-
Name
Oxaliplatin
- EINECS 274-613-9
- CAS No. 61825-94-3
- Article Data22
- CAS DataBase
- Density
- Solubility Soluble in water with heating and/or sonication
- Melting Point
- Formula C8H14N2O4Pt
- Boiling Point 193.6oC at 760 mmHg
- Molecular Weight 397.29
- Flash Point 75oC
- Transport Information UN 2811
- Appearance White crystalline solid
- Safety 26-36
- Risk Codes 36/37/38-40-42/43
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms Eloxatin;1-OHP;(1R,2R)-cyclohexane-1,2-diamine; oxalic acid; platinum(+2) cation;Eloxatin (TN);Trans-l-diaminocyclohexane oxalatoplatinum;Platinum, (1,2-cyclohexanediamine-N,N)(ethanedioato(2-)-O,O-, (SP-4-2-(1R-trans))-;Platinum, [ (1R,2R)-1,2-cyclohexanediamine-.kappa.N, .kappa.N][ethanedioato(2-)-.kappa.O1,.kappa.O2]-,(SP-4-2)-;(SP-4-2-(1R-trans))-(1,2-Cyclohexanediamine-N,N)(ethanedioato(2-)-O,O)platinum;RP 54780;Oxalatoplatinum;Oxaliplatin EP5;
- PSA 104.64000
- LogP 0.61450
Synthetic route

-
-
583-52-8
potassium oxalate

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In water at 60 - 70℃; for 5 - 6h; Product distribution / selectivity; Heating / reflux; | 67% |

-
-
583-52-8
potassium oxalate

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In water for 5 - 6h; Product distribution / selectivity; Heating / reflux; | 67% |
-
-
144-62-7
oxalic acid

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II) With water; silver nitrate at 20 - 45℃; for 2.08333h; Stage #2: oxalic acid With potassium hydroxide In water at 20℃; for 4h; pH=2.9; Product distribution / selectivity; | 62.9% |
| Stage #1: (1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II) With silver nitrate In water at 20℃; for 70h; Stage #2: With tetraethylammonium iodide In water for 16h; Stage #3: oxalic acid In water for 4h; | 54% |
| Stage #1: (1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II) With water; silver nitrate at 20℃; for 70h; Stage #2: oxalic acid In water at 20℃; for 4h; Product distribution / selectivity; | 40.3% |

-
-
583-52-8
potassium oxalate

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| Stage #1: trans-(R,R)-(1,2-diaminocyclohexane)diiodoplatinum(II) With acetic acid; silver(l) oxide In water at 80 - 90℃; for 4h; Stage #2: potassium oxalate In water at 60℃; for 5h; Product distribution / selectivity; | 60% |
-
-
61848-66-6
(1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II)

-
-
33081-83-3
bis(tetrabutylammonium)oxalate

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In i-Amyl alcohol; water at 85 - 88℃; for 8 - 10h; Product distribution / selectivity; | 50% |
-
-
61848-66-6
(1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II)

-
-
7732-18-5
water

-
-
144-62-7
oxalic acid

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II); water With silver nitrate at 20℃; for 48h; Stage #2: oxalic acid In water at 20℃; for 4h; Product distribution / selectivity; | A 44.1% B n/a |

-
-
61848-66-6
(1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II)

-
-
15843-43-3, 31038-77-4
silver oxalate

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In water | 41% |

-
-
144-62-7
oxalic acid

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| With silver(l) oxide In water at 60 - 100℃; for 13 - 14h; Product distribution / selectivity; | 38% |
-
-
61848-66-6
(1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II)

-
-
144-62-7
oxalic acid

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| With silver(l) oxide In water at 60 - 100℃; for 13 - 14h; Product distribution / selectivity; | 38% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; for 20h; pH=6; | 35% |
| With sodium hydroxide at 20℃; for 20h; pH=6; | 33% |

| Conditions | Yield |
|---|---|
| With silver(l) oxide In water at 90 - 100℃; for 10 - 12h; Product distribution / selectivity; | 30% |

| Conditions | Yield |
|---|---|
| With silver(l) oxide In water at 90 - 100℃; for 10 - 12h; Product distribution / selectivity; | 30% |

| Conditions | Yield |
|---|---|
| With potassium iodide; silver(l) oxide In water at 90 - 100℃; for 10 - 12h; Product distribution / selectivity; | 20% |

-
-
10025-99-7
potassium tetrachloroplatinate(II)

-
-
20439-47-8
(1R,2R)-1,2-diaminocyclohexane

-
-
144-62-7
oxalic acid

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| With silver(l) oxide In water at 90 - 100℃; for 10 - 12h; | 20% |


| Conditions | Yield |
|---|---|
| With acetic acid In water at 90℃; for 6h; pH=6.6 - 7.5; Product distribution / selectivity; | 15% |
| In water; acetone at 95℃; for 1.5h; Product distribution / selectivity; | 15% |

-
-
583-52-8
potassium oxalate

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In water for 6h; Product distribution / selectivity; Heating / reflux; |

| Conditions | Yield |
|---|---|
| In water at 45℃; for 8h; |

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| Product distribution / selectivity; |

-
-
144-62-7
oxalic acid

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In water at 20℃; for 4h; Product distribution / selectivity; |

-
-
61848-66-6
(1R,2R)-1,2-diaminocyclohexanedichloroplatinum(II)

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| purifn. by HPLC; elem.anal.;; |

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In water at room temp. in sunlight; chromy.; UV; | |
| With ascorbic acid In water at room temp. in the dark; chromy.; UV; |

-
-
62-76-0
sodium oxalate

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| at 20℃; for 2 - 4h; pH=12; Product distribution / selectivity; Absence of light; |


| Conditions | Yield |
|---|---|
| With oxalic acid In water for 4h; Product distribution / selectivity; |

-
-
583-52-8
potassium oxalate

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In water for 6h; Product distribution / selectivity; Heating / reflux; |

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| In aq. phosphate buffer pH=7; Electrochemical reaction; |

| Conditions | Yield |
|---|---|
| With sodium L-ascorbate In water |

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| With C49H42N7Ru(3+)*3F6P(1-); water-d2 for 1h; Irradiation; |

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| With sodium L-ascorbate In N,N-dimethyl-formamide; acetonitrile for 0.166667h; pH=7.4; Mechanism; Irradiation; |

| Conditions | Yield |
|---|---|
| With sodium L-ascorbate In N,N-dimethyl-formamide for 0.166667h; pH=7.4; Mechanism; Irradiation; |

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| With sodium L-ascorbate In aq. phosphate buffer at 37℃; under 760.051 Torr; pH=7.4; Kinetics; Darkness; |

| Conditions | Yield |
|---|---|
| for 12h; | 94% |

| Conditions | Yield |
|---|---|
| In water at 70℃; for 5h; | 92% |

-
-
61825-94-3
oxaliplatin

-
-
7722-84-1
dihydrogen peroxide

| Conditions | Yield |
|---|---|
| In water at 50℃; for 1h; | 90% |
| In water at 20℃; for 24h; Darkness; | 89% |
| In water at 70℃; for 5h; | 80.6% |
-
-
61825-94-3
oxaliplatin

-
-
7722-84-1
dihydrogen peroxide

| Conditions | Yield |
|---|---|
| In water at 50℃; for 5h; Darkness; | 90% |
| In water at 60 - 65℃; Darkness; | 74% |
| In water at 60℃; for 4h; | 71% |

| Conditions | Yield |
|---|---|
| at 50℃; for 1h; | 90% |


| Conditions | Yield |
|---|---|
| at 20℃; for 3h; Darkness; | 89.3% |
| at 20℃; Inert atmosphere; | 89.2% |
| at 60℃; Darkness; | 89% |

-
-
61825-94-3
oxaliplatin

-
-
128-09-6
N-chloro-succinimide

| Conditions | Yield |
|---|---|
| In ethanol for 8h; Darkness; Reflux; | 89% |
-
-
61825-94-3
oxaliplatin

-
-
516-12-1
N-iodo-succinimide

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 8h; Darkness; | 84% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 1h; | 83% |


| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 81% |

-
-
61825-94-3
oxaliplatin

-
-
7722-84-1
dihydrogen peroxide

-
-
111263-58-2, 111321-67-6, 111321-78-9, 198292-19-2
cis-oxalic acid (trans-1,2-cyclohexanediamine)dihydroxyplatinum(II)

| Conditions | Yield |
|---|---|
| at 50℃; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: oxaliplatin With dihydrogen peroxide In water at 20 - 50℃; for 27h; Darkness; Stage #2: succinic acid anhydride In N,N-dimethyl-formamide at 50℃; for 24h; Darkness; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: oxaliplatin With dihydrogen peroxide In water at 20 - 50℃; for 27h; Stage #2: propionic acid anhydride In N,N-dimethyl-formamide at 50℃; for 20h; Darkness; | 80% |

| Conditions | Yield |
|---|---|
| at 60℃; for 4h; | 78% |
| at 60℃; for 4h; | 76% |
| at 40℃; for 18h; |


| Conditions | Yield |
|---|---|
| Stage #1: oxaliplatin With dihydrogen peroxide; acetic acid at 20℃; for 24h; Darkness; Stage #2: succinic acid anhydride In N,N-dimethyl-formamide at 50℃; for 24h; Darkness; | 76% |

| Conditions | Yield |
|---|---|
| In water; acetone at 20℃; for 11.25h; | 75% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 48h; Darkness; | 75% |
| In water at 20℃; for 19h; Darkness; |


| Conditions | Yield |
|---|---|
| In water; acetone at 20℃; for 18.7833h; | 74% |

-
-
61825-94-3
oxaliplatin

-
-
128-08-5
N-Bromosuccinimide

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 12h; | 71% |
-
-
79-21-0
peracetic acid

-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| With acetic acid for 24.5h; | 66% |
-
-
61825-94-3
oxaliplatin

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water at 50℃; for 3h; Inert atmosphere; | 64.3% |
| With dihydrogen peroxide In water at 30℃; for 12h; Darkness; | |
| With dihydrogen peroxide In water at 60 - 70℃; for 4h; Inert atmosphere; |
-
-
61825-94-3
oxaliplatin

-
-
79-08-3
bromoacetic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In tetrahydrofuran at 20℃; for 24h; | 53% |
Oxaliplatin Chemical Properties
Oxaliplatin is a platinum-based chemotherapy drug in the same family as cisplatin and carboplatin. Compared to cisplatin the two amine groups are replaced by cyclohexyldiamine for improved antitumour activity. The chlorine ligands are replaced by the oxalato bidentate derived from oxalic acid in order to improve water solubility.
Product Name: Oxaliplatin (CAS NO.61825-94-3)
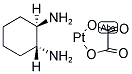
Molecular Formula: C8H14N2O4Pt
Molecular Weight: 397.29g/mol
Mol File: 61825-94-3.mol
Appearance: White Crystalline Solid
Boiling point: 193.6 °C at 760 mmHg
Flash Point: 75 °C
Stability: Stable. Store cool. Incompatible with oxidizing agents.
Enthalpy of Vaporization: 42.98 kJ/mol
Vapour Pressure: 0.46 mmHg at 25°C
H-Bond Donor: 2
H-Bond Acceptor: 6
Structure Descriptors of Oxaliplatin (CAS NO.61825-94-3):
IUPAC Name: (1R,2R)-cyclohexane-1,2-diamine; oxalate; platinum(4+)
Canonical SMILES: C1CCC(C(C1)N)N.C(=O)(C(=O)[O-])[O-].[Pt+4]
Isomeric SMILES: C1CC[C@H]([C@@H](C1)N)N.C(=O)(C(=O)[O-])[O-].[Pt+4]
InChI: InChI=1S/C6H14N2.C2H2O4.Pt/c7-5-3-1-2-4-6(5)8;3-1(4)2(5)6;/h5-6H,1-4,7-8H2;(H,3,4)(H,5,6);/q;;+4/p-2/t5-,6-;;/m1../s1
InChIKey: PCRKGXHIGVOZKB-BNTLRKBRSA-L
Product Categories: Pharmaceutical material and intermeidates; Active Pharmaceutical Ingredients; Antineoplastic; Intermediates & Fine Chemicals; Pharmaceuticals
Oxaliplatin History
Oxaliplatin was discovered in 1976 at Nagoya City University in Japan by Professor Yoshinori Kidani, who was granted U.S. Patent 4,169,846 over the drug in 1979. Oxaliplatin was subsequently in-licensed by Debiopharm, a Swiss drug company headquartered in Lausanne and developed as an advanced colorectal cancer treatment. Debio licensed the drug to Sanofi-Aventis in 1994. Eloxatin gained European approval in 1996 (firstly in France) and approval by the U.S. Food and Drug Administration (FDA) in 2002.
Oxaliplatin Uses
It is typically administered in combination with fluorouracil and leucovorin in a combination known as FOLFOX for the treatment of colorectal cancer.
Oxaliplatin Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 19800ug/kg (19.8mg/kg) | Drugs of the Future. Vol. 14, Pg. 529, 1989. | |
| rat | LD50 | intraperitoneal | 14300ug/kg (14.3mg/kg) | Drugs of the Future. Vol. 14, Pg. 529, 1989. |
Oxaliplatin Safety Profile
Safety Information of Oxaliplatin (CAS NO.61825-94-3):
Hazard Codes: Xn ,Xi
,Xi
Risk Statements: 36/37/38-40-42/43
R36/37/38:Irritating to eyes, respiratory system and skin.
R40:Limited evidence of a carcinogenic effect.
R42/43:May cause sensitization by inhalation and skin contact.
Safety Statements: 26-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
RIDADR: 2811
WGK Germany: 3
RTECS: TP2275850
HazardClass: 6.1(a)
PackingGroup: II
Oxaliplatin Specification
Oxaliplatin , its CAS NO. is 61825-94-3, the synonyms are (SP-4-2-(1R-trans))-(1,2-Cyclohexanediamine-N,N')(ethanedioato(2-)-O,O')platinum ; Dacplat ; Eloxatin ; Elplat ; JM-83 ; Lipoxal ; Oxalato(1R,2R-cyclohexanediammine)platinum(II) ; Oxalato(trans-(-)-1,2-cyclohexanediamine)platinum(II) ; Oxalato(trans-1-1,2-cyclohexanediamine)platinum(II) ; Oxalatoplatinum ; UNII-04ZR38536J ; cis-((1R,2R)-1,2-Cyclohexanediamine-N,N')(oxalato(2-)-O,O')platinum ; trans-l-Diaminocyclohexane oxalatoplatinum ; Platinum, ((1R,2R)-1,2-cyclohexanediamine-.kappa.N,.kappa.N')(ethanedioato(2-)-kappaO1,kappaO2)-, (SP-4-2)- ; Platinum, (1,2-cyclohexanediamine-N,N')(ethanedioato(2-)-O,O')-, (SP-4-2-(1R-trans))- .
Related Products
- Oxaliplatin
- Oxaliplatin
- 61825-98-7
- 61826-76-4
- 618-26-8
- 61827-42-7
- 61827-59-6
- 61827-71-2
- 61827-89-2
- 618-27-9
- 61830-21-5
- 61830-23-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View