| With rhodium supported catalyst In dichloromethane at 25℃; under 7500.75 Torr; for 0.25h; | 79% |

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite; silver nitrate In water at 50℃; | 77% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; iodine In dimethyl sulfoxide at 80℃; | 76% |
-
-
1487-18-9
(furan-2-yl)ethylene

-
-
6773-29-1
dimethyl diazomalonate

-
A
-
120508-12-5
2-((Z)-4-Oxo-hexa-2,5-dienylidene)-malonic acid dimethyl ester

-
B
-
120508-20-5
dimethyl 2-(furan-2-yl)cyclopropane-1,1,-dicarboxylate

| Conditions | Yield |
|---|---|
| With rhodium(II) acetate at 20℃; for 96h; | A 11% B 75% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; potassium iodide In dimethyl sulfoxide at 130℃; for 4h; Sealed tube; | 75% |

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite In dimethyl sulfoxide at 80℃; for 24h; Inert atmosphere; Sealed tube; regioselective reaction; | 73% |
-
-
1487-18-9
(furan-2-yl)ethylene

-
-
1107629-44-6
[{κ2-(P,O)-(2-anisyl)2PC6H4SO2O}Pd(Me)dmso]

| Conditions | Yield |
|---|---|
| In dichloromethane at 25℃; for 30h; Schlenk technique; | 70% |
-
-
1487-18-9
(furan-2-yl)ethylene

-
-
28144-70-9
anthranilic acid amide

-
-
26059-84-7
2-(furan-2-yl)quinazolin-4(3H)-one

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; tetra-(n-butyl)ammonium iodide at 80℃; for 8h; Green chemistry; | 70% |

| Conditions | Yield |
|---|---|
| With phenylsilane; Co(C15H19N2O5)*MeOH In ethanol; dichloromethane at 23℃; for 12h; | 68% |

| Conditions | Yield |
|---|---|
| With ammonium carbonate; tetra-(n-butyl)ammonium iodide In water at 20℃; for 3h; Electrochemical reaction; stereoselective reaction; | 68% |
-
-
1487-18-9
(furan-2-yl)ethylene

-
-
201230-82-2
carbon monoxide

-
-
108-91-8
cyclohexylamine

-
-
129762-34-1
N-<2-(2-Furyl)propyl>-N-cyclohexylimine

| Conditions | Yield |
|---|---|
| With hydrogen; carbonylhydridetris(triphenylphosphine)rhodium(I) at 100℃; under 76000.1 Torr; for 4h; | 65% |

-
-
1487-18-9
(furan-2-yl)ethylene

| Conditions | Yield |
|---|---|
| With potassium hydroxide semihydrate; phosphorus In water; dimethyl sulfoxide at 75℃; for 1.5h; Inert atmosphere; | 65% |
| With phosphorus; ammonia; tert-butyl alcohol In diethyl ether for 1h; | 20% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View