Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:4696-76-8
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:4696-76-8
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:4696-76-8
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryLeader Biochemical Group
PRODUCT DETAILS
Cas:4696-76-8
Min.Order:50 Kilogram
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryWuhan Fortuna Chemical Co.,Ltd
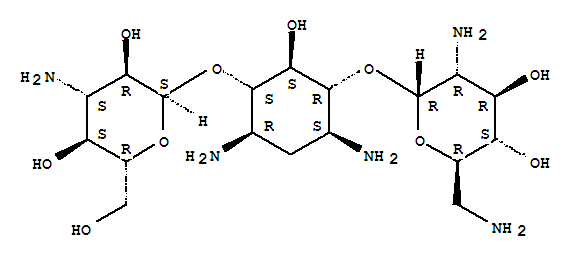
CAS RN.: 4696-76-8 EINECS: 225-170-5 Molecular Weight:
Cas:4696-76-8
Min.Order:25 Kilogram
Negotiable
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Cas:4696-76-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
1)quick response within 12 hours; 2)quality guarantee: all products are strictly tested by our qc, confirmed by qa and approved by third party lab in china, usa, canada, germany, uk, italy, france etc. 3) oem/odm available; 4) reasonable &
Cas:4696-76-8
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:4696-76-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Fonlynn Health Technology Co., Ltd.
Packing: According to customer requirements Delivery time: In stock or depands Port of shipment: Ningbo/Shanghai/Qingdao OEM/ODM:Welcome Sample:We can offer our existing samples at once Transportation:Express/Sea/Air Port:Ningbo/Shanghai/Qingd
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:4696-76-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
bekanamycinAppearance:white powder Storage:sealed, keep in the ventilated, dry Package:according to customers' requirements Application:health care Transportation:By air(EMS or EUB or FedEx or TNT ect...) or by sea(FOB or CIF or CNF ect...)or accordi
TaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Henan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung, LG, Merck, Thermo Fisher Scientific and so o
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:4696-76-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:4696-76-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Dawn Ray Pharmaceutical Co.,Ltd
≥85.0% HPLC Appearance:white powder Storage:Room temperature Package:25kg/drum or as required Application:used in medicine Transportation:by sea by air express Port:China main port
Cas:4696-76-8
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service an
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Hubei Vanz Pharm Co.,Ltd
ISO Factory/Good qualityAppearance:off white Storage:dry and cool place Package:10G/BAG; 1KG/BAG ;25KG/DRUM Application:Active pharaceutical ingredients Transportation:BY SEA,AIR,EXPRESS Port:SHANGHAI;BEIJING
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,or Intermediates,fine chemicals Transportation:air,sea,courier
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Sinoway Industrial Co., Ltd.
Why is SINOWAY:1) Specialized in pharmaceutical and healthcare industrial since 19872) ISO 9001:2015 & SGS audited supplier . 3) Accept various payment terms : T.T 30-60 days.4) We have warehouse in USA with quickly shipment . Application:API
Cas:4696-76-8
Min.Order:0
Negotiable
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Hubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Hangzhou Huarong Pharm Co., Ltd.
High qualityAppearance:White powder Storage:Room temperature Package:Aluminum bag Application:Used in producing API Transportation:By air Port:Beijing
Antimex Chemical Limied
hight degree of purity Application:Fine chemical intermediates, used as the main raw material for he synthesis of various pesticides, medicines, surfactants, polymer monomers, Ond Ontifungal agents
Cas:4696-76-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Fandachem Co.,Ltd
BekanamycinAppearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:pharmaceutical intermediate Transportation:by air, by sea, by express
Synthetic route
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
4696-76-8
kananmycin B

| Conditions | Yield |
|---|---|
| With sodium carbonate In water; isopropyl alcohol at 30℃; for 6h; Temperature; | 94% |
-
-
98-59-9
p-toluenesulfonyl chloride

-
-
4696-76-8
kananmycin B

-
-
60929-91-1, 119818-17-6
1,3,2',6',3''-penta-N-p-toluenesulfonylkanamycin B

| Conditions | Yield |
|---|---|
| With sodium carbonate In ethanol; dichloromethane; water at 25℃; for 16h; Reagent/catalyst; Solvent; Temperature; | 92.3% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
4696-76-8
kananmycin B

-
-
64271-30-3
1,3,2',6',3''-penta-N-BOC-kanamycin B

| Conditions | Yield |
|---|---|
| In water; dimethyl sulfoxide at 60℃; for 6h; | 90% |

| Conditions | Yield |
|---|---|
| In phosphate buffer at 0℃; for 5h; pH=8.0; | 84% |
-
-
4696-76-8
kananmycin B

-
-
207857-15-6
1,3-di(tert-butyloxycarbonyl)-2-(trifluoromethylsulfonyl)guanidine

| Conditions | Yield |
|---|---|
| With triethylamine In 1,4-dioxane; water at 20℃; | 80% |
-
-
4696-76-8
kananmycin B

-
-
1268528-26-2
pentaazidokanamycin B

| Conditions | Yield |
|---|---|
| With triflic azide; triethylamine; zinc(II) chloride In methanol; water at 20℃; Inert atmosphere; | 76% |
| Stage #1: kananmycin B With copper(II) sulfate In water for 0.25h; Stage #2: With triethylamine In water for 0.333333h; Cooling with ice; Further stages; | 1.66 g |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
4696-76-8
kananmycin B

-
-
132260-09-4
3,2',6'-tris-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Stage #1: kananmycin B With zinc(II) acetate dihydrate In dimethyl sulfoxide at 21℃; Stage #2: di-tert-butyl dicarbonate In dimethyl sulfoxide at 21℃; for 5h; | 66% |
| With zinc diacetate 1.) DMSO, RT, overnight, 2.) 5 h; Yield given. Multistep reaction; |
-
-
4696-76-8
kananmycin B

-
-
207857-15-6
1,3-di(tert-butyloxycarbonyl)-2-(trifluoromethylsulfonyl)guanidine

-
-
320616-55-5
C73H127N15O30

| Conditions | Yield |
|---|---|
| With triethylamine In 1,4-dioxane; water at 20℃; for 72h; | 65% |

| Conditions | Yield |
|---|---|
| With pyridine |
-
-
104-15-4
toluene-4-sulfonic acid

-
-
4696-76-8
kananmycin B

-
-
60929-91-1, 119818-17-6
1,3,2',6',3''-penta-N-p-toluenesulfonylkanamycin B

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 2h; Ambient temperature; |
-
-
501-53-1
benzyl chloroformate

-
-
4696-76-8
kananmycin B

-
-
65269-36-5
N,N'-bis-benzyloxycarbonyl-O4-(3-benzyloxycarbonylamino-α-D-3-deoxy-glucopyranosyl)-O6-(2,6-bis-benzyloxycarbonylamino-α-D-2,6-dideoxy-glucopyranosyl)-1L-2-deoxy-streptamine

| Conditions | Yield |
|---|---|
| With sodium carbonate In water Yield given; |

| Conditions | Yield |
|---|---|
| at 37℃; aminoglycoside-4'-nucleotidyltransferaze of Bacillus brevis, MgCl2, potassium phosphate buffer (pH 6.0); |
-
-
4696-76-8
kananmycin B

-
A
-
2037-48-1
2-deoxystreptamine

-
B
-
60183-25-7
kanosamine

-
C
-
59433-00-0
2,6-dideoxy-2,6-diamino-D-glucose

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 0.5h; Product distribution; Heating; other aminoglycosides and their carbenicillin products; |

-
-
4696-76-8
kananmycin B

-
-
64879-71-6
4'',6''-O-cyclohexylidenekanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dimethylformamide / 2 h / Ambient temperature 2: dimethylformamide / Ambient temperature View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-19-6
C61H97N5O22S

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 65 percent / pyridine / 64 h View Scheme | |
| Multi-step reaction with 6 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 65 percent / pyridine / 64 h View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-28-7
1-C-(hydroxymethyl)-6''-deoxy-6''-chlorokanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 65 percent / pyridine / 64 h 5: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 6: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 7: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 8: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 9: 88.5 percent / lithium chloride / dimethylformamide / 7 h / 90 °C 10: CF3COOH / Ambient temperature View Scheme | |
| Multi-step reaction with 12 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 65 percent / pyridine / 64 h 7: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 8: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 9: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 10: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 11: 88.5 percent / lithium chloride / dimethylformamide / 7 h / 90 °C 12: CF3COOH / Ambient temperature View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132297-01-9
1-C-(hydroxymethyl)-6''-deoxy-6''-acetamidokanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 12 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 65 percent / pyridine / 64 h 5: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 6: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 7: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 8: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 9: 95 percent / LiN3 / dimethylformamide / 23 h / 65 °C 10: 1.) triphenylphosphine, 2.) 33percent aqueous NH3 / 1.) pyridine, RT, 2 h, 2.) 24 h 11: 73 percent / ethanol / 1.5 h / Ambient temperature 12: CF3COOH / Ambient temperature View Scheme | |
| Multi-step reaction with 14 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 65 percent / pyridine / 64 h 7: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 8: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 9: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 10: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 11: 95 percent / LiN3 / dimethylformamide / 23 h / 65 °C 12: 1.) triphenylphosphine, 2.) 33percent aqueous NH3 / 1.) pyridine, RT, 2 h, 2.) 24 h 13: 73 percent / ethanol / 1.5 h / Ambient temperature 14: CF3COOH / Ambient temperature View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132297-00-8
1-C-(hydroxymethyl)-6''-deoxy-6''-azidokanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 65 percent / pyridine / 64 h 5: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 6: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 7: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 8: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 9: 95 percent / LiN3 / dimethylformamide / 23 h / 65 °C 10: CF3COOH / Ambient temperature View Scheme | |
| Multi-step reaction with 12 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 65 percent / pyridine / 64 h 7: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 8: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 9: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 10: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 11: 95 percent / LiN3 / dimethylformamide / 23 h / 65 °C 12: CF3COOH / Ambient temperature View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-14-1
3,2',6',3''-tetrakis-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 62 percent / H2 / 10percent Pd/C / methanol; H2O / 3 h / 2280 Torr View Scheme | |
| Multi-step reaction with 6 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 62 percent / H2 / 10percent Pd/C / methanol; H2O / 3 h / 2280 Torr View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-10-7
3,2',6'-tris-N-(tert-butoxycarbonyl)-3''-N-(trifluoroacetyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 1.1: zinc(II) acetate dihydrate / dimethyl sulfoxide / 21 °C 1.2: 5 h / 21 °C 2.1: dimethyl sulfoxide / 5 h / 21 °C View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-17-4
1-C-(hydroxymethyl)-3,2',6',3''-tetrakis-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 62 percent / H2 / 10percent Pd/C / methanol; H2O / 3 h / 2280 Torr 5: 45 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.75 h / 90 °C 6: 81 percent / triethylamine / CHCl3 / 0.5 h / Heating 7: 80.5 percent / hydrogen / Raney nickel W2 catalyst / methanol / 3 h / 3040 Torr View Scheme | |
| Multi-step reaction with 9 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 62 percent / H2 / 10percent Pd/C / methanol; H2O / 3 h / 2280 Torr 7: 45 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.75 h / 90 °C 8: 81 percent / triethylamine / CHCl3 / 0.5 h / Heating 9: 80.5 percent / hydrogen / Raney nickel W2 catalyst / methanol / 3 h / 3040 Torr View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-12-9
1-N-<(benzyloxy)carbonyl>-3,2',6'-tris-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-15-2
1-deamino-1-nitro-3,2',6',3''-tetrakis-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 62 percent / H2 / 10percent Pd/C / methanol; H2O / 3 h / 2280 Torr 5: 45 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.75 h / 90 °C View Scheme | |
| Multi-step reaction with 7 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 62 percent / H2 / 10percent Pd/C / methanol; H2O / 3 h / 2280 Torr 7: 45 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.75 h / 90 °C View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-16-3
1-C-(hydroxymethyl)-1-deamino-1-nitro-3,2',6',3''-tetrakis-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 62 percent / H2 / 10percent Pd/C / methanol; H2O / 3 h / 2280 Torr 5: 45 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.75 h / 90 °C 6: 81 percent / triethylamine / CHCl3 / 0.5 h / Heating View Scheme | |
| Multi-step reaction with 8 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 62 percent / H2 / 10percent Pd/C / methanol; H2O / 3 h / 2280 Torr 7: 45 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.75 h / 90 °C 8: 81 percent / triethylamine / CHCl3 / 0.5 h / Heating View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-27-6
C44H80N6O20

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 65 percent / pyridine / 64 h 5: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 6: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 7: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 8: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 9: 95 percent / LiN3 / dimethylformamide / 23 h / 65 °C 10: 1.) triphenylphosphine, 2.) 33percent aqueous NH3 / 1.) pyridine, RT, 2 h, 2.) 24 h View Scheme | |
| Multi-step reaction with 12 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 65 percent / pyridine / 64 h 7: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 8: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 9: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 10: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 11: 95 percent / LiN3 / dimethylformamide / 23 h / 65 °C 12: 1.) triphenylphosphine, 2.) 33percent aqueous NH3 / 1.) pyridine, RT, 2 h, 2.) 24 h View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-24-3
1-C-(hydroxymethyl)-6''-deoxy-6''-chloro-1,3,2',6',3''-pentakis-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 65 percent / pyridine / 64 h 5: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 6: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 7: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 8: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 9: 88.5 percent / lithium chloride / dimethylformamide / 7 h / 90 °C View Scheme | |
| Multi-step reaction with 11 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 65 percent / pyridine / 64 h 7: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 8: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 9: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 10: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 11: 88.5 percent / lithium chloride / dimethylformamide / 7 h / 90 °C View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-13-0
1-N-<(benzyloxy)carbonyl>-3,2',6',3''-tetrakis-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C View Scheme | |
| Multi-step reaction with 5 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C View Scheme |
-
-
4696-76-8
kananmycin B

-
-
132260-26-5
1-C-(hydroxymethyl)-6''-deoxy-6''-acetamido-1,3,2',6',3''-pentakis-N-(tert-butoxycarbonyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 73 percent / copper(II) acetate monohydrate / dimethylformamide / 1.5 h 3: dioxane; methanol / 0.5 h / 40 °C 4: 65 percent / pyridine / 64 h 5: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 6: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 7: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 8: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 9: 95 percent / LiN3 / dimethylformamide / 23 h / 65 °C 10: 1.) triphenylphosphine, 2.) 33percent aqueous NH3 / 1.) pyridine, RT, 2 h, 2.) 24 h 11: 73 percent / ethanol / 1.5 h / Ambient temperature View Scheme | |
| Multi-step reaction with 13 steps 1: 1.) Zn(OAc)2*2H2O / 1.) DMSO, RT, overnight, 2.) 5 h 2: 98 percent / dimethylsulfoxide / 1.5 h / Ambient temperature 3: 79 percent / dimethylformamide / 15 h / Ambient temperature 4: 33percent aqueous NH3 / tetrahydrofuran / 39 h / Ambient temperature 5: dioxane; methanol / 0.5 h / 40 °C 6: 65 percent / pyridine / 64 h 7: 76 percent / H2 / 10percent Pd/C / methanol / 2280 Torr 8: 40 percent / 3-chloroperbenzoic acid, 3-tert-butyl-5-methyl-4-hydroxyphenyl sulfide / 1,2-dichloro-ethane / 0.5 h / Heating 9: 72 percent / triethylamine / CHCl3 / 1.5 h / Heating 10: 1.) H2, (BOC)2O / 1.) Raney nickel W2 / 1.) methanol, 4 atm, 4 h, 2.) 20 h 11: 95 percent / LiN3 / dimethylformamide / 23 h / 65 °C 12: 1.) triphenylphosphine, 2.) 33percent aqueous NH3 / 1.) pyridine, RT, 2 h, 2.) 24 h 13: 73 percent / ethanol / 1.5 h / Ambient temperature View Scheme |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View