-
Name
Benzohydroxamic acid
- EINECS 207-797-6
- CAS No. 495-18-1
- Article Data175
- CAS DataBase
- Density 1.237 g/cm3
- Solubility 22 g/L (6 ºC)
- Melting Point 126-130 ºC
- Formula C7H7 N O2
- Boiling Point 327.1 °C at 760 mmHg
- Molecular Weight 137.138
- Flash Point 201 °C
- Transport Information
- Appearance brownish crystalline powder
- Safety Moderately toxic by ingestion. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx.
- Risk Codes R68
-
Molecular Structure
-
Hazard Symbols

- Synonyms Benzohydroxamicacid (6CI,8CI); BHAM; Benzenehydroxamic acid; Benzhydroxamic acid;Benzohydroximic acid; Benzoylhydroxylamine; Hydroxylamine, N-benzoyl-;N-Benzohydroxamic acid; N-Benzoylhydroxylamine; N-Hydroxybenzamide; NSC 147248;NSC 3136; Phenylhydroxamic acid
- PSA 49.33000
- LogP 1.19650
Synthetic route

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium hydrogencarbonate In water; ethyl acetate at 20℃; for 0.0833333h; | 99% |
| With hydroxylamine hydrochloride; sodium carbonate In diethyl ether; water at 0℃; for 1h; | 91.3% |
| With hydroxylamine hydrochloride; sodium carbonate In diethyl ether; water at 0℃; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: benzoic acid methyl ester With hydroxylamine hydrochloride; sodium hydroxide at 40 - 50℃; for 4h; Stage #2: With sulfuric acid at 10℃; | 98.3% |
| With hydroxylamine sulfate; sodium hydroxide In water for 2h; Reagent/catalyst; Solvent; Milling; | 96.15% |
| With hydroxylamine hydrochloride; potassium hydroxide In methanol at 20℃; for 12h; pH=10; | 90% |
-
-
13715-50-9
5,5-dimethyl-3-phenyl-<1,4,2>-diaoxazole

-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With Nafion-H In isopropyl alcohol for 30h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| With polymer-bound N-hydroxybenzenesulfonamide; sodium methylate In tetrahydrofuran; methanol at 20℃; for 7h; Angeli-Rimini's reaction; | 97% |
| With menadione; hydroxylamine hydrochloride; water; silver nitrate; triethylamine In tetrahydrofuran; ethanol at 20℃; for 0.5h; Angeli-Rimini Hydroxamic Synthesis; Irradiation; Green chemistry; | 85% |
| Multi-step reaction with 2 steps 1: [bis(acetoxy)iodo]benzene / acetonitrile / 1 h / 0 °C / Inert atmosphere 2: hydroxylamine / acetonitrile; water / 12 h / 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: N-chloro-succinimide; thio-xanthene-9-one / acetonitrile / 7 h / 20 °C / Irradiation 2: potassium carbonate; hydroxylamine hydrochloride / dichloromethane / 16 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; 1,3,5-trichloro-2,4,6-triazine; hydroxylamine hydrochloride; dmap In dichloromethane at 0 - 20℃; | 95% |
| Stage #1: benzoic acid With acetic anhydride for 0.0833333h; Stage #2: With hydroxylamine hydrochloride for 0.75h; Reagent/catalyst; | 95.81% |
| With N-Bromosuccinimide; hydroxylamine hydrochloride; triphenylphosphine In triethylamine; acetonitrile at 20℃; for 2h; Mitsunobu reaction; | 92% |

| Conditions | Yield |
|---|---|
| With methylamine In methanol for 2h; Irradiation; | 91% |
| With methylamine In methanol Irradiation; | 91% |
| With methylamine In methanol Quantum yield; Irradiation; | |
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; potassium hydroxide In methanol Reagent/catalyst; Concentration; | 91% |
| With potassium hydroxide; hydroxylamine hydrochloride In pyridine at 20℃; for 6h; Condensation; | 80% |
| With potassium hydroxide; hydroxylamine hydrochloride In methanol for 36h; Ambient temperature; | 77% |

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; triethylamine In tetrahydrofuran at 20℃; for 16h; | 91% |
-
-
155164-66-2
2-benzoyl-4,5-dichloropyridazin-3(2H)-one

-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With Amberlite(R) IRA-67 In acetonitrile at 20℃; for 0.833333h; | 90% |

-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; hydroxylamine In methanol at 20℃; for 12h; pH=10; | 90% |
-
-
144751-74-6
tert-butyl benzoyl((tert-butyldimethylsilyl)oxy)carbamate

-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With cesium fluoride; trifluoroacetic acid In dichloromethane at 0℃; for 6h; | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: benzyl alcohol With 1-hydroxy-pyrrolidine-2,5-dione; [bis(acetoxy)iodo]benzene In acetonitrile at 0 - 20℃; for 3h; Inert atmosphere; Green chemistry; Stage #2: With hydroxylamine In water; acetonitrile at 20℃; for 12h; Reagent/catalyst; Inert atmosphere; Green chemistry; | 82% |

-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 0℃; for 18h; | 80.6% |
-
-
7419-56-9
N-hydroxy-N-(1,1-dimethylethyl)benzamide

-
A
-
623-81-4
diethyl sulphite

-
B
-
495-18-1
Benzohydroxamic acid

-
C
-
93-89-0
benzoic acid ethyl ester

-
D
-
698-16-8
benzohydroximoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; ethanol In tetrachloromethane Product distribution; other solvent; | A n/a B 8% C 4% D 78% |

-
-
7419-56-9
N-hydroxy-N-(1,1-dimethylethyl)benzamide

-
A
-
495-18-1
Benzohydroxamic acid

-
B
-
93-89-0
benzoic acid ethyl ester

-
C
-
698-16-8
benzohydroximoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; ethanol 1.) CCl4, -30 deg C, 5 h; 2.) room temp., 2 h; Yield given. Multistep reaction; | A n/a B 4% C 78% |
| With thionyl chloride; ethanol 1.) CCl4, -30 deg C, 5 h; 2.) room temp., 2 h; Yield given. Multistep reaction; | A 8% B 4% C n/a |

-
-
93-89-0
benzoic acid ethyl ester

-
-
5470-11-1
hydroxylamine hydrochloride

-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| Stage #1: hydroxylamine hydrochloride With sodium hydroxide In water for 0.25h; Stage #2: benzoic acid ethyl ester With hydrogenchloride In ethanol at 40℃; for 72h; pH=2; | 78% |

| Conditions | Yield |
|---|---|
| With [hydroxy(tosyloxy)iodo]benzene In chloroform at 20 - 60℃; for 2.16667h; | 76% |
| With [hydroxy(tosyloxy)iodo]benzene In dimethyl sulfoxide at 20 - 80℃; for 1.5h; |
-
-
32685-16-8
potassium benzohydroxamate

-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With acetic acid Heating; | 75% |
-
-
139259-94-2
N-(acetyloxy)-N-(((4-methylphenyl)methyl)oxy)benzamide

-
A
-
589-18-4
4-Methylbenzyl alcohol

-
B
-
495-18-1
Benzohydroxamic acid

-
C
-
38418-10-9
4-methylbenzyl benzoate

-
D
-
100-52-7
benzaldehyde

-
E
-
93-97-0
benzoic acid anhydride

-
F
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; water-d2 In [D3]acetonitrile at 34.9℃; Rate constant; var. conc. of sulfuric acid; | A 54% B 1% C 18% D 4% E 6% F 68% |

-
-
36663-91-9
2-nitrobenzyl benzohydroxamate

-
A
-
495-18-1
Benzohydroxamic acid

-
B
-
55-21-0
benzamide

-
C
-
552-89-6
2-nitro-benzaldehyde

| Conditions | Yield |
|---|---|
| In water; acetonitrile Photolysis; | A 45% B 37% C 40% |

-
-
6723-30-4
O-2-tetrahydro-2H-pyranhydroxylamine

-
-
959-22-8
p-nitrophenylbenzoate

-
A
-
6584-61-8
O-(tetrahydro-2-pyranyl)benzohydroxamic acid

-
B
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With acetic acid In toluene at 115℃; under 4500.45 Torr; for 0.216667h; Microwave irradiation; | A 41% B n/a |

-
-
153136-98-2
4-acetyl-1-benzoyl-3-methyl-1H-pyrazole

-
A
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium carbonate In ethanol; water at 40℃; for 1h; | A 28% B 22% C 37% |

-
-
3532-25-0
N-benzyloxy benzamide

-
A
-
93-98-1
N-phenyl benzoyl amide

-
B
-
495-18-1
Benzohydroxamic acid

-
C
-
100-52-7
benzaldehyde

-
D
-
103-29-7
1,1'-(1,2-ethanediyl)bisbenzene

-
E
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| In acetone at 25℃; for 20h; Product distribution; Mechanism; Irradiation; | A 25% B 10% C 15% D 10% E 15% |

-
-
139259-98-6
p-nitrobenzyl N-acetoxybenzogydroxamate

-
A
-
619-73-8
4-nitrobenzyl chloride

-
B
-
495-18-1
Benzohydroxamic acid

-
C
-
4457-41-4
4-nitrobenzyl benzoate

-
D
-
100-52-7
benzaldehyde

-
E
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; water-d2 In [D3]acetonitrile at 34.9℃; Rate constant; var. conc. of sulfuric acid; | A 22% B n/a C 25% D 16% E 23% |


| Conditions | Yield |
|---|---|
| With alkali |
-
-
56-23-5
tetrachloromethane

-
-
959-32-0
N-(benzoyloxy)benzamide

-
A
-
495-18-1
Benzohydroxamic acid

-
B
-
65-85-0
benzoic acid

-
-
13591-25-8, 13591-32-7, 41401-05-2, 41401-06-3
dibenzylidene-diazoxane-N-oxide

-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With caro's acid |

| Conditions | Yield |
|---|---|
| With methanol; hydroxylamine at 40 - 50℃; |

| Conditions | Yield |
|---|---|
| With ethanol; hydroxylamine |
-
-
495-18-1
Benzohydroxamic acid

-
-
77219-88-6
bis(trimethylsilyl)benzohydroxamic acid

-
-
78313-15-2
mono(trimethylsilyl)benzohydroxamic acid

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 100% |
-
-
495-18-1
Benzohydroxamic acid

-
-
77219-88-6
bis(trimethylsilyl)benzohydroxamic acid

-
-
102-07-8
bis(diphenyl)urea

| Conditions | Yield |
|---|---|
| at 100℃; for 1.5h; | 100% |
-
-
495-18-1
Benzohydroxamic acid

| Conditions | Yield |
|---|---|
| With sodium periodate In dichloromethane; water; N,N-dimethyl-formamide for 2h; | 100% |

| Conditions | Yield |
|---|---|
| With tetrabutylammonium periodite In chloroform; N,N-dimethyl-formamide at 20℃; for 3h; | 100% |
-
-
33707-36-7
1-(methoxycarbonyl)-1,2-dihydropyridine

-
-
495-18-1
Benzohydroxamic acid

-
-
94936-52-4, 96212-66-7
methyl 3-benzoyl-2-oxa-3,5-diazabicyclo<2.2.2>oct-7-ene-5-carboxylate

| Conditions | Yield |
|---|---|
| With tetrapropylammonium periodate In dichloromethane at 0℃; for 1h; | 99% |
-
-
876938-53-3
Cyclohepta-1,3-diene

-
-
495-18-1
Benzohydroxamic acid

-
-
95687-88-0
N-benzoyl-8-oxa-9-azabicyclo<3.2.2>non-6-ene

| Conditions | Yield |
|---|---|
| With tetramethylammonium metaperiodate In chloroform; N,N-dimethyl-formamide | 99% |
| With tetrapropylammonium periodate In chloroform; N,N-dimethyl-formamide for 3.5h; Ambient temperature; | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: Benzohydroxamic acid With 4-methyl-morpholine; 1,3,5-trichloro-2,4,6-triazine In dichloroethane at 0℃; for 1.5h; Lossen rearrangement; Inert atmosphere; Stage #2: aniline In dichloroethane at 0 - 84℃; for 15h; Lossen rearrangement; Inert atmosphere; | 99% |
| Stage #1: Benzohydroxamic acid With ethyl 2-cyano-2-(4-nitrophenylsulfonyloxyimino)acetate; N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0℃; for 1.5h; Lossen Rearrangement; Stage #2: aniline In tetrahydrofuran at 20℃; for 4h; Lossen Rearrangement; | 83% |
| Stage #1: Benzohydroxamic acid With ethyl 2-(tert-butoxycarbonyloxyimino)-2-cyanoacetate; N-ethyl-N,N-diisopropylamine In dichloromethane at 0℃; for 0.25h; Green chemistry; Stage #2: aniline In dichloromethane for 6h; Lossen Rearrangement; Green chemistry; chemoselective reaction; | 82% |
| Stage #1: Benzohydroxamic acid With 2-chloro-1,3-dimethylimidazolinium chloride; triethylamine In dichloromethane at 20℃; for 15h; Rearrangement; Stage #2: aniline In dichloromethane at 20℃; for 4h; Addition; | 66% |

| Conditions | Yield |
|---|---|
| With sodium acetate In water at 20℃; for 2h; Reagent/catalyst; Time; Temperature; Solvent; | 99% |

| Conditions | Yield |
|---|---|
| With sodium acetate In water at 20℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| In water addn. of ligand in warm water to CuSO4*5H2O; (pptn.), standing for 6 h; filtration, washing (warm water), drying (vac., over P2O5); elem. anal.; | 98.5% |
-
-
495-18-1
Benzohydroxamic acid

-
-
1165952-91-9
cyclohexa-1,3-diene

-
-
70156-86-4, 96093-86-6, 146387-03-3
3-benzoyl-2,3-oxazabicyclo<2.2.2>oct-5-ene

| Conditions | Yield |
|---|---|
| With bis-[(trifluoroacetoxy)iodo]benzene In dichloromethane at 20℃; for 1h; Reagent/catalyst; Solvent; Diels-Alder Cycloaddition; Inert atmosphere; | 98% |
| With tert.-butylhydroperoxide; tetra-(n-butyl)ammonium iodide In dichloromethane at 20℃; for 18h; Catalytic behavior; Reagent/catalyst; Solvent; Diels-Alder Cycloaddition; Inert atmosphere; Green chemistry; | 98% |
| With chlorobis(cyclooctene)-iridium(I) dimer; dihydrogen peroxide In tetrahydrofuran; water at 0 - 20℃; for 20h; | 90% |
-
-
495-18-1
Benzohydroxamic acid

-
-
123-73-9
crotonaldehyde

-
-
114056-77-8
2-Benzoyl-3-methyl-5-hydroxyisoxazolidine

| Conditions | Yield |
|---|---|
| With TEAE-cellulose In methanol at 20℃; for 16h; | 98% |
| With diethylaminoethylcellulose at 0 - 20℃; | 98% |
| With diethylaminoethyl sephacel In dichloromethane; water for 2h; | 65% |
Benzohydroxamic acid Chemical Properties
IUPAC Name: N-Hydroxybenzamide
Synonyms: Benzamide, N-hydroxy- ; Benzohydroxamic acid ; Benzohydroxamic acid ; Benzohydroxamic acid ; (R-(R*,S*))-Alpha-Ethyl-2-piperidinemethanol ; 2-Piperidinemethanol, alpha-ethyl-, (R-(R*,S*))- (9CI)
Product Categories: AMIDE;Hydroxylamines;Hydroxylamines (N-Substituted);Inhibitors
Molecular Structure of Benzohydroxamic acid (CAS NO.495-18-1) : 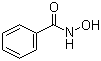
Molecular Formula of Benzohydroxamic acid (CAS NO.495-18-1) : C7H7NO2
Molecular Weight of Benzohydroxamic acid (CAS NO.495-18-1) : 137.14
CAS NO: 495-18-1
EINECS : 207-797-6
BRN : 1907585
Mol File: 495-18-1.mol
Index of Refraction: 1.578
Surface Tension: 51.9 dyne/cm
Density: 1.237 g/cm3
Flash Point: 201 °C
Enthalpy of Vaporization: 60.11 kJ/mol
Boiling Point: 327.1 °C at 760 mmHg
Vapour Pressure: 8.36E-05 mmHg at 25°C
Melting point: 126-130 ºC
Water solubility : 22 g/L (6 ºC
Storage temp: 0-6°C
Appearance:Rhombic crystals or light beige solid.
Solubility: soluble in alcohol; slightly soluble in ether, insoluble in benzene.
Benzohydroxamic acid Uses
Benzohydroxamic acid (CAS NO.495-18-1) is used as determination reagents of iron, vanadium and uranium .
Benzohydroxamic acid Toxicity Data With Reference
| 1. | mmo-sat 2500 nmol/plate | MUREAV Mutation Research. 135 (1984),139. | ||
| 2. | mma-sat 1 µmol/plate | MUREAV Mutation Research. 56 (1977),7. | ||
| 3. | orl-rat LD:>500 mg/kg | NCNSA6 National Academy of Sciences, National Research Council, Chemical-Biological Coordination Center, Review. 5 (1953),26. |
Benzohydroxamic acid Consensus Reports
Reported in EPA TSCA Inventory. EPA Genetic Toxicology Program.
Benzohydroxamic acid Safety Profile
Moderately toxic by ingestion. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx.
Hazard Codes  Xn
Xn
Risk Statements 68-40-20/21/22
R68:Possible risk of irreversible effects.
R40:Limited evidence of a carcinogenic effect.
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
Safety Statements 45-36/37-22
S22:Do not breathe dust.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S36/37:Wear suitable protective clothing and gloves.
WGK Germany 3
RTECS DF9650000
HS Code 29280090
Benzohydroxamic acid Specification
1.Reactivity Profile :Benzohydroxamic acid is an amide. Amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx).
2.Health Hazard :acute/chronic hazards : When heated to decomposition Benzohydroxamic acid emits toxic fumes of nitrogen oxides.
3.Fire Hazard :Flash point data for Benzohydroxamic acid are not available; however, Benzohydroxamic acid is probably combustible.
Related Products
- Benzohydroxamic acid
- 4952-68-5
- 495-31-8
- 495382-48-4
- 49538-43-4
- 49539-88-0
- 49540-00-3
- 495407-02-8
- 495-40-9
- 495-41-0
- 4954-14-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View