Hangzhou Fandachem Co.,Ltd
Hangzhou Fanda Chemical Co.,Ltd (FandaChem) , a China-based chemical company, specialize in exporting Trifluoromethyl iodide 85% 99.5% Iodotrifluoromethane Trifluoroiodomethane cas: 2314-97-8, Please contact us by email freely. We are leadi
Cas:2314-97-8
Min.Order:1 Metric Ton
Negotiable
Type:Other
inquiryDayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:2314-97-8
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Appearance:White or off-white Solid Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:2314-97-8
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Cas:2314-97-8
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:2314-97-8
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available ♦ W
Cas:2314-97-8
Min.Order:1 Kilogram
FOB Price: $290.0 / 590.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Cas:2314-97-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufactu
Cas:2314-97-8
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:2314-97-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present,
Cas:2314-97-8
Min.Order:1 Kilogram
FOB Price: $500.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Henan Kanbei Chemical Co.,LTD
1. Well-equipped, stable and high-quality raw material supply chain, to provide customers with the best quality products. 2.Strict quality inspection process and professional certificate testing to ensure the quality level of the final product. 3.E
Cas:2314-97-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquirySHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:2314-97-8
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
TaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Cas:2314-97-8
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Tianfu Chemical, built in 2009, is a professional supplier for API materials in China. With 10 years development, we have bulit our own factory and lab to produce a certain of products. And we have established stable business relationships with ma
Cas:2314-97-8
Min.Order:1 Kilogram
FOB Price: $200.0
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Trifluoromethyl Iodide CAS No.:2314-97-8 Molecule Formula:CF3I Molecule Weight:195.91 Purity: 99.0% Package: 25kg/drum Description:White or off-white powder Manufacture Standards:Enterprise Standard TESTING ITEMS
Cas:2314-97-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:2314-97-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
Trifluoromethyl iodide Basic information Product Name: Trifluoromethyl iodide Synonyms: PERFLUOROMETHYL IODIDE;CF3I;CIF3;FIC-1311;Freon13I1;freon13t1;iodotrifluoromethane(trifluoromethyliodide);Methane,trifluoroiodo- CAS: 2314-97-8 MF: C
Cas:2314-97-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:2314-97-8
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:2314-97-8
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Suzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Cas:2314-97-8
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryWin-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:2314-97-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:2314-97-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Kunshan Push Trading Co.,LTD
ADVANTAGE 1.Professional: More than 10 years chemical exporting experience. We have produced chemical more than fifteen years, 95% products are for export . More than 10 years chemical exporting experience. Good and stabilized factory price. 2.
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:2314-97-8
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
112425-78-2
S-(trifluoromethyl)diphenylsulfonium tetrafluoroborate

-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With sodium iodide In acetonitrile for 0.25h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With iodine byproducts: phophorus iodide; 280°C (48 h); | 95% |

| Conditions | Yield |
|---|---|
| With sodium iodide In acetonitrile at 70℃; for 2h; Inert atmosphere; | 86% |
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 15% |
| With iodine monofluoride; palladium on activated carbon; copper on carbon; platinum on activated charcoal at 250 - 550℃; Product distribution / selectivity; Vapor phase; |
-
-
432-02-0
tris(trifluoromethyl)arsine

-
-
7553-56-2
iodine

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
353-91-3
trifluoromethyl-arsonous acid diiodide

-
C
-
359-55-7
iodo-bis-trifluoromethyl-arsine

-
D
-
7784-45-4
arsenic triiodide

| Conditions | Yield |
|---|---|
| 100°C, 48 h; at high temp. more AsI3 and CF3I; | A 80% B 5% C 8% D n/a |
| 100°C, 48 h; at high temp. more AsI3 and CF3I; | A 80% B 5% C 8% D n/a |

-
-
1514-87-0
metyhyl chlorodifluoroacetate

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
124-38-9
carbon dioxide

-
C
-
1184-76-5
difluorodiiodomethane

-
D
-
74-88-4
methyl iodide

| Conditions | Yield |
|---|---|
| With potassium fluoride; copper(l) iodide; iodine In N,N-dimethyl-formamide at 120℃; for 3h; Mechanism; other bromodifluoroacetate deriv.; var. temp. and times of reaction; | A 77% B n/a C 10% D n/a |

-
-
1514-87-0
metyhyl chlorodifluoroacetate

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
1184-76-5
difluorodiiodomethane

-
C
-
74-88-4
methyl iodide

| Conditions | Yield |
|---|---|
| With potassium fluoride; copper(l) iodide; iodine In N,N-dimethyl-formamide at 120℃; for 3h; | A 77% B 10% C n/a |


| Conditions | Yield |
|---|---|
| With lithium iodide at 180℃; for 7h; | 73% |
-
-
354-34-7
trifluoroacetyl fluoride

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
354-36-9
trifluoroacetyl iodide

| Conditions | Yield |
|---|---|
| With lithium iodide at 180℃; for 7h; | A 73% B 26% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; copper(l) iodide; iodine In N,N-dimethyl-formamide at 80℃; for 5h; | A 70% B n/a |

| Conditions | Yield |
|---|---|
| With hydrogen iodide; oxygen at 500℃; Product distribution / selectivity; | 67% |
| With oxygen; iodine monofluoride at 550℃; Product distribution / selectivity; | 40% |
| With iodine monofluoride at 550℃; Product distribution / selectivity; | 40% |
-
-
432-02-0
tris(trifluoromethyl)arsine

-
-
74-88-4
methyl iodide

-
A
-
75-46-7
trifluoromethan

-
B
-
2314-97-8
iodotrifluoromethane

-
C
-
431-76-5
methylbistrifluoromethylarsine

-
D
-
359-55-7
iodo-bis-trifluoromethyl-arsine

-
E
-
57028-26-9
Trifluormethyl-methyl-iodarsin

| Conditions | Yield |
|---|---|
| 235°C, 24 h; no formation of F3CCH3; | A n/a B 64% C n/a D n/a E n/a |

-
-
371-76-6
bis(trifluoromethyl)mercury

-
-
7790-44-5
antimony triiodide

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
432-05-3
tris(trifluoromethyl)stibane

-
C
-
1479-46-5
Bis(trifluormethyl)-jodstiban

| Conditions | Yield |
|---|---|
| SbI3 and Hg(CF3)2 heated to 165°C for 8h; at -131°C sepd. (CF3)2SbI and Sb(CF3)3; | A n/a B 63% C 2% |

-
-
61110-81-4
dichloro-trifluoromethyl-λ3-iodane

-
-
2966-50-9
silver trifluoroacetate

-
A
-
75-72-9
chlorotrifluoromethane

-
B
-
2314-97-8
iodotrifluoromethane

-
C
-
60669-28-5
Trifluoromethyliodine(III) bis-trifluoroacetate

| Conditions | Yield |
|---|---|
| In acetonitrile at -40℃; | A n/a B n/a C 56% |

-
-
432-04-2
tris(trifluoromethyl)phosphine

-
-
74-88-4
methyl iodide

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
1605-54-5
methyl-bis-trifluoromethyl-phosphine

| Conditions | Yield |
|---|---|
| 240°C, 24 h; | A n/a B 54% |

-
-
432-05-3
tris(trifluoromethyl)stibane

-
A
-
2314-97-8
iodotrifluoromethane

-
C
-
1479-46-5
Bis(trifluormethyl)-jodstiban

| Conditions | Yield |
|---|---|
| With iodine shaking in sealed tube for 30 min; | A n/a B 14% C 54% |
| With I2 shaking in sealed tube for 30 min; | A n/a B 14% C 54% |
| With iodine room temp.; | |
| With iodine room temp.; |

-
-
359-64-8
iodo-bis-trifluoromethyl-phosphine

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
76-16-4
Hexafluoroethane

-
C
-
421-59-0
trifluoromethyldiiodophosphine

-
D
-
432-04-2
tris(trifluoromethyl)phosphine

-
E
-
13455-01-1
phosphorous triiodide

| Conditions | Yield |
|---|---|
| 205°C (48 h); | A 2% B 8% C 2% D 45% E n/a |

-
-
421-59-0
trifluoromethyldiiodophosphine

-
A
-
359-64-8
iodo-bis-trifluoromethyl-phosphine

-
B
-
2314-97-8
iodotrifluoromethane

-
C
-
432-04-2
tris(trifluoromethyl)phosphine

-
D
-
13455-01-1
phosphorous triiodide

| Conditions | Yield |
|---|---|
| 240°C (48 h); | A 41% B 13% C 23% D n/a |
| In neat (no solvent) heating to 240°C;; |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 40% |
| With iodine heptafluoride; dihydrogen peroxide; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |
| With dihydrogen peroxide; iodine pentafluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 40% |
| With iodine heptafluoride; dihydrogen peroxide; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |
| With dihydrogen peroxide; iodine pentafluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 40% |
| With iodine heptafluoride; dihydrogen peroxide; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |
| With dihydrogen peroxide; iodine pentafluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 40% |
| With iodine heptafluoride; dihydrogen peroxide; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |
| With dihydrogen peroxide; iodine pentafluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |
-
-
14410-21-0
trifluoromethyl disulphide

-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 40% |
| With iodine heptafluoride; dihydrogen peroxide; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |
| With dihydrogen peroxide; iodine pentafluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 30% |
-
-
432-04-2
tris(trifluoromethyl)phosphine

-
-
7553-56-2
iodine

-
A
-
359-64-8
iodo-bis-trifluoromethyl-phosphine

-
B
-
2314-97-8
iodotrifluoromethane

-
C
-
421-59-0
trifluoromethyldiiodophosphine

-
D
-
13455-01-1
phosphorous triiodide

| Conditions | Yield |
|---|---|
| 180°C, 24 h; | A 16% B 34% C 4% D n/a |

-
-
432-02-0
tris(trifluoromethyl)arsine

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
353-91-3
trifluoromethyl-arsonous acid diiodide

-
C
-
359-55-7
iodo-bis-trifluoromethyl-arsine

| Conditions | Yield |
|---|---|
| 100-105°C, 72 h; | A 28% B n/a C 22% |


| Conditions | Yield |
|---|---|
| With hydrogen; iodine monofluoride; 3 weight% Pd/C In dimethyl sulfoxide; N,N-dimethyl-formamide at 100℃; Product distribution / selectivity; | 20% |
| With iodine monofluoride; zinc; 3 weight% Pd/C In dimethyl sulfoxide; N,N-dimethyl-formamide at 100℃; Product distribution / selectivity; | 20% |
| With iodine Product distribution; Irradiation; thermal + multiple photon IR laser excitation; different laser energy fluence and temperatures; |
-
-
432-02-0
tris(trifluoromethyl)arsine

-
-
74-88-4
methyl iodide

-
A
-
2314-97-8
iodotrifluoromethane

-
B
-
76-16-4
Hexafluoroethane

-
C
-
431-76-5
methylbistrifluoromethylarsine

| Conditions | Yield |
|---|---|
| Irradiation (UV/VIS); with UV-light; | A 18% B 15% C n/a |

-
-
105062-51-9
CF13IO2Te2

-
A
-
75-73-0
carbon tetrafluoride

-
B
-
2314-97-8
iodotrifluoromethane

-
C
-
105062-50-8
CF8OTe

| Conditions | Yield |
|---|---|
| at 25℃; for 2h; | A n/a B n/a C 17% D n/a |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 15% |
| With potassium iodide at 200℃; | |
| With iodine monofluoride; palladium on activated carbon; copper on carbon; platinum on activated charcoal at 250 - 550℃; Product distribution / selectivity; Vapor phase; | |
| With hydrogen iodide at 350℃; for 0.00555556h; Catalytic behavior; Reagent/catalyst; | 65.19 %Chromat. |
| With 0.5% Pd/Al2O3; hydrogen; iodine at 150 - 350℃; Flow reactor; |
-
-
81290-20-2
(trifluoromethyl)trimethylsilane

-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 15% |
| With iodine; oxygen; palladium on activated charcoal at 350℃; Product distribution / selectivity; | |
| With sulfuric acid; iodine; oxygen; bis(acetylacetonate)oxovanadium In Hexafluorobenzene at 30 - 150℃; for 12h; Product distribution / selectivity; | |
| With iodine monofluoride; palladium on activated carbon; copper on carbon; platinum on activated charcoal at 250 - 550℃; Product distribution / selectivity; Vapor phase; |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine monofluoride; bis(acetylacetonate)oxovanadium In water Product distribution / selectivity; Heating / reflux; | 15% |
| With iodine monofluoride; palladium on activated carbon; copper on carbon; platinum on activated charcoal at 250 - 550℃; Product distribution / selectivity; Vapor phase; |
-
-
2314-97-8
iodotrifluoromethane

-
-
36525-64-1
perfluoro-NN'-dimethylethane-1,2-bis(amino-oxyl) diradical

| Conditions | Yield |
|---|---|
| for 24h; Irradiation; | 100% |
-
-
2314-97-8
iodotrifluoromethane

-
-
373-68-2
tetramethylammonium fluoride

-
-
81290-20-2
(trifluoromethyl)trimethylsilane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -60℃; for 2h; | 100% |


| Conditions | Yield |
|---|---|
| With Tetrakis(dimethylamino)ethylen In N,N-dimethyl-formamide at 0 - 20℃; for 12h; | 100% |
| With Tetrakis(dimethylamino)ethylen In N,N-dimethyl-formamide at 0 - 20℃; |

-
-
2314-97-8
iodotrifluoromethane

-
-
865305-65-3
1-Isopropyl-5-methoxy-2-(toluene-4-sulfonyl)-4-trifluoromethyl-benzene

| Conditions | Yield |
|---|---|
| Stage #1: 1-iodo-4-isopropyl-2-methoxy-5-(toluene-4-sulfonyl)-benzene With potassium fluoride; copper(l) iodide In DMF (N,N-dimethyl-formamide) at 120℃; Stage #2: iodotrifluoromethane In DMF (N,N-dimethyl-formamide) for 4.5h; | 100% |
| With potassium fluoride; copper(l) iodide In N,N-dimethyl-formamide at 20℃; for 4.5h; | 100% |

| Conditions | Yield |
|---|---|
| In acetonitrile byproducts: EtI; Ar-atmosphere; stirring (-23°C); evapn. (vac.); | 100% |


-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With potassium fluoride; copper(l) iodide In N,N-dimethyl-formamide at 120℃; for 4h; | 100% |
| With potassium fluoride; copper(l) iodide In N,N-dimethyl-formamide at 120℃; for 4.5h; | 100% |
-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With paraquat dichloride; triethylamine In N,N-dimethyl-formamide at -78 - 20℃; for 18h; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| at -78℃; for 0.333333h; Schlenk technique; Inert atmosphere; Reflux; | 100% |
| at -78℃; for 0.333333h; Inert atmosphere; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| at -78 - 23℃; Schlenk technique; Inert atmosphere; | 100% |
| at -78 - 23℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| at -78 - 23℃; Schlenk technique; Inert atmosphere; | 100% |
| at -78 - 23℃; Inert atmosphere; | 100% |
| at -78℃; |
-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With paraquat dichloride; triethylamine In N,N-dimethyl-formamide at -78 - 20℃; for 24h; | 99.6% |
-
-
2314-97-8
iodotrifluoromethane

-
-
1666-13-3
diphenyl diselenide

-
-
5173-02-4
phenyl trifluoromethylselenide

| Conditions | Yield |
|---|---|
| With Tetrakis(dimethylamino)ethylen In N,N-dimethyl-formamide at 0 - 20℃; | 99% |
| With rongalite In water; N,N-dimethyl-formamide at 20℃; | 28% |
| With Tetrakis(dimethylamino)ethylen In N,N-dimethyl-formamide at 0 - 20℃; |

| Conditions | Yield |
|---|---|
| In chloroform byproducts: EtI; 2.5 equiv. CF3I, -40°C, 10 min (pptn.); removal of volatiles (vac.); | 99% |
| In not given absence of air and moisture; in presence of 1 equiv. HCF2I; |

-
-
2314-97-8
iodotrifluoromethane

-
-
60898-04-6
triluoromethyl(triphenylphosphine)gold(I)

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: MeI; 4 equiv. of CF3I, standing in sealed tube at 35°C for 45 min; evapn.; elem. anal.; | 99% |
-
-
2314-97-8
iodotrifluoromethane

-
-
73894-44-7
1-(8-Bromo-7-methoxy-1-phenyl-1,2,4,5-tetrahydro-benzo[d]azepin-3-yl)-2,2,2-trifluoro-ethanone

-
-
73894-49-2
2,2,2-Trifluoro-1-(7-methoxy-1-phenyl-8-trifluoromethyl-1,2,4,5-tetrahydro-benzo[d]azepin-3-yl)-ethanone

| Conditions | Yield |
|---|---|
| With copper In N,N-dimethyl-formamide at 150℃; | 98% |
-
-
2314-97-8
iodotrifluoromethane

-
-
73894-46-9
1-(8-Bromo-6-chloro-7-methoxy-1-phenyl-1,2,4,5-tetrahydro-benzo[d]azepin-3-yl)-2,2,2-trifluoro-ethanone

-
-
73894-50-5
1-(6-Chloro-7-methoxy-1-phenyl-8-trifluoromethyl-1,2,4,5-tetrahydro-benzo[d]azepin-3-yl)-2,2,2-trifluoro-ethanone

| Conditions | Yield |
|---|---|
| With copper In N,N-dimethyl-formamide at 150℃; | 98% |

| Conditions | Yield |
|---|---|
| With fac-tris(2-phenylpyridinato-N,C2')iridium(III); 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran; acetonitrile at 20℃; Sealed tube; Inert atmosphere; Irradiation; diastereoselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With pyrrolidine; Diphenylacetaldehyde In dichloromethane at 20℃; for 16h; Inert atmosphere; Irradiation; | 98% |
| With Eosin Y; sodium thiosulfate In acetonitrile at 20℃; for 1h; Inert atmosphere; Irradiation; | 53% |

| Conditions | Yield |
|---|---|
| With 5,10-di(4-trifluoromethylphenyl)-5,10-dihydrophenazine; potassium formate In N,N-dimethyl acetamide at 40℃; for 6h; Irradiation; | 98% |
-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine In dimethyl sulfoxide at 20℃; for 36h; Schlenk technique; Irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With platinum(II) (2-phenylpyridinato-N,C(2'))(2,4-pentanedionato-O,O); 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; for 6h; Quantum yield; Sealed tube; Irradiation; Inert atmosphere; | 97% |
| With tris(1,10-phenanthroline)ruthenium(II) dichloride; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile for 2h; Solvent; Inert atmosphere; | 96% |
| With tris(1,10-phenanthroline)ruthenium(II) dichloride; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; Inert atmosphere; Irradiation; regioselective reaction; | 95% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; tri-(9-anthryl)borane In acetonitrile at 20℃; for 3h; Reagent/catalyst; Solvent; Wavelength; Inert atmosphere; Irradiation; | 93% |
-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; eosin y In N,N-dimethyl-formamide at 20℃; for 0.5h; Reagent/catalyst; Concentration; Solvent; Time; Inert atmosphere; Irradiation; | 97% |
-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine In dimethyl sulfoxide at 20℃; for 36h; Schlenk technique; Irradiation; | 97% |

| Conditions | Yield |
|---|---|
| With (dppf)Ni(o-tol)Cl; sodium t-butanolate In Hexafluorobenzene at 50℃; for 16h; Inert atmosphere; Schlenk technique; Sealed tube; | 97% |
-
-
2314-97-8
iodotrifluoromethane

-
-
74465-47-7
(2R,3R,4R,5R)-2-(acetoxymethyl)-5-(6-bromo-9H-purin-9-yl)tetrahydrofuran-3,4-diyl diacetate

-
-
76513-94-5
(2R,3R,4R,5R)-2-(acetoxymethyl)-5-(6-(trifluoromethyl)-9H-purin-9-yl)tetrahydrofuran-3,4-diyl diacetate

| Conditions | Yield |
|---|---|
| With copper(l) iodide; zinc In N,N-dimethyl-formamide at 70℃; for 10h; | 96% |
-
-
2314-97-8
iodotrifluoromethane

-
-
223463-13-6
2-iodo-5-bromopyridine

-
-
7440-66-6
zinc

-
-
436799-32-5
5-bromo-2(trifluoromethyl)pyridine

| Conditions | Yield |
|---|---|
| Stage #1: iodotrifluoromethane; zinc With 1,3-dimethyl-3,4,5,6-tetrahydro-2(1H)-pyrimidinone at 20℃; for 2h; Inert atmosphere; Stage #2: 2-iodo-5-bromopyridine With copper(l) iodide at 50℃; for 24h; Catalytic behavior; Reagent/catalyst; Inert atmosphere; chemoselective reaction; | 96% |


| Conditions | Yield |
|---|---|
| With platinum(II) (2-phenylpyridinato-N,C(2'))(2,4-pentanedionato-O,O); 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; Sealed tube; Irradiation; Inert atmosphere; | 96% |
-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; eosin y In N,N-dimethyl-formamide at 20℃; for 0.5h; Inert atmosphere; Irradiation; | 96% |
-
-
2314-97-8
iodotrifluoromethane

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; eosin y In N,N-dimethyl-formamide at 20℃; for 0.5h; Inert atmosphere; Irradiation; | 96% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

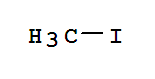











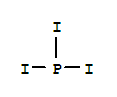


 Xi,
Xi, Xn
Xn