-
Name
2-Thiopheneboronic acid
- EINECS 612-186-6
- CAS No. 6165-68-0
- Article Data35
- CAS DataBase
- Density 1.321 g/cm3
- Solubility
- Melting Point 138-140 °C(lit.)
- Formula C4H5BO2S
- Boiling Point 287.917 °C at 760 mmHg
- Molecular Weight 127.959
- Flash Point 127.928 °C
- Transport Information
- Appearance white to light yellow crystal powder
- Safety 26-36/37-37/39-36
- Risk Codes 22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms 2-Thiopheneboronicacid (6CI,7CI,8CI);Boronic acid, 2-thienyl- (9CI);2-Thienylboric acid;Thien-5-ylboronic acid;thiophen-2-ylboronic acid;Thiophene-2-boronic acid;
- PSA 68.70000
- LogP -0.57210
Synthetic route
-
-
1158984-92-9
2-thiopheneboronic acid MIDA ester

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In tetrahydrofuran; water at 23℃; for 0.333333h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Iodothiophene With diisopropopylaminoborane; triethylamine; triphenylphosphine; palladium dichloride In tetrahydrofuran at 65℃; for 12h; Alcaraz-Vaultier borylation; Inert atmosphere; Stage #2: With methanol In tetrahydrofuran at 0℃; Inert atmosphere; Further stages; | 99% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
193978-23-3
2-thiopheneboronic acid pinacol ester

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water; acetone at 20℃; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromothiophene With tris(dibenzylideneacetone)dipalladium(0) chloroform complex; diisopropopylaminoborane; triethylamine; triphenylphosphine In tetrahydrofuran at 65℃; for 12h; Alcaraz-Vaultier borylation; Inert atmosphere; Stage #2: With methanol In tetrahydrofuran at 0℃; Inert atmosphere; Further stages; | 87% |
| With n-butyllithium; diethyl ether at -60℃; anschliessend Behandeln mit Tributylborat und Behandeln des Reaktionsgemisches mit wss. Salzsaeure; | |
| Stage #1: 2-bromothiophene With Trimethyl borate In N,N-dimethyl-formamide at 20℃; Electrochemical reaction; Mg consumable anode, supporting electrolyte; Stage #2: at 0℃; Acid hydrolysis; Further stages.; | |
| With hydrogenchloride; borane; magnesium In tetrahydrofuran; ice-water |

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| With water; silica gel at 20℃; for 3h; Inert atmosphere; | 84% |
-
-
1003-09-4
2-bromothiophene

-
-
67-56-1
methanol

-
-
55124-35-1
diisopropylamine borane

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| Stage #1: diisopropylamine borane With magnesium; phenylmagnesium bromide In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: 2-bromothiophene In tetrahydrofuran at 70℃; Stage #3: methanol Further stages; | 82% |

-
-
188290-36-0
thiophene

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| Stage #1: thiophene With n-butyllithium In tetrahydrofuran at -78 - -20℃; Stage #2: With Triisopropyl borate In tetrahydrofuran at -78 - 20℃; | 78% |
| Stage #1: thiophene With n-butyllithium In tetrahydrofuran at -78 - -20℃; Stage #2: With Triisopropyl borate In tetrahydrofuran at -78 - 20℃; Stage #3: With hydrogenchloride In tetrahydrofuran Further stages.; | 78% |
| Stage #1: thiophene With n-butyllithium In tetrahydrofuran at 0℃; Inert atmosphere; Stage #2: With Trimethyl borate In tetrahydrofuran at -78 - 20℃; Stage #3: With sulfuric acid In tetrahydrofuran | 55% |
| Stage #1: thiophene With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 2h; Inert atmosphere; Stage #2: With Trimethyl borate In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromothiophene With n-butyllithium In diethyl ether; hexane at -78℃; for 4h; Inert atmosphere; Stage #2: Trimethyl borate In diethyl ether; hexane at -78 - 20℃; Inert atmosphere; Stage #3: With hydrogenchloride In water | 78% |
-
-
1003-09-4
2-bromothiophene

-
-
22092-92-8
diisopropopylaminoborane

-
-
7732-18-5
water

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromothiophene; diisopropopylaminoborane With magnesium In tetrahydrofuran at 65℃; for 4h; Inert atmosphere; Stage #2: water With hydrogenchloride In tetrahydrofuran at 65℃; for 0.25h; Barbier Coupling Reaction; Inert atmosphere; | 74% |


| Conditions | Yield |
|---|---|
| With water In tetrahydrofuran slow addn. of 10 mmol of the Grignard reagent to a stirred soln. of borane in THF (40 mmol); the mixt. was poured into ice-water and acidified with 10% HCl;; extn. into ether three-times; the combined extracts were dried over NaSO4; removal of solvent under reduced pressure; trituration with petroleum ether; recrystn. from H2O;; | 68% |
-
-
121-43-7
Trimethyl borate

-
-
5713-61-1
thiophen-2-yl magnesium bromide

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| Grignard reaction; | 65% |

| Conditions | Yield |
|---|---|
| Stage #1: thiophene With n-butyllithium In tetrahydrofuran; hexane at -78 - -20℃; for 1h; Inert atmosphere; Stage #2: triethyl borate In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Stage #3: With hydrogenchloride; water In tetrahydrofuran; hexane Inert atmosphere; | 65% |
| With n-butyllithium In tetrahydrofuran thiophene and n-BuLi in hexane mixed in THF at 0°C under N2; B(OEt)2 added dropwise at -70°C; temp. raised to room temp. and aq. H2SO4 added followed CH2Cl2; org. layer extd. with aq. NaOH; to aq. layer added aq. HCl and compd. extd. with dichloromethane; | 43% |
-
-
96-43-5
2-thienyl chloride

-
-
121-43-7
Trimethyl borate

-
A
-
188290-36-0
thiophene

-
B
-
98-03-3
thiophene-2-carbaldehyde

-
C
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| With aluminium In N,N-dimethyl-formamide byproducts: Al(3+); Electrolysis; electrolysis carried out in a single-compartment cell at room temp., starting materials dissolved in DMF containing KBr or n-Bu4NBr, solvent andexcess B(OCH3)3 evaporated under vacuum, medium hydrolysed at 0° C with an HCl or H2SO4 soln.; extraction with Et2O (3 x 20 ml if the solvent evaporated or 3 x 60 ml otherwise), dried over sodium or magnesium sulfate and concentrated undervacuum, side product phenol formation avoided by work-up under inert at mosphere, recrystallized in CH2Cl2; | A n/a B n/a C 59% |

-
-
1003-09-4
2-bromothiophene

-
-
121-43-7
Trimethyl borate

-
A
-
188290-36-0
thiophene

-
B
-
98-03-3
thiophene-2-carbaldehyde

-
C
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| With magnesium In N,N-dimethyl-formamide byproducts: Mg(2+); Electrolysis; electrolysis carried out in a single-compartment cell at room temp., starting materials dissolved in DMF containing KBr or n-Bu4NBr, solvent andexcess B(OCH3)3 evaporated under vacuum, medium hydrolysed at 0° C with an HCl or H2SO4 soln.; extraction with Et2O (3 x 20 ml if the solvent evaporated or 3 x 60 ml otherwise), dried over sodium or magnesium sulfate and concentrated undervacuum, side product phenol formation avoided by work-up under inert at mosphere, recrystallized in CH2Cl2; | A n/a B n/a C 57% |
| With aluminium In N,N-dimethyl-formamide byproducts: Al(3+); Electrolysis; electrolysis carried out in a single-compartment cell at room temp., starting materials dissolved in DMF containing KBr or n-Bu4NBr, solvent andexcess B(OCH3)3 evaporated under vacuum, medium hydrolysed at 0° C with an HCl or H2SO4 soln.; extraction with Et2O (3 x 20 ml if the solvent evaporated or 3 x 60 ml otherwise), dried over sodium or magnesium sulfate and concentrated undervacuum, side product phenol formation avoided by work-up under inert at mosphere, recrystallized in CH2Cl2; | A n/a B n/a C 56% |


| Conditions | Yield |
|---|---|
| Stage #1: 2-bromothiophene With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; Stage #2: triethyl borate In tetrahydrofuran; hexane at -78 - 20℃; Stage #3: With hydrogenchloride; water Cooling; | 45% |

-
A
-
6165-68-0
thiophene boronic acid

-
B
-
6165-69-1
Thien-3-ylboronic acid

-
C
-
1379794-79-2
thiophene-2,3-diyldiboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: C12H19BLiNO2S With thiophene; triethyl borate Inert atmosphere; Stage #2: With sulfuric acid In water | A 9% B 9% C n/a |

-
-
2786-07-4
2-thienyl lithium

-
-
60-29-7
diethyl ether

-
-
688-74-4
boric acid tributyl ester

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| anschliessend Behandeln mit wss. Salzsaeure; |


| Conditions | Yield |
|---|---|
| With diethyl ether at -75℃; Behandeln des Reaktionsgemisches mit wss. Schwefelsaeure; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-thienyl chloride With Trimethyl borate In N,N-dimethyl-formamide at 20℃; Electrochemical reaction; Al consumable anode, supporting electrolyte; Stage #2: at 0℃; Acid hydrolysis; Further stages.; | |
| With methanol; tetrakis(dimethylamido)diborane; chloro(2-dicyclohexylphosphino-2′,4′,6′-triisopropyl-1,1′-biphenyl)[2-(2′-amino-1,1′-biphenyl)]palladium(II); potassium acetate; XPhos at 20 - 60℃; for 5.0833h; Inert atmosphere; Sealed tube; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; n-butyllithium | |
| With hydrogenchloride; n-butyllithium |

| Conditions | Yield |
|---|---|
| With water; magnesium In tetrahydrofuran a 2.0 M soln. of borane (40 mmol) in THF was added to flame dried Mg turnings (12 mmol) at 0°C; slow addn. of the aryl bromide (10 mmol)in THF; stirring at room temp. for 12-16 h; the mixt. was poured into ice-water and acidified with 10% HCl;; extn. into ether three-times; the combined extracts were dried over NaSO4; removal of solvent under reduced pressure; trituration with petroleum ether; recrystn. from H2O;; |

| Conditions | Yield |
|---|---|
| In not given hydrolysis of appropriate N-methyliminodiacetic acid boronate; |

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| Stage #1: C12H19BLiNO2S With thiophene; triethyl borate Inert atmosphere; Stage #2: With sulfuric acid In water |

| Conditions | Yield |
|---|---|
| With triethyl borate In tetrahydrofuran at -78℃; for 1h; Inert atmosphere; Schlenk technique; | 10 g |

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 20℃; Inert atmosphere; Schlenk technique; | |
| With hydrogenchloride In water for 0.5h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| Stage #1: thiophene With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Stage #2: Trimethyl borate In tetrahydrofuran at -78℃; for 1h; | |
| Stage #1: thiophene With n-butyllithium In tetrahydrofuran at -78℃; for 1h; Stage #2: Trimethyl borate In tetrahydrofuran at -78 - 20℃; for 1h; |

| Conditions | Yield |
|---|---|
| With sodium carbonate; trans-PdBr(N-Succ)(PPh3)2 In tetrahydrofuran at 60℃; for 16h; | 100% |
| With copper fluorapatite In methanol at 20℃; for 2h; Green chemistry; | 95% |
| With C34H54N2O2; palladium diacetate; potassium carbonate; p-toluenesulfonyl chloride In water at 0 - 25℃; for 0.25h; Reagent/catalyst; Green chemistry; | 92% |
-
-
908254-94-4
7-[di(phenyloxy)phosphoryloxy]-8-(tert-butoxycarbonyl)-9-phenyl-8-azaspiro[4.5]deca-6,9-diene

-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate; bis-triphenylphosphine-palladium(II) chloride In tetrahydrofuran; ethanol; water for 3h; Suzuki-Miyaura coupling; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In ethanol; water; toluene for 18h; Inert atmosphere; Reflux; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; sodium carbonate In water; isopropyl alcohol at 80℃; for 0.666667h; Suzuki Coupling; Microwave irradiation; | 91% |
| With bis(triphenylphosphine)palladium(II) dichloride; potassium carbonate In 1,2-dimethoxyethane; water at 80℃; for 14h; | 86% |
-
-
6165-68-0
thiophene boronic acid

-
-
100-00-5
4-chlorobenzonitrile

-
-
59156-21-7
2-(4-nitrophenyl)thiophene

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; tricyclohexylphosphine; dichloro bis(acetonitrile) palladium(II) at 85 - 90℃; for 5h; Suzuki-Miyaura reaction; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane; potassium carbonate; palladium diacetate In N,N-dimethyl-formamide at 110℃; for 19h; Suzuki-Miyaura cross-coupling; | 93% |
| With potassium phosphate tribasic trihydrate; (2-mesityl-1H-inden-3-yl)dicyclohexyl phosphine; palladium diacetate; Aliquat 336 In water at 20 - 100℃; for 8h; Suzuki-Miyaura Coupling; Inert atmosphere; | 86% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,2-dimethoxyethane; water at 70℃; for 10h; Suzuki-Miyaura reaction; | 73% |
-
-
6165-68-0
thiophene boronic acid

-
-
6630-33-7
ortho-bromobenzaldehyde

-
-
99902-07-5
2-(2-thienyl)benzaldehyde

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In 1,2-dimethoxyethane; water for 3.25h; Suzuki-Miyaura Coupling; Inert atmosphere; Reflux; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In 1,2-dimethoxyethane; water for 3.25h; Suzuki-Miyaura Coupling; Inert atmosphere; Reflux; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In water; N,N-dimethyl-formamide at 80℃; for 12h; Inert atmosphere; Schlenk technique; | 89% |
-
-
6165-68-0
thiophene boronic acid

-
-
1027067-02-2
Thiophen-2-yl-imidazo[1,2-a]pyrazin-8-ylamine

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; methanol; water | 100% |

-
-
6165-68-0
thiophene boronic acid

-
-
1007402-19-8
5-bromo-3-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluorodecane-1-sulfonyl)-3-(2-nitrobenzyl)-1-propyl-1,3-dihydroindol-2-one

-
-
1007402-26-7
3-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluorodecane-1-sulfonyl)-3-(2-nitrobenzyl)-1-propyl-5-thiophen-2-yl-1,3-dihydroindol-2-one

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,4-dioxane; water at 80℃; for 5h; Suzuki coupling; | 100% |
-
-
852923-47-8
2-butynyl-[(2E)-4-hydroxy-2-butenyl]propanedioic acid dimethyl ester

-
-
6165-68-0
thiophene boronic acid

-
-
1061620-29-8
(4Z)-3-ethenyl-4-[1-(thiophen-2-yl)ethylidene]-1,1-cyclopentanedicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 80℃; for 3h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With C11H8Cl4N2O3Pd; tetrabutylammomium bromide; potassium carbonate In methanol; water; N,N-dimethyl-formamide at 100℃; for 0.166667h; Suzuki Coupling; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; tetrabutylammomium bromide; potassium carbonate In chloroform; water at 80 - 100℃; for 0.166667h; Suzuki Coupling; | 99% |
| With 2-[5-(4-methylphenyl)isoxazol-3-yl]-5-(5-phenylisoxazol-3-yl)-1,3,4-oxadiazole·2PdCl2; tetrabutylammomium bromide; potassium carbonate In water; N,N-dimethyl-formamide at 100℃; for 0.0166667h; Suzuki Coupling; | 98% |
-
-
6165-68-0
thiophene boronic acid

-
-
147497-32-3
6-bromo-3,4-dihydroisoquinolin-1(2H)-one

-
-
1309955-17-6
6-(thiophen-2-yl)-3,4-dihydroisoquinolin-1(2H)-one

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium carbonate In ethanol; toluene Suzuki Coupling; Inert atmosphere; Reflux; | 100% |
| With tetrabutylammomium bromide; palladium diacetate; potassium carbonate In water at 70℃; for 2.5h; Suzuki coupling; Inert atmosphere; | 83% |
-
-
6165-68-0
thiophene boronic acid

-
-
1354742-82-7
2-(carboxymethyl)-5-(thiophen-2-yl)benzoic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water at 150℃; for 0.5h; Suzuki coupling; Inert atmosphere; Microwave irradiation; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water at 150℃; for 0.5h; Suzuki Coupling; Microwave irradiation; |
-
-
6165-68-0
thiophene boronic acid

-
-
1378020-97-3
N-(adamantan-1-yl)-5-bromo-1-pentyl-1H-indole-3-carboxamide

-
-
1378020-81-5
N-(adamantan-1-yl)-1-pentyl-5-(thiophen-2-yl)-1H-indole-3-carboxamide

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate; triphenylphosphine In 1,2-dimethoxyethane; ethanol; water at 150℃; for 0.166667h; Suzuki coupling; | 100% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium hydrogencarbonate In 1,2-dimethoxyethane; water for 9h; Suzuki-Miyaura Coupling; Reflux; | 100% |
-
-
6165-68-0
thiophene boronic acid

-
-
1286738-68-8
6-(4-bromo-2-chlorophenyl)-8-ethyl-2-(4-(4-methylpiperazin-1-yl)phenylamino)pyrido[2,3-d]pyrimidin-7(8H)-one

-
-
1286738-65-5
C30H29ClN6OS

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium phosphate In water; N,N-dimethyl-formamide at 140℃; for 0.5h; Reagent/catalyst; Solvent; Inert atmosphere; Microwave irradiation; | 100% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 2.5h; Reflux; | 100% |

-
-
6165-68-0
thiophene boronic acid

-
-
1345882-37-2
5-bromo-2,3-dimethylisoindolin-1-one

-
-
1567837-17-5
2,3-dimethyl-5-(thiophen-2-yl)isoindolin-1-one

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium carbonate In ethanol; toluene Suzuki Coupling; Inert atmosphere; Reflux; | 100% |
-
-
6165-68-0
thiophene boronic acid

-
-
20870-78-4
5-bromo-2-indolin-2-one

-
-
189748-08-1
5-(thiophen-2-yl)indolin-2-one

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium carbonate In ethanol; toluene Suzuki Coupling; Inert atmosphere; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; methanesulfonic acid(2-dicyclohexylphosphino-2′,4′,6′-triisopropyl-1,1′-biphenyl)[2-(2′-amino-1,1′-biphenyl)]palladium(II) In water; dimethyl sulfoxide at 20℃; for 20h; Reagent/catalyst; Solvent; Suzuki Coupling; | 100% |
-
-
6165-68-0
thiophene boronic acid

-
-
27725-15-1
2-(2-hydroxy-5-methylphenylsulfinyl)-4-methylphenol

| Conditions | Yield |
|---|---|
| With magnesium sulfate In tetrahydrofuran at 20℃; for 20h; Inert atmosphere; | 100% |
-
-
6165-68-0
thiophene boronic acid

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; 1,2-bis-(diphenylphosphino)ethane In 1,4-dioxane; water at 80℃; Suzuki Coupling; Inert atmosphere; | 100% |
-
-
6165-68-0
thiophene boronic acid

-
-
89-63-4
4-Chloro-2-nitroaniline

-
-
405170-93-6
2-nitro-4-thiophen-2-yl-phenylamine

| Conditions | Yield |
|---|---|
| With potassium phosphate; (2-dicyclohexylphosphino-2’,4’,6’-triisopropyl-1,1 ‘-biphenyl)[2-(2’-amino-1,1‘-biphenyl)]palladium(II) methanesulfonate In 1,4-dioxane; water for 20h; Sealed tube; Schlenk technique; Inert atmosphere; | 100% |
-
-
6165-68-0
thiophene boronic acid

-
-
95-56-7
2-hydroxybromobenzene

-
-
106584-13-8
2-(2'-hydroxyphenyl)thiophene

| Conditions | Yield |
|---|---|
| With bis(tri-t-butylphosphine)palladium(0); potassium carbonate In tetrahydrofuran; water at 80℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; | 100% |
-
-
6165-68-0
thiophene boronic acid

-
-
619-58-9
4-iodobenzoic acid

-
-
29886-62-2
4-thiophen-2-yl-benzoic acid

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium carbonate In 1,4-dioxane; water for 12h; Suzuki Coupling; Reflux; | 99% |
| With tris(dibenzylideneacetone)dipalladium (0); Sasrin resin; potassium carbonate; benzotriazol-1-ol; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide; triethylamine; trifluoroacetic acid 1) CH2Cl2, rt, 20 h; 2) DMF, rt, 20 h; 3) CH2Cl2, rt, 30 min; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With potassium carbonate; [PS-PEG-adppp-Pd(η3-C3H5)]Cl In water at 50℃; for 12h; Suzuki-Miyaura coupling; | 99% |
| Stage #1: thiophene boronic acid; iodobenzene With potassium phosphate In 1,2-dimethoxyethane at 80℃; for 6h; Suzuki-Miyaura Coupling; Inert atmosphere; Stage #2: In 1,2-dimethoxyethane; water at 100℃; for 10h; | 94% |
| With potassium phosphate; C24H44BClN2P2Pd In toluene at 80℃; for 1h; Catalytic behavior; Suzuki-Miyaura Coupling; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; [PS-PEG-adppp-Pd(η3-C3H5)]Cl In water at 50℃; for 12h; Suzuki-Miyaura coupling; | 99% |
| With tetrakis(triphenylphosphine) palladium(0); sodium hydrogencarbonate In tetrahydrofuran; water for 24h; Suzuki cross-coupling; Inert atmosphere; Reflux; | 60% |
| With sodium phosphate; poly(N-vinyl-2-pyrrolidone); palladium In ethanol; water for 48h; Suzuki cross-coupling reaction; Heating; | 26% |
| With potassium fluoride In ethanol; water at 140℃; for 0.0333333h; Microwave irradiation; | 100 %Chromat. |
-
-
6165-68-0
thiophene boronic acid

-
-
586-77-6
4-bromo-N,N-dimethylaniline

-
-
88613-62-1
1-N,N-dimethylamino-4-(2-thienyl)benzene

| Conditions | Yield |
|---|---|
| With potassium fluoride; tris(dibenzylideneacetone)dipalladium (0); tri-tert-butyl phosphine In tetrahydrofuran at 20℃; for 2h; Suzuki cross-coupling; | 99% |
| With sodium carbonate; (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In ethanol at 55℃; Suzuki Coupling; | |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium carbonate In ethanol at 55℃; Suzuki coupling; |

| Conditions | Yield |
|---|---|
| With potassium carbonate; [PS-PEG-adppp-Pd(η3-C3H5)]Cl In water at 50℃; for 12h; Suzuki-Miyaura coupling; | 99% |
| With C18H9F5NOP(2-)*Pd(2+)*C3H7NO; caesium carbonate In N,N-dimethyl-formamide at 50℃; for 24h; Suzuki-Miyaura Coupling; Schlenk technique; Inert atmosphere; | 70% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; [PS-PEG-adppp-Pd(η3-C3H5)]Cl In water at 50℃; for 12h; Suzuki-Miyaura coupling; | 99% |
| With sodium carbonate In ethanol; water at 80℃; for 0.5h; Catalytic behavior; Suzuki-Miyaura Coupling; | 92% |
| With sodium carbonate; resin-plug bound Pd In N,N-dimethyl-formamide Suzuki coupling; | 75% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; [PS-PEG-adppp-Pd(η3-C3H5)]Cl In water at 50℃; for 12h; Suzuki-Miyaura coupling; | 99% |
| With potassium carbonate In ethanol; water at 70℃; for 1h; Catalytic behavior; Suzuki-Miyaura Coupling; | 93% |
| With potassium carbonate In water at 60℃; for 1h; Suzuki-Miyaura Coupling; Green chemistry; | 91% |
2-Thiopheneboronic acid Specification
The 2-Thiopheneboronic acid, with the CAS registry number 6165-68-0, is also known as Boronic acid, B-2-thienyl-. It belongs to the product categories of Boron Compounds; Azoles; Blocks; Boronic Acids; Boron, Nitrile, Thio & TM-Cpds; Heterocycles; Substituted Boronic Acids; Boronic Acids; Boronic Acid; Organoborons; Thiophene; B (Classes of Boron Compounds); Organic or Inorganic Borate. This chemical's molecular formula is C4H5BO2S and molecular weight is 127.96. What's more, its IUPAC name is called Thiophen-2-ylboronic acid. It should be stored in a cool, dry and well-ventilated place.
Physical properties about 2-Thiopheneboronic acid are: (1)ACD/LogP: 1.267; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.27; (4)ACD/LogD (pH 7.4): 1.23; (5)ACD/BCF (pH 5.5): 5.40; (6)ACD/BCF (pH 7.4): 4.93; (7)ACD/KOC (pH 5.5): 116.38; (8)ACD/KOC (pH 7.4): 106.24; (9)#H bond acceptors: 2; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 68.7 Å2; (13)Index of Refraction: 1.562; (14)Molar Refractivity: 31.43 cm3; (15)Molar Volume: 96.885 cm3; (16)Polarizability: 12.46×10-24cm3; (17)Surface Tension: 49.189 dyne/cm; (18)Density: 1.321 g/cm3; (19)Flash Point: 127.928 °C; (20)Enthalpy of Vaporization: 55.672 kJ/mol; (21)Boiling Point: 287.917 °C at 760 mmHg; (22)Vapour Pressure: 0.00100 mmHg at 25 °C.
Uses of 2-Thiopheneboronic acid: (1) it is used as synthetic intermediates; (2) it is used to produce other chemicals. For example, it can react with 2,6-dibromo-pyridine to get 2,6-di-thiophen-2-yl-pyridine. This reaction needs reagent tetrakis-(triphenylphosphine)palladium and solvent 1,2-dimethoxy-ethane. The reaction time is 4 hours. The yield is 55 %.
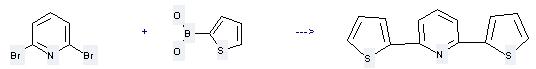
When you are dealing with this chemical, you should be very careful. This chemical may cause inflammation to the skin, eyes and respiratory system or other mucous membranes. Therefore, you should wear suitable protective clothing, gloves and eye/face protection. In case of contacting with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1) SMILES: B(c1cccs1)(O)O
(2) InChI: InChI=1S/C4H5BO2S/c6-5(7)4-2-1-3-8-4/h1-3,6-7H
(3) InChIKey: ARYHTUPFQTUBBG-UHFFFAOYSA-N
Related Products
- 2-Thiopheneboronic acid
- 6165-69-1
- 6165-75-9
- 6165-76-0
- 61657-67-8
- 61658-41-1
- 616-62-6
- 61665-08-5
- 61667-16-1
- 616-69-3
- 61670-79-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View