LIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:52488-36-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:52488-36-5
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryWuhan Fortuna Chemical Co.,Ltd
Unique advantages for 4-Bromoindole Cas 52488-36-5 Guaranteed the purity High quality & competitive price Quality control Fast feedback Prompt shipment Appearance:Liquid Storage:0-6°C Package:250kg/drum or as you required Applica
Cas:52488-36-5
Min.Order:1 Kilogram
FOB Price: $30.0
Type:Trading Company
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hubei DiBo chemical co., LTD
Name:4-Bromoindole CAS NO: 52488-36-5 Grade:Medical scientific research and export Molecular formula:C7H5BrN2 Molecular weight:197.032 Product Quality 12 years of chemical raw materials Mature operation of the industry System stability
Cas:52488-36-5
Min.Order:25 Kilogram
FOB Price: $1.0 / 2.0
Type:Other
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 6 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:52488-36-5
Min.Order:1 Kilogram
FOB Price: $2000.0
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:52488-36-5
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Product Name 4-Bromoindole / 4-bromo-1H-indole CAS 52488-36-5 MF C8H6BrN Type Pharmaceutical Intermediates Content Min 98.0% A
Cas:52488-36-5
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquiryHubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:powder Storage:Store in sealed containers at cool & dry pla
Cas:52488-36-5
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:52488-36-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:52488-36-5
Min.Order:1 Kilogram
FOB Price: $24.0 / 30.0
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:52488-36-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Wuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Cas:52488-36-5
Min.Order:10 Kilogram
Negotiable
Type:Trading Company
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Afine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:52488-36-5
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:52488-36-5
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquirySHANGHAI SYSTEAM BIOCHEM CO., LTD
We are one of a few suppliers that can offer custom synthesis service of this product We are specialized in custom synthesis, chemical/pharmaceutical/ pesticides outsourcing and contract research. We are committed to prov
Cas:52488-36-5
Min.Order:100 Gram
FOB Price: $100.0 / 2000.0
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: 4-Bromoindole CAS No.:52488-36-5 Molecule Formula:C7H5BrN2 Molecule Weight:197.03 Purity: 98.0% Package: 200kg/drum Description:Light yellow liquid Manufacture Standards:Enterprise Standard TESTING IT
Cas:52488-36-5
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
4-Bromoindole Basic information Product Name: 4-Bromoindole Synonyms: 4-BROMO-1H-INDOLE,4-BROMOINDOLE;4-BROMO-1H-INDAZOLE;4-BROMO-INDOLE pure;4-Bromoindole ,95%;4-Bromo-1H-indole , TECH;1H-Indole,4-broMo-;4-Bromoindole 96%;4-BROMOINDOLE CAS:
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:52488-36-5
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryHangzhou Huarong Pharm Co., Ltd.
We Huarong Pharm can provide Customized Synthesis & Process R&D & APIs and intermediates Production & Quality Research & Registration Application, especially our GMP validation service which complies with SFDA, FDA, WHO and EU EMPA. O
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:52488-36-5
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Wuhu Nuowei Chemistry Technologies Co., Ltd.
Wuhu Nuovo Chemical Technology Co., Ltd. was established in August 2014, mainly engaged in the development, production and sales of ionic liquids, ribose, nucleosides, nucleotides and related chemicals; Products are mainly used in new energy, new ma
Synthetic route
-
-
90481-72-4
4-bromo-1-[(4-methylphenyl)sulfonyl]-1H-indole

-
-
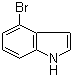
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol for 12h; Heating; | 98% |
-
-
1180007-97-9
4-bromo-1-(trifluoromethylsulfonyl)indole

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 100℃; for 12h; | 96% |
-
-
86626-38-2
4-bromo-2,3-dihydro-1H-indole

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium carbonate In ethyl acetate at 120℃; for 24h; Sealed tube; Green chemistry; | 95% |
| With TEMPOL; oxygen; copper(l) chloride at 50 - 60℃; for 4.5h; Inert atmosphere; | 95% |
| With oxygen; sodium t-butanolate In dimethyl sulfoxide at 60℃; under 760.051 Torr; for 4h; | 48% |
-
-
1196981-04-0
1-(4-bromo-1H-indol-1-yl)-2,2-dimethylpropan-1-one

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With water; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran for 3h; Reflux; | 94% |
-
-
101909-45-9
4-bromo-1-methoxycarbonylindole

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol for 1h; Heating; | 88.5% |
-
-
90481-68-8
1-bromo-2-ethenyl-3-nitrobenzene

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With carbon monoxide; triphenylphosphine; palladium diacetate In acetonitrile under 3040 Torr; for 15h; Heating; | 84% |
| With carbon monoxide; triphenylphosphine; palladium diacetate In acetonitrile under 3040 Torr; for 15h; Product distribution; Heating; other catalysts, phosphine, bases, solvents; palladium-catalyzed reductive heteroannulation of 2-nitrostyrenes in the presence of CO; | 84% |
| Multi-step reaction with 2 steps 1: 96.6 percent / Fe / acetic acid; ethanol / 5 h / Heating 2: 46.3 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 22 h / 100 °C / sealed tube View Scheme | |
| Multi-step reaction with 4 steps 1: 96.6 percent / Fe / acetic acid; ethanol / 5 h / Heating 2: pyridine / 0.33 h / Heating 3: 76.7 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 18 h / Heating 4: 98 percent / 20percent aq. NaOH / methanol / 12 h / Heating View Scheme |
-
-
67-56-1
methanol

-
-
201230-82-2
carbon monoxide

-
-
90481-68-8
1-bromo-2-ethenyl-3-nitrobenzene

-
A
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With 1,3-bis-(diphenylphosphino)propane; palladium diacetate In N,N-dimethyl-formamide under 3040 Torr; for 22h; Heating; | A 76% B 17% |

-
-
1244651-69-1
N-(3-bromo-2-(2-(trimethylsilyl)ethynyl)phenyl)-2-hydroxy-2-methylpropanamide

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In N,N-dimethyl-formamide at 140℃; for 56h; Inert atmosphere; | 75% |
| With sodium hydroxide In N,N-dimethyl-formamide at 140℃; for 5h; Inert atmosphere; | 75% |

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With potassium fluoride; tris-(dibenzylideneacetone)dipalladium(0); t-BuBrettPhos; potassium bromide In 1,4-dioxane at 130℃; for 16h; Inert atmosphere; | 74% |
-
-
86626-38-2
4-bromo-2,3-dihydro-1H-indole

-
-
98-95-3
nitrobenzene

-
A
-
52488-36-5
4-bromo-1H-indole

-
B
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| With nickel-nitrogen-doped carbon framework In water at 145℃; for 18h; Inert atmosphere; Sealed tube; Green chemistry; | A 70% B 74% |

-
-
109277-82-9
2-(2-amino-6-bromophenyl)ethan-1-ol

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| tris(triphenylphosphine)ruthenium(II) chloride In toluene for 6h; Heating; | 73% |
| tris(triphenylphosphine)ruthenium(II) chloride In toluene for 6h; Rate constant; Heating; | 73% |
| With C72H79O35Ru4; o-phenylenebis(diphenylphosphine); oxygen In toluene at 130℃; for 4h; Inert atmosphere; | 71% |
-
-
105205-47-8
[(E)-2-(2-Bromo-6-nitro-phenyl)-vinyl]-dimethyl-amine

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With zinc In acetic acid at 85℃; for 2h; | 70% |
| With acetic acid; zinc In methanol; dichloromethane Heating; | 67% |

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With zinc In acetic acid Reduction; cyclization; | 70% |
-
-
101909-43-7
4-bromo-1H-indole-3-carboxylic acid methyl ester

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol for 1.5h; Heating; | 69.1% |
| With sodium hydroxide In methanol for 1.5h; Heating; | 60.5% |
-
-
86626-38-2
4-bromo-2,3-dihydro-1H-indole

-
-
100-00-5
4-chlorobenzonitrile

-
A
-
52488-36-5
4-bromo-1H-indole

-
B
-
106-47-8
4-chloro-aniline

| Conditions | Yield |
|---|---|
| With nickel-nitrogen-doped carbon framework In water at 145℃; for 18h; Inert atmosphere; Sealed tube; Green chemistry; | A 53% B 52% |

-
-
90481-69-9
3-bromo-2-ethenylaniline

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); p-benzoquinone; lithium chloride In tetrahydrofuran at 100℃; for 22h; sealed tube; | 46.3% |
| Multi-step reaction with 3 steps 1: pyridine / 0.33 h / Heating 2: 76.7 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 18 h / Heating 3: 98 percent / 20percent aq. NaOH / methanol / 12 h / Heating View Scheme |
-
-
16732-64-2
4-bromo-1H-indole-2-carboxylic acid

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With quinoline; copper(I) bromide at 250℃; |
-
-
85355-50-6
(2-Bromo-6-nitro-phenyl)-acetaldehyde

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium acetate; iron; acetic acid In ethanol; water for 2h; Heating; Yield given; |
-
-
85355-40-4
Acetic acid 2-(2-bromo-6-nitro-phenyl)-1-chloro-ethyl ester

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium acetate; iron; acetic acid In ethanol; water for 2h; Heating; |
-
-
55289-35-5
2-bromo-6-nitrotoluene

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromo-6-nitrotoluene; N,N-dimethyl-formamide dimethyl acetal With pyrrolidine In N,N-dimethyl-formamide at 110℃; for 5h; Stage #2: With acetic acid; zinc at 85℃; for 3h; | 25.2 g |
-
-
603-83-8
3-nitro-o-tolylamine

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: aq. HBr / 0.33 h / Heating 1.2: aq. NaNO2 / 0.25 h / 0 - 5 °C 1.3: 90 percent / CuBr; aq. HBr / 0.33 h / 100 °C 2.1: pyrrolidine / dimethylformamide / 5 h / 110 °C 2.2: 25.2 g / aq. HOAc; zinc / 3 h / 85 °C View Scheme | |
| Multi-step reaction with 6 steps 2: 98 percent / Br2, benzoyl peroxide / CCl4 / 14 h / Heating; Irradiation 3: CHCl3 / 0.17 h / Heating 4: 85.1 percent / Et3N / CH2Cl2 / 1 h / Ambient temperature 5: 96.6 percent / Fe / acetic acid; ethanol / 5 h / Heating 6: 46.3 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 22 h / 100 °C / sealed tube View Scheme | |
| Multi-step reaction with 8 steps 2: 98 percent / Br2, benzoyl peroxide / CCl4 / 14 h / Heating; Irradiation 3: CHCl3 / 0.17 h / Heating 4: 85.1 percent / Et3N / CH2Cl2 / 1 h / Ambient temperature 5: 96.6 percent / Fe / acetic acid; ethanol / 5 h / Heating 6: pyridine / 0.33 h / Heating 7: 76.7 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 18 h / Heating 8: 98 percent / 20percent aq. NaOH / methanol / 12 h / Heating View Scheme |
-
-
55289-35-5
2-bromo-6-nitrotoluene

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: CuI; DMF / 0.33 h / 180 °C / 6000.6 - 7500.75 Torr / microwave irradiation 2: 67 percent / Zn; AcOH / methanol; CH2Cl2 / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 1.) Triton B, 2.) Zn, CaCl2 / 1.) Me2SO, 95 deg C, 1 h, 2.) H2O, reflux 2: 73 percent / RuCl2(PPh3)3 / toluene / 6 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: pyrrolidine / dimethylformamide / 110 °C 2: 70 percent / Zn / acetic acid / 2 h / 85 °C View Scheme |
-
-
942-24-5
3-methoxycarbonylindole

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: thallium(III) trifluoroacetate / trifluoroacetic acid / 2 h / 20 °C 1.2: 63 percent / CuBr2 / dimethylformamide / 1 h / 120 °C 2.1: 69.1 percent / aq. NaOH / methanol / 1.5 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 1.) TTFA, 2.) CuBr2 / 1.) TFA, room temp., 2 h; 2.) DMF, 120 deg C, 1 h 2: 60.5 percent / 40percent NaOH / methanol / 1.5 h / Heating View Scheme |
-
-
59255-95-7
2-bromo-6-nitroaniline

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: H2SO4, isoamyl nitrite / ethanol / 1 h / 0 °C 2: LiCl, HCl, CuCl*2H2O / acetone; H2O / 1.) 2 h, 3 to 10 deg C, 2.) 2 h, room temp., 3.) 2 h, room temp. 3: Fe, HOAc, sodium acetate dihydrate / ethanol; H2O / 2 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: H2SO4, isoamyl nitrite / ethanol / 1 h / 0 °C 2: LiCl, HCl, CuCl*2H2O / acetone; H2O / 1.) 2 h, 3 to 10 deg C, 2.) 2 h, room temp., 3.) 2 h, room temp. 3: Fe, HOAc, sodium acetate dihydrate / ethanol; H2O / 2 h / Heating View Scheme |
-
-
85355-33-5
2-Bromo-6-nitro-benzenediazonium; hydrogen sulfate

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: LiCl, HCl, CuCl*2H2O / acetone; H2O / 1.) 2 h, 3 to 10 deg C, 2.) 2 h, room temp., 3.) 2 h, room temp. 2: Fe, HOAc, sodium acetate dihydrate / ethanol; H2O / 2 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: LiCl, HCl, CuCl*2H2O / acetone; H2O / 1.) 2 h, 3 to 10 deg C, 2.) 2 h, room temp., 3.) 2 h, room temp. 2: Fe, HOAc, sodium acetate dihydrate / ethanol; H2O / 2 h / Heating View Scheme |
-
-
58579-54-7
3-bromo-2-(bromomethyl)-1-nitrobenzene

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: CHCl3 / 0.17 h / Heating 2: 85.1 percent / Et3N / CH2Cl2 / 1 h / Ambient temperature 3: 96.6 percent / Fe / acetic acid; ethanol / 5 h / Heating 4: 46.3 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 22 h / 100 °C / sealed tube View Scheme | |
| Multi-step reaction with 6 steps 1: CHCl3 / 0.17 h / Heating 2: 85.1 percent / Et3N / CH2Cl2 / 1 h / Ambient temperature 3: 96.6 percent / Fe / acetic acid; ethanol / 5 h / Heating 4: pyridine / 0.33 h / Heating 5: 76.7 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 18 h / Heating 6: 98 percent / 20percent aq. NaOH / methanol / 12 h / Heating View Scheme |
-
-
90481-71-3
N-tosyl-3-bromo-2-ethenylaniline

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 76.7 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 18 h / Heating 2: 98 percent / 20percent aq. NaOH / methanol / 12 h / Heating View Scheme |
-
-
90481-67-7
(2-Bromo-6-nitro-benzyl)-triphenyl-phosphonium; bromide

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 85.1 percent / Et3N / CH2Cl2 / 1 h / Ambient temperature 2: 96.6 percent / Fe / acetic acid; ethanol / 5 h / Heating 3: 46.3 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 22 h / 100 °C / sealed tube View Scheme | |
| Multi-step reaction with 5 steps 1: 85.1 percent / Et3N / CH2Cl2 / 1 h / Ambient temperature 2: 96.6 percent / Fe / acetic acid; ethanol / 5 h / Heating 3: pyridine / 0.33 h / Heating 4: 76.7 percent / p-benzoquinone, LiCl, PdCl2(CH3CN)2 / tetrahydrofuran / 18 h / Heating 5: 98 percent / 20percent aq. NaOH / methanol / 12 h / Heating View Scheme |
-
-
118665-02-4
2-(2-bromo-6-nitrophenyl)ethan-1-ol

-
-
52488-36-5
4-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Zn/CaCl2/H2O / ethanol / Heating 2: 73 percent / RuCl2(PPh3)3 / toluene / 6 h / Heating View Scheme |
-
-
52488-36-5
4-bromo-1H-indole

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
98600-34-1
4-bromo-1H-indole-3-carbaldehyde

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 20℃; for 1h; Vilsmeier-Hack formylation; | 100% |
| Stage #1: N,N-dimethyl-formamide With trichlorophosphate Cooling with ice; Stage #2: 4-bromo-1H-indole In N,N-dimethyl-formamide at 20℃; Cooling with ice; Stage #3: With water; potassium hydroxide In N,N-dimethyl-formamide Reflux; | 100% |
| Stage #1: 4-bromo-1H-indole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 25℃; Stage #2: With potassium hydroxide In water Reflux; | 100% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
73183-34-3
bis(pinacol)diborane

-
-
388116-27-6
indole-4-boronic acid pinacol ester

| Conditions | Yield |
|---|---|
| With potassium acetate; dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2 In dimethyl sulfoxide at 90℃; for 4h; | 100% |
| With potassium acetate; dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2 In dimethyl sulfoxide at 90℃; for 4h; | 100% |
| With tris-(dibenzylideneacetone)dipalladium(0); potassium acetate; XPhos In 1,4-dioxane at 110℃; for 20h; Miyaura borylation; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 20℃; for 3h; | 100% |
| Stage #1: 4-bromo-1H-indole With sodium hydride In tetrahydrofuran at 0℃; for 0.5h; Schlenk technique; Stage #2: methyl iodide In tetrahydrofuran at 0 - 20℃; Schlenk technique; | 100% |
| With sodium hydride In tetrahydrofuran at 20℃; for 4h; | 98% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
676448-17-2
4-bromoindole-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With dmap In tetrahydrofuran for 0.25h; | 100% |
| In dichloromethane at 20℃; for 8h; | 99% |
| With dmap In dichloromethane at 20℃; for 8h; Inert atmosphere; | 98% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
86864-60-0
2-(tert-butyldimethylsilyloxy)ethyl bromide

-
-
603305-12-0
4-bromo-1-[3-(tert-butyldimethylsilyloxy)prop-1-yl]-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1H-indole With sodium hydride In DMF (N,N-dimethyl-formamide) Stage #2: 2-(tert-butyldimethylsilyloxy)ethyl bromide In DMF (N,N-dimethyl-formamide) at 0 - 20℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With iodine; potassium iodide In ethanol; water at 45℃; for 12.0833h; | 99.5% |
| With iodine; potassium iodide In ethanol; water at 20 - 60℃; | 92% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
479423-30-8
N-(tert-butoxycarbonyl)-α-(phenylsulfonyl)-4-bromobenzylamine

| Conditions | Yield |
|---|---|
| With potassium fluoride; C46H34I4O6 In m-xylene at 20℃; for 12h; Friedel-Crafts Alkylation; Schlenk technique; enantioselective reaction; | 99% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
155396-71-7
tert-butyl N-(phenyl(phenylsulfonyl)methyl)carbamate

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate In toluene at 20℃; for 48h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; regioselective reaction; | 99% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
64-17-5
ethanol

-
-
201230-82-2
carbon monoxide

-
-
50614-84-1
ethyl indole-4-carboxylate

| Conditions | Yield |
|---|---|
| With 1,1'-bis-(diphenylphosphino)ferrocene; bis(benzonitrile)palladium(II) dichloride; triethylamine at 130℃; under 18751.5 Torr; for 20h; | 98% |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide; mineral oil | 98% |
| Stage #1: 4-bromo-1H-indole With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.5h; Stage #2: 1-Bromopentane In N,N-dimethyl-formamide at 0 - 20℃; for 6h; | 98% |
| Stage #1: 4-bromo-1H-indole With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.5h; Stage #2: 1-Bromopentane In N,N-dimethyl-formamide at 0 - 20℃; for 6h; | 98% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
421-83-0
trifluoromethane sulfonyl chloride

| Conditions | Yield |
|---|---|
| With 1,2,2,3,4,4-hexamethylphosphetane 1-oxide; phenylsilane In 1,4-dioxane at 40℃; Schlenk technique; Sealed tube; Inert atmosphere; | 98% |
| With trimethylphosphane In tetrahydrofuran at -78 - 20℃; for 52h; regioselective reaction; | 74% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate In ethanol; water at 80℃; for 4h; Suzuki-Miyaura Coupling; | 98% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
1216305-58-6
4-bromo-3-chloro-1H-indole

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide In N,N-dimethyl-formamide at 20℃; for 12h; Inert atmosphere; | 97% |
| With 1,3-dichloro-5,5-dimethylhydantoin In 1,4-dioxane at 25℃; for 3h; | 88% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
1593-60-8
p-toluenesulfonylhydroxylamine

| Conditions | Yield |
|---|---|
| With 1-hydroxy-pyrrolidine-2,5-dione; iodine In butan-1-ol at 120℃; for 15h; Inert atmosphere; Sealed tube; regioselective reaction; | 97% |

| Conditions | Yield |
|---|---|
| In water; acetic acid for 48h; Ambient temperature; | 96.3% |
| With acetic acid In water at 0 - 25℃; |

-
-
52488-36-5
4-bromo-1H-indole

-
-
201230-82-2
carbon monoxide

-
-
14135-70-7
N-(1-methyl-2-phenylethyl)piperazine

| Conditions | Yield |
|---|---|
| With 1,1'-bis-(diphenylphosphino)ferrocene; bis(benzonitrile)palladium(II) dichloride; triethylamine In toluene at 130℃; under 18751.5 Torr; for 20h; | 96% |

-
-
52488-36-5
4-bromo-1H-indole

-
-
75-36-5
acetyl chloride

-
-
195874-03-4
1-(4-bromo-1H-indol-3-yl)ethan-1-one

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In toluene | 96% |
| With tin(IV) chloride In toluene at 0℃; for 2h; Friedel-Crafts reaction; | 95% |
| With tin(IV) chloride In toluene at 0℃; for 2h; | 75% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
16629-19-9
thiophene-2-sulfonyl chloride

-
-
1001394-83-7
4-bromo-1-(2-thienylsulfonyl)-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In water for 25h; | 96% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
13154-24-0
triisopropylsilyl chloride

-
-
412048-44-3
4-Bromo-1-(tri-isopropylsilyl)-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1H-indole With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; for 0.5h; Stage #2: triisopropylsilyl chloride In tetrahydrofuran; mineral oil at 0 - 20℃; | 96% |
| With sodium hydride In tetrahydrofuran at 0 - 20℃; | 88% |
| With sodium hydride In tetrahydrofuran at 20℃; Inert atmosphere; Cooling with ice; | 82% |
| With NaH In dichloromethane; N,N-dimethyl-formamide | 3.44 g (63%) |
-
-
52488-36-5
4-bromo-1H-indole

-
-
98-09-9
benzenesulfonyl chloride

-
-
412048-77-2
1‐(benzenesulfonyl)‐4‐bromo‐1H‐indole

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium hydroxide In dichloromethane at 20℃; for 1h; | 95% |
| Stage #1: 4-bromo-1H-indole With sodium hydride In tetrahydrofuran at 0℃; for 1h; Stage #2: benzenesulfonyl chloride In tetrahydrofuran at 20℃; for 2h; | 93% |
| Stage #1: 4-bromo-1H-indole With sodium hydride In tetrahydrofuran at 20℃; for 1h; Stage #2: benzenesulfonyl chloride In tetrahydrofuran for 1h; | 87% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
6482-24-2
2-Bromoethyl methyl ether

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1H-indole With sodium hydride In 1,2-dimethoxyethane; dimethyl sulfoxide at 20℃; for 0.5h; Stage #2: 2-Bromoethyl methyl ether In 1,2-dimethoxyethane; dimethyl sulfoxide at 20℃; | 95% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
42899-76-3
pyridine-3-sulfonyl chloride hydrochloride

-
-
1001394-86-0
4-bromo-1-(pyridine-3-ylsulfonyl)-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In water for 0.75h; | 95% |

| Conditions | Yield |
|---|---|
| With C20H24Cl2N2O2Zr In ethanol at 25 - 27℃; for 0.416667h; | 95% |

| Conditions | Yield |
|---|---|
| With bis(η3-allyl-μ-chloropalladium(II)); N,N,N′,N′-tetra(diphenylphosphinomethyl)-1,2-ethylenediamine; potassium carbonate In N,N-dimethyl acetamide; butan-1-ol at 110℃; for 20h; Suzuki Coupling; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 70℃; for 0.5h; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1H-indole With sodium hydride In tetrahydrofuran; N,N-dimethyl-formamide; mineral oil at 0℃; for 1h; Inert atmosphere; Stage #2: bromethyl methyl ether In tetrahydrofuran; N,N-dimethyl-formamide; mineral oil at 0 - 23℃; for 1h; | 95% |
-
-
52488-36-5
4-bromo-1H-indole

-
-
618-41-7
Benzenesulfinic acid

-
-
945000-87-3
4-bromo-3-(phenylthio)-1H-indole

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; toluene-4-sulfonic acid In 1,2-dichloro-ethane at 80℃; for 12h; regioselective reaction; | 95% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 Xi
Xi