-
Name
Cyclohexanecarbonitrile
- EINECS 212-157-4
- CAS No. 766-05-2
- Article Data142
- CAS DataBase
- Density 0.91 g/cm3
- Solubility
- Melting Point 11 °C(lit.)
- Formula C7H11N
- Boiling Point 199.8 °C at 760 mmHg
- Molecular Weight 109.171
- Flash Point 68.8 °C
- Transport Information UN 3276 6.1/PG 3
- Appearance
- Safety 36/37-45-36/37/39-26-23
- Risk Codes 24/25-36/37/38-23/24/25
-
Molecular Structure
-
Hazard Symbols
 T
T
- Synonyms Cyanocyclohexane;Cyclohexanecarboxylic acid nitrile;Cyclohexanenitrile;Cyclohexyl cyanide;Hexahydrobenzonitrile;NSC 17557;
- PSA 23.79000
- LogP 2.09028
Synthetic route

| Conditions | Yield |
|---|---|
| With nickel(II) chloride dihydrate In acetonitrile at 80℃; Molecular sieve; Inert atmosphere; | 100% |
| With MIL-100 (Fe)-NH4F In o-xylene at 153 - 160℃; for 1h; Dean-Stark; Inert atmosphere; | 97% |
| With phosphoric acid diethyl ester 2-phenylbenzimidazol-1-yl ester; triethylamine In acetonitrile at 20℃; for 0.916667h; | 94% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide In acetonitrile at 20℃; for 4h; Reagent/catalyst; Inert atmosphere; UV-irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With pyridine; Oxone; 4-acetylamino-2,2,6,6-tetramethyl-1-piperidinoxy; Pyridine hydrobromide In dichloromethane at 20℃; for 12h; Green chemistry; | 97% |
| With pyridine; 4-acetylamino-2,2,6,6-tetramethylpiperidine-1-oxoammonium tetrafluoroborate In dichloromethane at 20℃; for 12h; Inert atmosphere; | 95% |
| With water; potassium hydroxide at 25℃; pH=13.6; Electrochemical reaction; | 95% |

| Conditions | Yield |
|---|---|
| With sulfonamide In sulfolane at 120℃; for 3h; | 95% |
-
-
19888-79-0
1-cyclohexanecarboxaldehyde N,N-dimethylhydrazone

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With pyridine; dihydrogen peroxide; acetic acid; methyltrioxorhenium(VII) In water; acetonitrile for 0.25h; | 95% |
| Multi-step reaction with 2 steps 1: benzene / 7 h / Heating 2: NaOMe / methanol / 4 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| With ammonia at 210℃; Inert atmosphere; | 95% |
| With nitrogen; ammonia at 240℃; Heating; | 88% |
| With ammonium hydroxide; dihydrogen peroxide In water; acetonitrile at 50℃; for 8h; | 76% |

| Conditions | Yield |
|---|---|
| With ammonia; iodine In tetrahydrofuran; water at 20℃; for 0.166667h; | 94% |
| Stage #1: cyclohexanecarbaldehyde With 3 A molecular sieve; hydroxylamine hydrochloride; 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane at 20℃; for 10h; Stage #2: With ethyl phosphodichloridite In dichloromethane at 20℃; for 5h; Further stages.; | 92% |
| With bismuth(lll) trifluoromethanesulfonate; acetylhydroxamic acid In acetonitrile for 24h; Reflux; | 88% |

| Conditions | Yield |
|---|---|
| With trichloromethyl chloroformate In various solvent(s) 0-5 deg C then heated to 60 deg C, 5 min; | 93% |
| With lead acetate In dichloromethane for 12h; Reflux; | 90% |
| With oxalyl dichloride; triethylamine In dimethyl sulfoxide; acetonitrile at 20℃; for 0.666667h; Swern Oxidation; | 87% |
-
-
61540-43-0
Dimethyl-dithiocarbamic acid 1-cyano-cyclohexyl ester

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride In benzene at 80℃; for 5h; | 93% |
-
-
24225-08-9
methanesulfonyl cyanide

-
-
105398-69-4
Cyclohexanecarboxylic acid 2-thioxo-2H-pyridin-1-yl ester

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| In dichloromethane at 0℃; Irradiation; | 90% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -78℃; for 0.5h; cyanation; | 90% |
-
-
30950-35-7
(E)-N-(cyclohexylmethylidene)hydroxylamine

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; 4 A molecular sieve In acetonitrile at 80℃; for 0.166667h; | 90% |
-
-
1569-69-3
Cyclohexanethiol

-
-
10442-39-4
tetra-n-butylammonium cyanide

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With triphenylphosphine; 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 20℃; for 5h; | 90% |
-
-
709-83-1
2-(cyclohexyloxy)tetrahydro-2H-pyran

-
-
10442-39-4
tetra-n-butylammonium cyanide

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With triphenylphosphine; 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile for 7h; Heating; | 90% |
-
-
110-82-7
cyclohexane

-
-
172876-96-9
3-oxo-1λ3-benzo[d][1,2]iodaoxole-1(3H)-carbonitrile

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With tert-Butyl peroxybenzoate In ethyl acetate at 110℃; for 16h; Solvent; Temperature; Reagent/catalyst; Time; Sealed tube; Inert atmosphere; Glovebox; | 90% |
-
-
19158-51-1
P-toluenesulfonyl cyanide

-
-
105398-69-4
Cyclohexanecarboxylic acid 2-thioxo-2H-pyridin-1-yl ester

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| In dichloromethane at 0℃; Irradiation; | 88% |

| Conditions | Yield |
|---|---|
| With thionyl chloride; sulfonamide In sulfolane at 120℃; for 3h; | 88% |
| With sulfuric acid; acetonitrile at 95℃; for 4h; | 59% |
| Multi-step reaction with 2 steps 1.1: thionyl chloride / tetrahydrofuran / 1 h / 50 °C 1.2: 0.08 h / 0 °C 2.1: palladium diacetate; Selectfluor; acetonitrile / 18 h / 20 °C View Scheme | |
| With indium(III) chloride; acetonitrile at 200℃; under 11251.1 Torr; for 14h; | 86 %Chromat. |
-
-
61827-29-0
1-(1-Cyanocyclohexyl)-2-carbomethoxyhydrazine

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With sodium cyanide In methanol at 17℃; for 4.7h; electrolysis; | 86% |
-
-
3218-02-8
cyclohexylmethylamine

-
A
-
766-05-2
cyclohexane carbonitrile

-
B
-
1122-56-1
cyclohexylcarboxamide

-
C
-
98-89-5
Cyclohexanecarboxylic acid

| Conditions | Yield |
|---|---|
| With water; oxygen at 140℃; under 3800.26 Torr; for 24h; | A n/a B 86% C n/a |

-
-
7677-24-9
trimethylsilyl cyanide

-
-
2719-27-9
cyclohexanylcarbonyl chloride

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); triphenylphosphine In toluene at 110℃; for 1h; Schlenk technique; Inert atmosphere; | 86% |
-
-
19158-51-1
P-toluenesulfonyl cyanide

-
-
121883-35-0
cyclohexylzinc iodide

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 25℃; | 84% |
-
-
27702-91-6
1-(1-Cyanocyclohexyl)-2-acetylhydrazine

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With sodium cyanide In methanol at 17℃; for 4.7h; electrolysis; | 83% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine; 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 20℃; for 0.166667h; | 83% |
-
-
4362-48-5
2-cyclohexyl-1,3-dioxolane

-
A
-
4715-11-1
cyclohexanecarbaldoxime

-
B
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; toluene-4-sulfonic acid In ethanol for 2h; Heating; | A 8% B 81% |
-
-
102804-62-6
Allyl 1-cyanocyclohexanecarboxylate

-
A
-
766-05-2
cyclohexane carbonitrile

-
B
-
1855-63-6
cyclohex-1-enecarbonitrile

| Conditions | Yield |
|---|---|
| tris(dibenzylideneacetone)dipalladium(0) chloroform complex; triphenylphosphine In various solvent(s) for 1h; Heating; Yields of byproduct given; | A n/a B 81% |
-
-
175351-40-3
pyridin-2-yl cyanate

-
-
931-50-0
cyclohexylmagnesium bromide

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 0.166667h; | 80% |
-
-
10424-96-1
N'-[1-Cyclohexyl-meth-(Z)-ylidene]-N,N-dimethyl-hydrazine

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium carbonate; acetonitrile at 55℃; for 18h; | 80% |
-
-
67-56-1
methanol

-
-
201230-82-2
carbon monoxide

-
-
19158-51-1
P-toluenesulfonyl cyanide

-
A
-
4630-82-4
methyl cyclohexylcarboxylate

-
B
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: carbon monoxide; P-toluenesulfonyl cyanide; (Prop-2-ene-1-sulfonyl)-cyclohexane With 1,1'-azobis(1-cyanocyclohexanenitrile) In n-heptane at 100℃; under 98800 Torr; Stage #2: methanol In n-heptane at 20℃; | A 80% B 15% |

-
-
50450-21-0
dimethylbromosulphonium bromide

-
-
1122-56-1
cyclohexylcarboxamide

-
A
-
766-05-2
cyclohexane carbonitrile

-
B
-
67-68-5
dimethyl sulfoxide

| Conditions | Yield |
|---|---|
| In acetonitrile for 3h; Reflux; | A 80% B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With n-butyllithium; N-ethyl-N,N-diisopropylamine; lithium diisopropyl amide In tetrahydrofuran; hexane at -15 - 8℃; Stage #2: 2-ethyl-1-bromobutane In tetrahydrofuran; hexane at 3 - 26℃; Product distribution / selectivity; | 100% |
| Stage #1: cyclohexane carbonitrile With methylmagnesium chloride; diethylamine In tetrahydrofuran at 20 - 25℃; for 1.5h; Stage #2: 2-ethyl-1-bromobutane In tetrahydrofuran at 45 - 50℃; for 2h; Product distribution / selectivity; | 97.6% |
| With methylmagnesium chloride; diethylamine In tetrahydrofuran at 45.3 - 70.2℃; for 2.25h; Product distribution / selectivity; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: allyl bromide In tetrahydrofuran at -78 - 20℃; Inert atmosphere; | 100% |
| Stage #1: cyclohexane carbonitrile With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.5h; Stage #2: allyl bromide In tetrahydrofuran at 20℃; | 94% |
| Stage #1: cyclohexane carbonitrile With lithium diisopropyl amide In tetrahydrofuran Inert atmosphere; Stage #2: allyl bromide Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; hydrogen; palladium on activated charcoal In methanol at 20℃; for 24h; | 100% |
| With palladium 10% on activated carbon; hydrogen In methanol at 20℃; under 760.051 Torr; for 24h; | 99% |
-
-
64-17-5
ethanol

-
-
766-05-2
cyclohexane carbonitrile

-
-
43002-69-3
ethyl cyclohexanecarboximidate hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 0 - 20℃; for 12h; Inert atmosphere; | 100% |
| With hydrogenchloride In 1,4-dioxane at 20℃; for 48h; | 74% |
| With hydrogenchloride for 3h; Inert atmosphere; Cooling with ice; | 73% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether at 0℃; for 0.333333h; | 100% |
| With hydrogenchloride at 0℃; for 0.333333h; |

| Conditions | Yield |
|---|---|
| With [RuH(tBu-PNP(-))(CO)]; water In tert-butyl alcohol at 50℃; for 24h; | 99% |
| With water; tricyclohexylphosphine; {Rh(OMe)(cod)}2 In isopropyl alcohol at 25℃; for 72h; | 96% |
| With N-ethyl-N-hydroxy-ethanamine; water; copper diacetate In ethanol at 35℃; for 3h; | 89% |

| Conditions | Yield |
|---|---|
| With [Ru(H)(BH4)(CO)(PPh3)(3-(di-tert-butylphosphino)-N-((1-methyl-1H-imidazol-2 yl)methyl)propylamine)]; hydrogen In isopropyl alcohol at 70℃; for 3h; Inert atmosphere; Autoclave; | 99% |
| With ammonia; hydrogen In toluene at 120℃; under 22502.3 Torr; for 16h; Autoclave; | 92% |
| With hydrogen In ethanol at 88℃; under 37503.8 Torr; for 0.5h; | 91% |
-
-
100-39-0
benzyl bromide

-
-
766-05-2
cyclohexane carbonitrile

-
-
104367-54-6
1-benzylcyclohexanecarbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: benzyl bromide In tetrahydrofuran; hexane at -78 - 20℃; | 99% |
| Stage #1: cyclohexane carbonitrile With lithium diisopropyl amide In tetrahydrofuran at -78 - 20℃; for 1h; Stage #2: benzyl bromide With methyl cyanoformate In tetrahydrofuran at -78 - 20℃; for 2h; chemoselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.333333h; Stage #2: Phenyl azide With N,N,N,N,N,N-hexamethylphosphoric triamide In tetrahydrofuran at -78 - 20℃; for 1h; Stage #3: dimethyl sulfate at 20℃; for 1h; | 99% |


| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.333333h; Stage #2: Phenyl azide With N,N,N,N,N,N-hexamethylphosphoric triamide In tetrahydrofuran at -78 - 20℃; for 1h; Stage #3: dimethyl sulfate at -78℃; for 1h; | 99% |


| Conditions | Yield |
|---|---|
| With potassium hexamethylsilazane In toluene at 60℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: iodobenzene; cyclohexane carbonitrile With iodine; magnesium In tetrahydrofuran at 83 - 85℃; Stage #2: With sulfuric acid In water at 20 - 25℃; for 2h; | 99% |
-
-
766-05-2
cyclohexane carbonitrile

-
-
95-53-4
o-toluidine

-
-
205990-43-8
2-cyclohexylcarbonyl-6-methylaniline

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile; o-toluidine With boron trichloride In toluene at 20℃; for 1h; Addition; Stage #2: With aluminium trichloride In acetonitrile for 5h; Rearrangement; Heating; Stage #3: With hydrogenchloride In acetonitrile for 2.5h; Hydrolysis; Heating; | 98.4% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride | 98% |
| With aluminum (III) chloride at 120℃; |

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With hydrogenchloride; methanol In diethyl ether at 0℃; Stage #2: With ammonia In ethanol | 98% |
| Multi-step reaction with 2 steps 1: 31 g / HCl (gas) / diethyl ether; methanol / 0 °C 2: 9 g / NH3 / ethanol / 24 h / 0 °C View Scheme | |
| Stage #1: cyclohexane carbonitrile With hydrogenchloride; ethanol for 3h; Pinner Amidine Synthesis; Inert atmosphere; Cooling with ice; Stage #2: With ammonia In methanol at 20℃; for 24h; Pinner Amidine Synthesis; Inert atmosphere; | 835 mg |
-
-
766-05-2
cyclohexane carbonitrile

-
-
75-65-0
tert-butyl alcohol

-
-
6941-24-8
cyclohexanecarboxylic acid N-t-butylamide

| Conditions | Yield |
|---|---|
| With 1-methyl-3-(4-sulfonylbutyl)-1H-imidazol-3-ium trifluoromethanesulfonate at 60℃; for 5h; Ritter reaction; Inert atmosphere; Ionic liquid; | 98% |
| With silica boron-sulfuric acid nanoparticles at 20℃; for 0.333333h; Ritter reaction; Neat (no solvent); | 91% |

| Conditions | Yield |
|---|---|
| With sodium triethylborohydride In neat (no solvent) at 20℃; for 3h; Inert atmosphere; Glovebox; Green chemistry; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: bromobenzene; cyclohexane carbonitrile With iodine; magnesium In tetrahydrofuran at 80 - 85℃; Stage #2: With sulfuric acid In water at 20 - 25℃; for 2h; | 98% |

| Conditions | Yield |
|---|---|
| With gallium(III) trichloride; 2,4-lutidine In 2-methyltetrahydrofuran; isopropyl alcohol at 85℃; for 4h; Reagent/catalyst; Solvent; Inert atmosphere; | 97.6% |

-
-
27570-08-7, 60592-52-1, 6303-59-9
4-(2-bromo-vinyl)-anisole

-
-
766-05-2
cyclohexane carbonitrile

-
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| With gallium(III) trichloride; 2,4-lutidine In 2-methyltetrahydrofuran; isopropyl alcohol at 70℃; for 6h; Reagent/catalyst; Solvent; Inert atmosphere; | 97.5% |

-
-
66482-29-9, 66482-30-2, 125428-11-7
1-(2-bromovinyl)-4-chlorobenzene

-
-
766-05-2
cyclohexane carbonitrile

-
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| With gallium(III) trichloride; 2,4-lutidine In 2-methyltetrahydrofuran; isopropyl alcohol at 80℃; for 5h; Reagent/catalyst; Solvent; Inert atmosphere; | 97.3% |

-
-
1372806-96-6
(E)-(5-bromopent-3-en-2-yloxy)tertbutyldimethylsilane

-
-
766-05-2
cyclohexane carbonitrile

-
-
1372806-98-8
C18H33NOSi

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: (E)-(5-bromopent-3-en-2-yloxy)tertbutyldimethylsilane In tetrahydrofuran; hexane at -78 - 25℃; | 97% |
-
-
540-88-5
acetic acid tert-butyl ester

-
-
766-05-2
cyclohexane carbonitrile

-
-
6941-24-8
cyclohexanecarboxylic acid N-t-butylamide

| Conditions | Yield |
|---|---|
| With silica sulfuric acid at 60℃; for 3.08333h; Reagent/catalyst; Temperature; Ritter Amidation; Green chemistry; | 97% |
-
-
38771-21-0
4-bromobut-1-yne

-
-
766-05-2
cyclohexane carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 4-bromobut-1-yne In tetrahydrofuran; hexane at -78 - 24℃; for 14h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile; chlorobenzene With iodine; magnesium In tetrahydrofuran at 85℃; Stage #2: With sulfuric acid In water at 20 - 25℃; for 2h; | 97% |
-
-
67-56-1
methanol

-
-
766-05-2
cyclohexane carbonitrile

-
-
94052-40-1
methyl cyclohexanecarboximidate hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In hexane at 0℃; for 3h; | 96% |
| With hydrogenchloride; diethyl ether | |
| With hydrogenchloride In methanol; diethyl ether at 0℃; | 31 g |
-
-
40400-13-3
2-Iodobenzyl bromide

-
-
766-05-2
cyclohexane carbonitrile

-
-
445006-90-6
1-(2-iodobenzyl)cyclohexanecarbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexane carbonitrile With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: 2-Iodobenzyl bromide In tetrahydrofuran; hexane at -78 - 20℃; Further stages.; | 96% |
Cyclohexanecarbonitrile Specification
The Cyclohexanecarbonitrile with CAS registry number of 766-05-2 is also known as Cyclohexanecarboxylic acid nitrile. The IUPAC name and product name are tha same. It belongs to product categories of C6 to C7; Cyanides/Nitriles; Nitrogen Compounds. Its EINECS registry number is 212-157-4. In addition, the formula is C7H11N and the molecular weight is 109.17. This chemical at low levels causes damage to health that should be sealed in ventilated and dry place without light at room temperature.
Physical properties about Cyclohexanecarbonitrile are: (1)ACD/LogP: 1.61; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.61; (4)ACD/LogD (pH 7.4): 1.61; (5)ACD/BCF (pH 5.5): 9.9; (6)ACD/BCF (pH 7.4): 9.9; (7)ACD/KOC (pH 5.5): 179.53; (8)ACD/KOC (pH 7.4): 179.53; (9)#H bond acceptors: 1; (10)Index of Refraction: 1.451; (11)Molar Refractivity: 32.32 cm3; (12)Molar Volume: 119.9 cm3; (13)Surface Tension: 33.2 dyne/cm; (14)Density: 0.91 g/cm3; (15)Flash Point: 68.8 °C; (16)Enthalpy of Vaporization: 43.6 kJ/mol; (17)Boiling Point: 199.8 °C at 760 mmHg; (18)Vapour Pressure: 0.335 mmHg at 25 °C.
Preparation of Cyclohexanecarbonitrile: it is prepared by reaction of cyclohexanecarboxylic acid amide. The reaction needs reagents bis(trichloromethyl) carbonate, triethylamine and solvent CHCl3 at the temperature of 50 °C for 2 hours. The yield is about 76%.
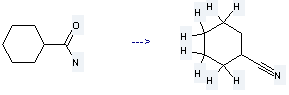
Uses of Cyclohexanecarbonitrile: it is used to produce 1-(6-methyl-4-oxo-2-phenyl-3,4-dihydro-2H-[1,3]oxazin-2-yl)-cyclohexanecarbonitrile by reaction with 6-methyl-2-phenyl-[1,3]oxazin-4-one. The reaction occurs with reagents BuLi, N-isopropyl-N-cyclohexylamine and solvent tetrahydrofuran at the temperature of -70 °C for 2 hours. The yield is about 83%.
![Cyclohexanecarbonitrile is used to produce 1-(6-methyl-4-oxo-2-phenyl-3,4-dihydro-2H-[1,3]oxazin-2-yl)-cyclohexanecarbonitrile by reaction with 6-methyl-2-phenyl-[1,3]oxazin-4-one.](/UserFilesUpload/Uses of Cyclohexanecarbonitrile.png)
When you are using this chemical, please be cautious about it. As a chemical, it is irritating to eyes, respiratory system and skin. Besides, it is toxic by inhalation, in contact with skin and if swallowed. During using it, wear suitable protective clothing, gloves and eye/face protection. Do not breathe gas/fumes/vapour/spray. If contact with eyes accidently, rinse immediately with plenty of water and seek medical advice. In case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: C1CCC(CC1)C#N
2. InChI: InChI=1S/C7H11N/c8-6-7-4-2-1-3-5-7/h7H,1-5H2
3. InChIKey: VBWIZSYFQSOUFQ-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 100mg/kg (100mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Zentralblatt fuer Arbeitsmedizin und Arbeitsschutz. Vol. 19, Pg. 225, 1969. |
| rabbit | LDLo | skin | 200mg/kg (200mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Zentralblatt fuer Arbeitsmedizin und Arbeitsschutz. Vol. 19, Pg. 225, 1969. |
| rat | LD50 | oral | 80mg/kg (80mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Zentralblatt fuer Arbeitsmedizin und Arbeitsschutz. Vol. 19, Pg. 225, 1969. |
Related Products
- Cyclohexanecarbonitrile
- Cyclohexanecarbonitrile, 3-(acetyloxy)-3-methyl-6-(1-methylethyl)-2-(3-oxobutyl)-,[1S-(1a,2a,3a,6b)]- (9CI)
- Cyclohexanecarbonitrile, 4-oxo-
- Cyclohexanecarbonitrile, 5-methyl-2-(1-methylethyl)- (9CI)
- Cyclohexanecarbonitrile,1-(3-fluorophenyl)-
- Cyclohexanecarbonitrile,1-(4-fluorophenyl)-
- Cyclohexanecarbonitrile,1-(4-methoxyphenyl)-
- Cyclohexanecarbonitrile,2-oxo-
- 76-60-8
- 766-08-5
- 76608-88-3
- 76609-47-7
- 766-09-6
- 76609-88-6
- 76610-84-9
- 766-11-0
- 7661-32-7
- 76613-71-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View