Changchun Artel lmport and Export trade company
minimum order qty. 10 gram
Cas:488-23-3
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:488-23-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:488-23-3
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:488-23-3
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
High purity CAS 488-23-3 1,2,3,4-Tetramethylbenzene in stock Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED intermediat
Cas:488-23-3
Min.Order:1 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:488-23-3
Min.Order:1 Kilogram
FOB Price: $100.0 / 150.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after the dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available &diam
Cas:488-23-3
Min.Order:1 Kilogram
FOB Price: $7.0 / 11.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Cas:488-23-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
1,2,3,4-Tetramethylbenzene CAS:488-23-3 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic
Cas:488-23-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:488-23-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:488-23-3
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:488-23-3
Min.Order:0 Metric Ton
Negotiable
Type:Other
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:488-23-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:488-23-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:Colorless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:Used in organic
Jiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Cas:488-23-3
Min.Order:0
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:488-23-3
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryShanghai Minstar Chemical Co., Ltd
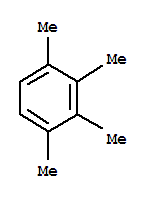
1,2,3,4-Tetramethylbenzene Basic information Product Name: 1,2,3,4-Tetramethylbenzene Synonyms: 1,2,3,4-tetramethyl-benzen;LABOTEST-BB LT00852111;DUROL;1,2,3,4-TETRAMETHYLBENZENE;Prehenitene;1,2,3,4-Tetramethylbenzene,tech.90%;Benzene, 1,2,3,4
Cas:488-23-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:488-23-3
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Golden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Win-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:488-23-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Lonwin Chemical Group Limited
Shanghai Lonwin Chemical company is a subsidiary of Lonwin Industry Group Limited, was established in 2011 and is headquartered in Shanghai, adjacent to China National Convention and Exhibition Center and Hongqiao transportation hub.Lonwinchem is
Cas:488-23-3
Min.Order:100 Kilogram
FOB Price: $100.0 / 150.0
Type:Other
inquiryAecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Hubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Synthetic route
-
-
38108-82-6
1,2-bis(bromomethyl)-3,6-dimethylbenzene

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 5℃; Temperature; Reagent/catalyst; Inert atmosphere; Reflux; | 97.3% |
| With tetrahydrofuran; lithium aluminium tetrahydride |
-
-
95-93-2
1,2,4,5-tetramethylbenzene

-
-
75-36-5
acetyl chloride

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
527-53-7
1,2,3,5-Tetramethylbenzene

-
C
-
2142-79-2
2,3,5,6-tetramethylacetophenone

| Conditions | Yield |
|---|---|
| With aluminium trichloride In tetrachloromethane 1.) 5 deg C, 1 h; 2.) 25 deg C, 1 h; | A 2.3% B 0.1% C 94.6% |

-
-
527-53-7
1,2,3,5-Tetramethylbenzene

-
-
108-24-7
acetic anhydride

-
A
-
95-93-2
1,2,4,5-tetramethylbenzene

-
B
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
C
-
2142-78-1
1-(2,3,4,6-tetramethyl-phenyl)-ethanone

-
D
-
2142-79-2
2,3,5,6-tetramethylacetophenone

| Conditions | Yield |
|---|---|
| With aluminium trichloride In carbon disulfide at 50℃; for 0.5h; Further byproducts given; | A 1.8% B 1.9% C 92.8% D 2.2% |

-
-
95-93-2
1,2,4,5-tetramethylbenzene

-
-
108-24-7
acetic anhydride

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
2142-79-2
2,3,5,6-tetramethylacetophenone

-
C
-
15517-58-5
1,1′-(2,3,5,6-tetramethyl-1,4-phenylene)bis(ethan-1-one)

| Conditions | Yield |
|---|---|
| aluminium trichloride In carbon disulfide 1.) 25 deg C, 1 h; 2.) 50 deg C, 3 h; | A 2.9% B 69.6% C 27.5% |


| Conditions | Yield |
|---|---|
| Stage #1: Cp2TiCl2 With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: dimethylacetylene In tetrahydrofuran; hexane at -10℃; for 3h; Stage #3: With propyl cyanide In tetrahydrofuran; hexane at 50℃; for 1h; | 53% |
-
-
18611-39-7
1,2-Diisopropyliden-3,4-dichlor-cyclobutan

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
1124-20-5
2-(3-methylphenyl)propene

-
C
-
76886-19-6
1-chloro-2-isopropyl-6-methylbenzene

| Conditions | Yield |
|---|---|
| at 250℃; for 2.5h; | A 51% B 11% C 30% |

-
-
18611-37-5
4-Chlor-2-chlormethylen-3-isopropyliden-1,1-dimethyl-cyclobutan

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
18611-39-7
1,2-Diisopropyliden-3,4-dichlor-cyclobutan

-
C
-
76886-19-6
1-chloro-2-isopropyl-6-methylbenzene

| Conditions | Yield |
|---|---|
| at 300℃; for 2.5h; Mechanism; pyrolysis of halogen derivatives of dimethylenecyclobutane and methylenecyclobutene investigated; | A 48% B n/a C 2% |

-
-
18611-37-5
4-Chlor-2-chlormethylen-3-isopropyliden-1,1-dimethyl-cyclobutan

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
1124-20-5
2-(3-methylphenyl)propene

-
C
-
76886-19-6
1-chloro-2-isopropyl-6-methylbenzene

| Conditions | Yield |
|---|---|
| at 300℃; for 2.5h; | A 48% B 28% C 2% |

-
-
115-10-6
Dimethyl ether

-
-
106-42-3
para-xylene

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
700-12-9
pentamethylbenzene,

-
C
-
87-85-4
Hexamethylbenzene

| Conditions | Yield |
|---|---|
| at 450℃; Leiten ueber Aluminiumoxyd-Siliciumdioxyd-Kontakte; |

-
-
95-93-2
1,2,4,5-tetramethylbenzene

-
-
527-53-7
1,2,3,5-Tetramethylbenzene

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid at 85℃; Erhitzen der gebildeten 1.2.3.4-Tetramethyl-benzol-sulfonsaeure-(5) mit wss. H2SO4 auf 145grad; |

| Conditions | Yield |
|---|---|
| With sulfuric acid Darstellung der Sulfonsaeure; |
-
-
17851-27-3
1-ethyl-2,4,5-trimethylbenzene

-
-
488-23-3
1,2,3,4-Tetramethylbenzene
-
-
559-64-8
sumaresinolic acid

-
A
-
582-16-1
2,7-dimethylnaphthalene

-
B
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
C
-
2131-43-3
1,2,5,6-tetramethylnaphthalene

-
D
-
486-34-0
1,2,7-trimethylnaphthalene

| Conditions | Yield |
|---|---|
| at 310℃; under 12 Torr; anschliessendes Erhitzen mit Selen auf 340-350grad; |


| Conditions | Yield |
|---|---|
| With diethyl ether; sodium |
-
-
41381-36-6
2-bromo-1,3,4-trimethyl-benzene

-
-
74-88-4
methyl iodide

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| With sodium; benzene at 150℃; im Einschlussrohr; |
-
-
95-63-6
1,2,4-Trimethylbenzene

-
A
-
95-47-6
o-xylene

-
B
-
106-42-3
para-xylene

-
C
-
95-93-2
1,2,4,5-tetramethylbenzene

-
D
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
E
-
527-53-7
1,2,3,5-Tetramethylbenzene

-
F
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With hydrogen; H Mordenite at 350℃; under 760 Torr; for 7h; Product distribution; variation of the catalyst; | |
| With USY zeolite (spiltover hydrogen); hydrogen In gas at 200℃; for 0.333333h; Product distribution; effect of spiltover hydrogen on the stabilization of catalytic activity; also on illared montmorillonite; |

-
-
95-63-6
1,2,4-Trimethylbenzene

-
A
-
95-47-6
o-xylene

-
B
-
106-42-3
para-xylene

-
C
-
526-73-8
1,2,3-trimethylbenzene

-
D
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
E
-
108-38-3
m-xylene

-
F
-
108-67-8
1,3,5-trimethyl-benzene

| Conditions | Yield |
|---|---|
| With HY zeolite at 200℃; under 12.8 Torr; Product distribution; Rate constant; Equilibrium constant; kinetics, Ea, mechanism; var. temp.; |

-
-
34764-71-1
2',3',4',5'-tetramethylacetophenone

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| With methoxybenzene; trifluoroacetic acid at 110℃; Rate constant; |
-
-
74692-88-9
1,2,3,4-tetramethyl-[5-3H]benzene

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| With acetic acid; trifluoroacetic acid Rate constant; changes in Vol percent TFA in HOAc; |

| Conditions | Yield |
|---|---|
| In acetonitrile Thermodynamic data; Rate constant; ΔG; |
-
-
130563-79-0
Anthracene-2,6,9,10-tetracarbonitrile; compound with 1,2,3,4-tetramethyl-benzene

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
80721-78-4
2,6,9,10-tetracyanoanthracene

| Conditions | Yield |
|---|---|
| In acetonitrile Thermodynamic data; Rate constant; ΔG; |
-
-
66312-03-6
2,3,4,5-Tetramethyltetracyclo<4.4.0.02,4.03,5>deca-7,9-dien-1,6-dicarbonsaeureanhydrid

-
A
-
85-44-9
phthalic anhydride

-
B
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
C
-
3031-15-0
1,2,3,4-tetramethylnaphthalene

-
D
-
80673-73-0
3,9b-Dihydro-9b-hydroxy-1,2,3a-trimethyl-3-methylencyclopenta<2>benzopyran-5(3aH)-on

-
E
-
80684-79-3
3,4,5,6,9,10,11,12-Octamethylpentacyclo<6.4.2.02,7.03,6.09,12>tetradeca-4,10,13-trien-2,7-dicarbonsaeureanhydrid

| Conditions | Yield |
|---|---|
| at 145℃; for 0.833333h; Product distribution; |

-
-
95-93-2
1,2,4,5-tetramethylbenzene

-
-
75-36-5
acetyl chloride

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
2142-79-2
2,3,5,6-tetramethylacetophenone

| Conditions | Yield |
|---|---|
| aluminium trichloride In carbon disulfide at 25℃; for 1h; Product distribution; other acetylation agent, other solvents, other conditions, further tetramethylbenzenes; |

-
-
527-53-7
1,2,3,5-Tetramethylbenzene

-
-
34764-71-1
2',3',4',5'-tetramethylacetophenone

-
A
-
95-93-2
1,2,4,5-tetramethylbenzene

-
B
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
C
-
2142-78-1
1-(2,3,4,6-tetramethyl-phenyl)-ethanone

-
D
-
2142-79-2
2,3,5,6-tetramethylacetophenone

| Conditions | Yield |
|---|---|
| With aluminium trichloride In nitromethane at 20℃; for 15h; Product distribution; Mechanism; further solvent, methyl substituted acetophenones, with or without methyl substituted benzenes; | A 29.6 % Chromat. B 4.6 % Chromat. C 0.9 % Chromat. D 16.2 % Chromat. |


| Conditions | Yield |
|---|---|
| In various solvent(s) at -20℃; Rate constant; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| at 400℃; |
-
-
95-93-2
1,2,4,5-tetramethylbenzene

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
527-53-7
1,2,3,5-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| at 26.9℃; Zusammensetzung des Gleichgewichtsgemisches; |

-
-
95-93-2
1,2,4,5-tetramethylbenzene

-
A
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
B
-
527-53-7
1,2,3,5-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| at 426.9℃; Zusammensetzung des Gleichgewichtsgemisches; |

-
-
16957-70-3
2-methylpent-2-enoic acid

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylpent-2-enoic acid With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 25℃; for 2h; Inert atmosphere; Stage #2: With aluminum (III) chloride In dichloromethane at 8℃; Inert atmosphere; Stage #3: 1,2,3,4-Tetramethylbenzene In dichloromethane at 25℃; for 4h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; palladium diacetate; trifluoroacetic acid at 50℃; for 18h; Inert atmosphere; Sealed tube; | 98% |
-
-
50-00-0
formaldehyd

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
55743-69-6
5,6-bis(bromomethyl)-1,2,3,4-tetramethylbenzene

| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid at 100℃; for 8h; | 97% |
| With sulfuric acid; acetic acid; sodium bromide for 3h; Heating; | 6% |
| With hydrogen bromide; acetic acid |
-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
54509-71-6
1-iodo-2,3,4,5-tetramethylbenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; iodine; nitric acid In acetic acid at 50 - 55℃; for 5h; | 97% |
| With iodine; Selectfluor In acetonitrile at 55 - 65℃; for 1h; | 85% |

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
16836-95-6
silver(I) 4-methylbenzenesulfonate

-
-
75751-43-8
[Ir(C6H2(CH3)4)((CHCHC2H4)2)](1+)*OSO2C6H4CH3(1-)=[Ir(C6H2(CH3)4)((CHCHC2H4)2)]OSO2C6H4CH3

| Conditions | Yield |
|---|---|
| In ethanol; dichloromethane; acetone (N2); silver tosylate treated with acetone/EtOH/C6H2(CH3)4, treated with Ir complex/CH2Cl2, stirred for 14 h at room temp.; filtered, concd., Et2O added, decanted, dried (vac.), recrystd. from CH2Cl2/Et2O/pentane and CH2Cl2/Et2O; elem. anal.; | 97% |

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
75-36-5
acetyl chloride

-
A
-
95-93-2
1,2,4,5-tetramethylbenzene

-
B
-
34764-71-1
2',3',4',5'-tetramethylacetophenone

| Conditions | Yield |
|---|---|
| aluminium trichloride In carbon disulfide at 25℃; for 3h; | A 2.9% B 96.4% |
| With aluminium trichloride In carbon disulfide at 25℃; for 3h; | A 4.2% B 91.8% |

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
19219-82-0
1,2,3,4-tetramethyl-2,5-dichlorobenzene

| Conditions | Yield |
|---|---|
| With benzyl(trimethyl)ammonium tetrachloroiodate In acetic acid for 20h; Ambient temperature; | 96% |
| With 1,3-dichloro-5,5-dimethylhydantoin; acetic acid at 90℃; for 2.5h; | 33% |
| With 1,3-dichloro-5,5-dimethylhydantoin In acetic acid at 90℃; for 2.5h; | 33% |
| With aluminium trichloride; sulfuryl dichloride |
-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
A
-
40101-36-8
1-Bromo-2,3,4,5-tetramethylbenzene

-
B
-
36321-73-0
1,2-dibromo-3,4,5,6-tetramethylbenzene

| Conditions | Yield |
|---|---|
| With benzyltrimethylazanium tribroman-2-uide; zinc(II) chloride In acetic acid for 2h; Ambient temperature; | A 96% B 85% |
-
-
80-59-1
Tiglic acid

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
163842-93-1
2,3,4,5,6,7-tetramethylindane-1-one

| Conditions | Yield |
|---|---|
| Stage #1: Tiglic acid With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane for 12h; Inert atmosphere; Stage #2: 1,2,3,4-Tetramethylbenzene With aluminum (III) chloride In dichloromethane at 8 - 25℃; for 4h; Inert atmosphere; | 96% |
-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
75-36-5
acetyl chloride

-
A
-
2142-79-2
2,3,5,6-tetramethylacetophenone

-
B
-
34764-71-1
2',3',4',5'-tetramethylacetophenone

| Conditions | Yield |
|---|---|
| With aluminium trichloride In chloroform at 25℃; for 1h; | A 4.1% B 95.3% |


| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid at 80℃; for 3h; | 95% |
-
-
381-98-6
trifluoromethyacrylic acid

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
1219921-42-2
2-trifluoromethyl-3-(4',5',6',7'-tetramethylphenyl)-1-indanone

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 0 - 20℃; Friedel Crafts reaction; | 95% |
-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
14202-27-8, 51830-12-7, 67315-16-6, 70095-87-3, 70494-79-0, 139686-29-6, 139686-31-0, 139686-32-1
(+/-) methyl (4,5)-trans-epoxy-(2E)-hexenoate

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane at -78 - -20℃; for 2h; | 94% |
-
-
52462-29-0
[ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
88946-78-5
bis[dichloro-(η(6)-1,2,3,4-tetramethylbenzene)ruthenium]

| Conditions | Yield |
|---|---|
| In neat (no solvent) Ru-complex was stirred in excess refluxing arene under N2 for 24 h; soln. was cooled, ppt. collected, washed with hexane; | 94% |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
1193-82-4
racemic methyl phenyl sulfoxide

-
-
1402011-74-8
S-methyl-S-phenyl-2,3,4,5-tetramethylphenyl sulfonium triflate

| Conditions | Yield |
|---|---|
| In diethyl ether at 0 - 5℃; Inert atmosphere; | 93.67% |

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
1193-82-4
racemic methyl phenyl sulfoxide

-
-
1025373-45-8
trifluoromethanesulfonic acid anhydride

-
-
1402011-74-8
S-methyl-S-phenyl-2,3,4,5-tetramethylphenyl sulfonium triflate

| Conditions | Yield |
|---|---|
| In diethyl ether at 0 - 5℃; Inert atmosphere; | 93.67% |

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
108-24-7
acetic anhydride

-
A
-
15517-58-5
1,1′-(2,3,5,6-tetramethyl-1,4-phenylene)bis(ethan-1-one)

-
B
-
34764-71-1
2',3',4',5'-tetramethylacetophenone

-
C
-
69313-51-5
1,3-diacetyl-2,4,5,6-tetramethylbenzene

| Conditions | Yield |
|---|---|
| With aluminium trichloride In carbon disulfide at 50℃; for 2h; | A 5.4% B 90.6% C 2.8% |

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
117965-56-7
2,3-bis(bromomethyl)-1,4-bis(dibromomethyl)benzene

| Conditions | Yield |
|---|---|
| With bromine In tetrachloromethane for 1h; Heating; Irradiation; | 90% |
-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
814-68-6
acryloyl chloride

-
-
711-43-3
4,5,6,7-tetramethyl-1H-indan-1-one

| Conditions | Yield |
|---|---|
| With aluminium trichloride Cyclization; acylation; Heating; | 90% |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
24824-93-9
chloromethyl p-tolyl sulfoxide

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
1374153-80-6
(chloromethyl)(2,3,4,5-tetramethylphenyl)(p-tolyl)sulfonium triflate

| Conditions | Yield |
|---|---|
| In diethyl ether at 0 - 5℃; Inert atmosphere; | 89.74% |

-
-
24824-93-9
chloromethyl p-tolyl sulfoxide

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

| Conditions | Yield |
|---|---|
| Stage #1: chloromethyl p-tolyl sulfoxide; 1,2,3,4-Tetramethylbenzene With trifluoromethylsulfonic anhydride In diethyl ether at -60℃; for 4h; Stage #2: With tetrafluoroboric acid In diethyl ether for 0.5h; | 89% |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
54234-79-6, 67529-46-8, 109120-75-4, 21865-07-6
propyl phenyl sulfoxide

| Conditions | Yield |
|---|---|
| In diethyl ether at -78℃; for 2h; Inert atmosphere; | 89% |

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
36321-73-0
1,2-dibromo-3,4,5,6-tetramethylbenzene

| Conditions | Yield |
|---|---|
| With bromine; iodine In dichloromethane for 3h; | 88% |
| With bromine; zinc(II) chloride In acetic acid at 20℃; for 14h; | 85% |
| With bromine; zinc(II) chloride In acetic acid at 20℃; for 14h; | 85% |

| Conditions | Yield |
|---|---|
| With N,N,N-trimethylbenzenemethanaminium dichloroiodate; zinc(II) chloride In acetic acid at 70℃; for 24h; | 88% |
| With sulfuric acid; iodine; periodic acid In water; acetic acid at 50 - 55℃; Suzuki iodination; | 83% |
| With iodine; Selectfluor In acetonitrile at 55 - 65℃; for 2.5h; | 67% |
| With sulfuric acid; iodine; periodic acid In acetic acid | |
| With sulfuric acid; iodine; periodic acid In acetic acid |
-
-
96-48-0
4-butanolide

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
147792-79-8
4-(2,3,4,5-Tetramethylphenyl)buttersaeure

| Conditions | Yield |
|---|---|
| With aluminium trichloride at 60℃; for 2h; | 88% |
-
-
35660-94-7
(E)-2-methylbut-2-enoyl chloride

-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
163842-93-1
2,3,4,5,6,7-tetramethylindane-1-one

| Conditions | Yield |
|---|---|
| With aluminium trichloride In carbon disulfide Heating; | 88% |
| With aluminium trichloride Cyclization; acylation; Heating; | 87% |
| Stage #1: (E)-2-methylbut-2-enoyl chloride With aluminum (III) chloride In dichloromethane Inert atmosphere; Stage #2: 1,2,3,4-Tetramethylbenzene In dichloromethane at 25℃; for 4h; Inert atmosphere; | 77.25 g |
-
-
488-23-3
1,2,3,4-Tetramethylbenzene

-
-
42887-62-7
2,3,4,5-tetramethyl-1-nitrobenzene

| Conditions | Yield |
|---|---|
| With silver nitrate; boron trifluoride In acetonitrile at 25℃; for 8h; | 86% |
| With silver nitrate; boron trifluoride In acetonitrile at 25℃; for 5h; competitive nitration benzene, relative rate; | |
| With nitric acid |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View















 F
F