Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:42924-53-8
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou JINLAN Pharm-Drugs Technology Co., Ltd
We can provide GMP validation service that complies with SFDA, FDA, WHO and EU EMPA.Excellent registration team could help us easlily to register our products in different countries.If you and your customer are interested in some products or need C
Cas:42924-53-8
Min.Order:1 Gram
Negotiable
Type:Manufacturers
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:42924-53-8
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
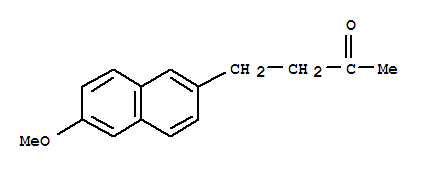
Product Name: Nabumetone Synonyms: 4-[6-(trideuteriomethoxy)-2-naphthalenyl]-2-butanone;NABUMETONE, EP STANDARD;NABUMETONE, MM(CRM STANDARD);NABUMETONE, IMP. F (EP): 6,6'-DIMETHOXY-2,2'-BINAPHTHALENYL MM(CRM STANDARD);NABUMETONE, IMPURITY
Cas:42924-53-8
Min.Order:1 Kilogram
FOB Price: $230.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:42924-53-8
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available ♦ W
Cas:42924-53-8
Min.Order:1 Kilogram
FOB Price: $7.0 / 9.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:42924-53-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Nabumetone CAS:42924-53-8 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates,
Cas:42924-53-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:42924-53-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:42924-53-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryLonwin Chemical Group Limited
Nabumetone CAS: 42924-53-8 Specification Item Standard Structural data confirmation 1. H-NMR: The data of structure are identical with reference standard 2. HPLC-MS: Ensure the mo
Henan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:42924-53-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Hefei Zhaobo Technology Co., Ltd.
Nabumetone cas 42924-53-8 Purity: 99% Min Application: Intermediates Appearance: Powder Package: Bag Delivery: 3-5days Our Advantage & Service 1.Top quality: Using high quality material and establishing a strict quality control syste
Cas:42924-53-8
Min.Order:1 Kilogram
FOB Price: $10.0 / 50.0
Type:Trading Company
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Hebei Miheng Trading Co., Ltd.
Nabumetone CAS 42924-53-8 Our advantage 1.Top quality: Using high quality material and establishing a strict quality control system,assigning specific persons in charge of each part of production,from raw material purchase to assembly. 2 Pr
Cas:42924-53-8
Min.Order:1 Kilogram
FOB Price: $50.0 / 100.0
Type:Trading Company
inquirySHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:42924-53-8
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryTaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providing h
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Suzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:Active Pharmaceutical I
Golden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Hubei Vanz Pharm Co.,Ltd
ISO Factory/Good qualityAppearance:off white Storage:dry and cool place Package:10G/BAG; 1KG/BAG ;25KG/DRUM Application:Active pharaceutical ingredients Transportation:BY SEA,AIR,EXPRESS Port:shenzhen/shanghai
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Synthetic route
-
-
127053-22-9, 56600-90-9
4-(2-methoxynaphthalene-6-yl)but-3-en-2-one

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With hydrogen In toluene at 50℃; under 3750.38 Torr; for 1h; Catalytic behavior; Temperature; Pressure; Inert atmosphere; Schlenk technique; Autoclave; | 99% |
| With 1% Pd on activated carbon; hydrogen In water at 45℃; under 760.051 Torr; for 15h; Reagent/catalyst; Green chemistry; chemoselective reaction; | 95% |
| With hydrogen In N,N-dimethyl-formamide at 100℃; | 90% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; silver(I) acetate; acetic acid at 25℃; for 24h; | 99% |
-
-
73356-31-7
4-(6-methoxy-2-naphthalenyl)-4-hydroxybut-3-en-2-one

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| palladium on charcoal catalyst In acetic acid | 93% |
| Multi-step reaction with 3 steps 1: acetic acid; hydrogen; palladium on activated charcoal / 20 °C 2: sulfuric acid 3: hydrogen / ethyl acetate / 4 h / 20 °C / 760.05 Torr View Scheme | |
| Multi-step reaction with 3 steps 1: hydrogen; palladium 10% on activated carbon; sulfuric acid / ethyl acetate 2: sulfuric acid 3: hydrogen / ethyl acetate / 4 h / 20 °C / 760.05 Torr View Scheme |

| Conditions | Yield |
|---|---|
| With hydrogen; Pd/Mg-Al hydrotalcite at 74.84℃; under 3750.38 Torr; for 1.25h; | A 11 % Chromat. B 89% |


| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; tetra-(n-butyl)ammonium iodide; nickel dibromide In pyridine; N,N-dimethyl-formamide at 70℃; Electrochemical reaction; | 85% |
| Stage #1: methyl vinyl ketone With [2,2]bipyridinyl; water; cobalt(II) bromide In N,N-dimethyl-formamide at 20℃; for 0.166667h; Stage #2: 2-Bromo-6-methoxynaphthalene With pyridine; trifluoroacetic acid; lithium bromide; zinc In N,N-dimethyl-formamide at 80℃; for 0.333333h; | 50% |
-
-
598-32-3
2-hydroxy-3-butene

-
-
5111-65-9
2-Bromo-6-methoxynaphthalene

-
A
-
42924-53-8
nabumetone

-
B
-
127053-21-8
4-(6-methoxy-2-naphthyl)-3-butene-2-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxy-3-butene With dicyclohexylmethylamine; tetrabutylammomium bromide In N,N-dimethyl acetamide; water at 20℃; for 0.25h; Heck reaction; Stage #2: 2-Bromo-6-methoxynaphthalene; 4'-hydroxyacetophenone oxime palladacycle catalyst In N,N-dimethyl acetamide; water at 120℃; for 9h; Heck reaction; Further stages.; | A 82% B n/a |
| With sodium hydrogencarbonate; bis-triphenylphosphine-palladium(II) chloride In various solvent(s) at 140℃; for 5h; Yield given; | A n/a B 9% |


-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium acetate; sodium acetate trihydrate; palladium In methanol; water; hydrogen; toluene | 79.7% |
| With sodium hydroxide; sodium acetate; sodium acetate trihydrate; palladium In methanol; water; hydrogen; toluene | 79.7% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium acetate; palladium In water; hydrogen; isopropyl alcohol; toluene | 78.5% |
| With sodium hydroxide; sodium acetate; palladium In water; hydrogen; isopropyl alcohol; toluene | 78.5% |
| With sodium hydroxide; sodium acetate; palladium In water; hydrogen; isopropyl alcohol; toluene | 74.8% |
| With sodium hydroxide; sodium acetate; palladium In water; hydrogen; isopropyl alcohol; toluene | 74.8% |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium formate; sodium hydrogencarbonate; Pd-benzothiazole carbene at 130℃; for 3h; Heck reaction; | 75% |
| With tetrabutylammomium bromide; sodium hydrogencarbonate; palladium dichloride at 120℃; for 6h; Heck coupling; | 74% |
| Stage #1: 2-hydroxy-3-butene With dicyclohexylmethylamine; tetrabutylammomium bromide In N,N-dimethyl acetamide at 20℃; for 0.25h; Heck reaction; Stage #2: 2-Bromo-6-methoxynaphthalene; 4'-hydroxyacetophenone oxime palladacycle catalyst In N,N-dimethyl acetamide at 120℃; for 24h; Heck reaction; Further stages.; | 90 % Spectr. |
| With Fe3O4@chitosan nanoparticles/Pd In ethanol at 140℃; for 24h; |

| Conditions | Yield |
|---|---|
| With [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; carbon monoxide In neat (no solvent) at 200℃; for 72h; Temperature; Autoclave; | 75% |
| With hydrogen; magnesium oxide; palladium at 59.84℃; under 3750.38 Torr; for 1h; | 98 % Chromat. |
-
-
3453-33-6
6-methoxynaphthalene-2-carbaldehyde

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| 67% | |
| Multi-step reaction with 2 steps 1: aq. NaOH 2: H2 / Pd-C / ethyl acetate View Scheme | |
| Multi-step reaction with 2 steps 1: N,N-dimethyl-formamide / 210 °C / Continuous flow 2: hydrogen / N,N-dimethyl-formamide / 100 °C View Scheme | |
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / 0.12 h / 70 °C / Microwave irradiation; Continuous flow 2: hydrogen / N,N-dimethyl-formamide / 100 °C View Scheme | |
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / 4 h 2: hydrogen / ethyl acetate / 4 h / 20 °C / 760.05 Torr View Scheme |
-
-
73356-31-7
4-(6-methoxy-2-naphthalenyl)-4-hydroxybut-3-en-2-one

-
A
-
65726-24-1
4-(6-methoxynaphthalen-2-yl)butan-2-ol

-
B
-
42924-53-8
nabumetone

-
C
-
220786-56-1, 98386-83-5
(±)-4-hydroxy-4-(6-methoxy-2-naphthyl)-2-butanone

| Conditions | Yield |
|---|---|
| With sulfuric acid; palladium 10% on activated carbon; hydrogen In ethyl acetate | A 5% B 65% C 5% |


| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; chloro-trimethyl-silane; nickel(II) bromide 2-methoxyethyl ether complex; zinc In tetrahydrofuran; N,N-dimethyl acetamide at 40℃; for 1h; Sealed tube; Inert atmosphere; | 54% |


-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium acetate; sodium hydrogensulfite; palladium In methanol; water; hydrogen; N,N-dimethyl-formamide; isopropyl alcohol | 52.8% |
| With sodium hydroxide; sodium acetate; sodium hydrogensulfite; palladium In methanol; water; hydrogen; N,N-dimethyl-formamide; isopropyl alcohol | 52.8% |
-
-
136579-95-8
methyl 3-(6-methoxynaphthalen-2-yl)propanoate

-
-
917-64-6
methyl magnesium iodide

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -55℃; | 51% |
-
-
917-64-6
methyl magnesium iodide

-
-
42924-52-7
2-Methoxynaphthalen-6-propionitril

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| In diethyl ether Heating; |

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water for 6h; Heating; | 4 g |
-
-
73022-40-9
2-(Bromomethyl)-6-methoxynaphthalene

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: K2CO3 / acetone / 3 h / Heating 2: 4 g / KOH / H2O / 6 h / Heating View Scheme |
-
-
42924-51-6
2-(2-bromoethyl)-6-methoxynaphthalene

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. ethanol / Heating 2: diethyl ether / Heating View Scheme |
-
-
32725-05-6, 56523-40-1
2-<1-hydroxyethyl>-6-methoxynaphthalene

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: PBr3 / benzene / Heating 2: aq. ethanol / Heating 3: diethyl ether / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: triphenylphosphine; 1H-imidazole; iodine / dichloromethane / 20 °C 2: [2,2]bipyridinyl; nickel(II) bromide 2-methoxyethyl ether complex; zinc; chloro-trimethyl-silane / N,N-dimethyl acetamide; tetrahydrofuran / 1 h / 40 °C / Sealed tube; Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: 1H-imidazole; triphenylphosphine; iodine / dichloromethane / 0 - 20 °C / Inert atmosphere 2: (2-(dimethylamino)-N-(quinolin-8-yl)acetamido)nickel(II) chloride; manganese / N,N-dimethyl-formamide / 16 h / 20 °C / Sealed tube View Scheme |
-
-
23981-48-8
methyl 6-methoxy-2-naphthylacetate

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: LiAlH4 / diethyl ether / Heating 2: PBr3 / benzene / Heating 3: aq. ethanol / Heating 4: diethyl ether / Heating View Scheme |
-
-
598-32-3
2-hydroxy-3-butene

-
-
5111-65-9
2-Bromo-6-methoxynaphthalene

-
B
-
42924-53-8
nabumetone

-
C
-
56600-77-2
3-(6-methoxy-2-naphthyl)-2-butanone

| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxy-3-butene With dicyclohexylmethylamine; tetrabutylammomium bromide In N,N-dimethyl acetamide at 20℃; for 0.25h; Heck reaction; Stage #2: 2-Bromo-6-methoxynaphthalene; Kaiser oxime-derived complex on resin base In N,N-dimethyl acetamide at 120℃; for 24h; Heck reaction; Further stages. Title compound not separated from byproducts.; |

-
-
598-32-3
2-hydroxy-3-butene

-
-
5111-65-9
2-Bromo-6-methoxynaphthalene

-
A
-
42924-53-8
nabumetone

-
B
-
56600-77-2
3-(6-methoxy-2-naphthyl)-2-butanone

| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxy-3-butene With dicyclohexylmethylamine; tetrabutylammomium bromide In N,N-dimethyl acetamide at 20℃; for 0.25h; Heck reaction; Stage #2: 2-Bromo-6-methoxynaphthalene; Kaiser oxime-derived complex on resin base In N,N-dimethyl acetamide at 120℃; for 14h; Heck reaction; Further stages. Title compound not separated from byproducts.; |

-
-
1250259-21-2
ethyl-5-(6-methoxy-2-naphthyl)-β-keto-pentanoate

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With water; potassium hydroxide In ethanol Conversion of starting material; |
-
-
135-19-3
β-naphthol

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: acetic acid; bromine / 0.5 h / 30 - 35 °C 2.1: acetic acid; tin / 3 h / 100 - 120 °C 3.1: potassium carbonate / acetone; water / 4 h / 10 - 50 °C / Cooling 4.1: magnesium; iodine / tetrahydrofuran / 5 h / Reflux 4.2: 1.75 h / 10 - 90 °C 4.3: 1 h / 20 °C 5.1: sodium hydroxide / water / 4 h 6.1: hydrogen / ethyl acetate / 4 h / 20 °C / 760.05 Torr View Scheme |
-
-
16239-18-2
1,6-dibromo-2-naphthol

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: acetic acid; tin / 3 h / 100 - 120 °C 2.1: potassium carbonate / acetone; water / 4 h / 10 - 50 °C / Cooling 3.1: magnesium; iodine / tetrahydrofuran / 5 h / Reflux 3.2: 1.75 h / 10 - 90 °C 3.3: 1 h / 20 °C 4.1: sodium hydroxide / water / 4 h 5.1: hydrogen / ethyl acetate / 4 h / 20 °C / 760.05 Torr View Scheme |
-
-
15231-91-1
6-bromo-naphthalen-2-ol

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: potassium carbonate / acetone; water / 4 h / 10 - 50 °C / Cooling 2.1: magnesium; iodine / tetrahydrofuran / 5 h / Reflux 2.2: 1.75 h / 10 - 90 °C 2.3: 1 h / 20 °C 3.1: sodium hydroxide / water / 4 h 4.1: hydrogen / ethyl acetate / 4 h / 20 °C / 760.05 Torr View Scheme |
-
-
5111-65-9
2-Bromo-6-methoxynaphthalene

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: magnesium; iodine / tetrahydrofuran / 5 h / Reflux 1.2: 1.75 h / 10 - 90 °C 1.3: 1 h / 20 °C 2.1: sodium hydroxide / water / 4 h 3.1: hydrogen / ethyl acetate / 4 h / 20 °C / 760.05 Torr View Scheme | |
| Multi-step reaction with 3 steps 1.1: n-butyllithium / diethyl ether / 0.5 h / 0 °C / Inert atmosphere 1.2: 1.42 h / Inert atmosphere 2.1: triphenylphosphine; 1H-imidazole; iodine / dichloromethane / 20 °C 3.1: [2,2]bipyridinyl; nickel(II) bromide 2-methoxyethyl ether complex; zinc; chloro-trimethyl-silane / N,N-dimethyl acetamide; tetrahydrofuran / 1 h / 40 °C / Sealed tube; Inert atmosphere View Scheme |
-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With tris(pentafluorophenyl)borate; water-d2 In tetrahydrofuran at 100℃; for 12h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; regioselective reaction; | 99% |
-
-
42924-53-8
nabumetone

-
-
1225055-49-1
4-(5-iodo-6-methoxynaphthalen-2-yl)butan-2-one

| Conditions | Yield |
|---|---|
| With indium(III) triflate; N-iodo-succinimide In acetonitrile at 23℃; for 8h; Inert atmosphere; Darkness; | 97% |
-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
5736-88-9
p-butoxybenzaldehyde

-
-
1363178-37-3
1-(4-butoxyphenyl)-5-(6-methoxynaphthalen-2-yl)-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; p-butoxybenzaldehyde In ethanol at 25℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 25℃; for 22h; pH=4 - 5; Mannich reaction; | 97% |

-
-
42924-53-8
nabumetone

-
-
65726-24-1
4-(6-methoxynaphthalen-2-yl)butan-2-ol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol at -10 - 20℃; for 4h; | 95% |
| With sodium tetrahydroborate In ethanol | |
| With sodium tetrahydroborate In ethanol at -10℃; for 3h; Inert atmosphere; | |
| With sodium tetrahydroborate In ethanol at 0 - 20℃; for 3h; Schlenk technique; Inert atmosphere; |
-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
104-87-0
4-methyl-benzaldehyde

-
-
1363178-36-2
5-(6-methoxynaphthalen-2-yl)-1-p-tolyl-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; 4-methyl-benzaldehyde In ethanol at 20℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 20℃; for 26h; pH=4 - 5; Mannich reaction; | 95% |

-
-
42924-53-8
nabumetone

-
-
68427-21-4
Nabumetone hydroxyimine

| Conditions | Yield |
|---|---|
| With pyridine; hydroxylamine hydrochloride In ethanol Heating; | 94% |
| With hydroxylamine hydrochloride |
-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With nickel(II) tetrafluoroborate hexahydrate; ammonia; hydrogen; bis(2-diphenylphosphinoethyl)phenylphosphine In 2,2,2-trifluoroethanol at 120℃; for 24h; chemoselective reaction; | 91% |
| With ammonia; hydrogen In tetrahydrofuran at 120℃; for 15h; | 90% |
| With N,N'-bis(salicylidene)-1,2-phenylene-diaminocobalt(II); ammonia; hydrogen In tetrahydrofuran; water at 130℃; for 24h; Autoclave; | 82% |
| With ammonium hydroxide; hydrogen at 140℃; under 48754.9 Torr; for 20h; |
-
-
42924-53-8
nabumetone

-
-
38676-25-4
4-Diazo-2,6-dibromo-2,5-cyclohexadienone

| Conditions | Yield |
|---|---|
| With bis{rhodium[3,3'-(1,3-phenylene)bis(2,2-dimethylpropanoic acid)]} at 20℃; for 16h; Inert atmosphere; Schlenk technique; Molecular sieve; | 91% |
-
-
42924-53-8
nabumetone

-
-
31862-15-4
2,6-dichloro-4-diazocyclohexa-2,5-dien-1-one

| Conditions | Yield |
|---|---|
| With bis{rhodium[3,3'-(1,3-phenylene)bis(2,2-dimethylpropanoic acid)]} at 20℃; for 16h; Inert atmosphere; Schlenk technique; Molecular sieve; | 90% |
-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
1363178-35-1
1-(4-bromophenyl)-5-(6-methoxynaphthalen-2-yl)-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; 4-bromo-benzaldehyde In ethanol at 25℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 25℃; for 24h; pH=4 - 5; Mannich reaction; | 89% |

-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
6287-38-3
3,4-dichlorobenzaldehyde

-
-
1206182-19-5
1-(3,4-dichlorophenyl)-5-(6-methoxynaphthalen-2-yl)-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; 3,4-dichlorobenzaldehyde In ethanol at 30℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 30℃; for 29h; pH=4 - 5; Mannich reaction; | 89% |

-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
620-23-5
m-tolyl aldehyde

-
-
1206182-16-2
5-(6-methoxynaphthalen-2-yl)-1-m-tolyl-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; m-tolyl aldehyde In ethanol at 31℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 31℃; for 23h; pH=4 - 5; Mannich reaction; | 89% |

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With bis{rhodium[3,3'-(1,3-phenylene)bis(2,2-dimethylpropanoic acid)]} In 1,2-dichloro-ethane at 50℃; for 16h; Inert atmosphere; Schlenk technique; Molecular sieve; | 89% |
-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
123-08-0
4-hydroxy-benzaldehyde

-
-
1363178-38-4
1-(4-hydroxyphenyl)-5-(6-methoxynaphthalen-2-yl)-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; 4-hydroxy-benzaldehyde In ethanol at 20℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 20℃; for 25h; pH=3 - 4; Mannich reaction; | 88% |

-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
1206182-20-8
5-(6-methoxynaphthalen-2-yl)-1-(4-methoxyphenyl)-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; 4-methoxy-benzaldehyde In ethanol at 32℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 32℃; for 28h; pH=3 - 4; Mannich reaction; | 87% |

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Stage #1: nabumetone With ammonium hydroxide; hydrogen at 60℃; under 7500.75 Torr; for 20h; Autoclave; Stage #2: With hydrogenchloride In diethyl ether | 87% |
-
-
76-84-6
triphenylmethyl alcohol

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| Stage #1: triphenylmethyl alcohol; nabumetone With potassium phosphate; chloro(1,5-cyclooctadiene)rhodium(I) dimer; potassium tert-butylate; C69H57N2O2(1+)*BF4(1-)*H(1+) In toluene at 125℃; for 24h; Sealed tube; Stage #2: With sodium tetrahydroborate In methanol at 0 - 20℃; for 1h; | 86% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; chloroform; water at 20 - 32℃; for 27h; | 85% |


| Conditions | Yield |
|---|---|
| Stage #1: nabumetone With sodium hydroxide In methanol at 20℃; for 0.0833333h; Stage #2: benzaldehyde In methanol at 20℃; for 16h; | 82% |
-
-
2295-31-0
2,4-thiazolidinedion

-
-
42924-53-8
nabumetone

| Conditions | Yield |
|---|---|
| With piperidine; benzoic acid In toluene Heating; | 80% |
-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
1363178-31-7
1-(3-fluorophenyl)-5-(6-methoxynaphthalen-2-yl)-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; 3-Fluorobenzaldehyde In ethanol at 13℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 13℃; for 23h; pH=4 - 5; Mannich reaction; | 80% |

-
-
455-14-1
4-trifluoromethylphenylamine

-
-
42924-53-8
nabumetone

-
-
100-52-7
benzaldehyde

-
-
1236218-15-7
5-(6-methoxynaphthalen-2-yl)-1-phenyl-1-(4-(trifluoromethyl)phenylamino)pentan-3-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-trifluoromethylphenylamine; benzaldehyde In ethanol at 31℃; for 0.333333h; Mannich reaction; Stage #2: nabumetone With hydrogenchloride In ethanol; chloroform; water at 31℃; for 25h; pH=4 - 5; Mannich reaction; | 79% |

Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 Xn
Xn