Wuhan Fortuna Chemical Co.,Ltd
1.High quality : the purity is 99% min . through multiple producing procedures. 2.Competitive price : low price because of our skilled production technolpgy ,save the production cost at most , and give big profit room to our customers
Hangzhou Think Chemical Co. Ltd
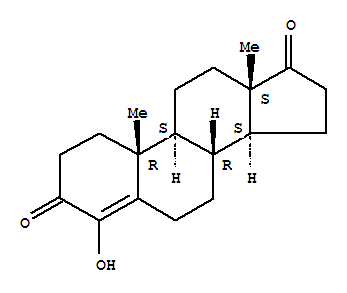
Formestane CAS No.:566-48-3 Name: Formestane Synonyms: 4-Hydroxyandrost-4-ene-3,17-dione Molecular Structure Mol
Cas:566-48-3
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Other
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:566-48-3
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryShanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:566-48-3
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
LIDE PHARMACEUTICALS LIMITED
LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc.
Cas:566-48-3
Min.Order:1 Kilogram
FOB Price: $0.9 / 1.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:566-48-3
Min.Order:1 Kilogram
FOB Price: $15.0 / 50.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Chemical Name: Formestane CAS No.: 566-48-3 Molecular Fomula: C20H28O2 Molecular weight: 300.4351 Appearance:colorless liquid Assay:99%min Appearance: Colorless Liquid Storage:Preserve in well-closed, light-resistant and airtight containers. P
Hubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best s
Cas:566-48-3
Min.Order:10 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:566-48-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates, steroids, pharmaceutical i
Cas:566-48-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Terui OP New Material Technology Co., Ltd.
We generally use secure shipping methods that do not require you to do any customs clearance. Free shipping to your address without worrying about any customs issues. We have a professional customs clearance team to 100% guarantee the safe delivery o
Cas:566-48-3
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:566-48-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryWuhan Wonda Pharm Limited
1.High Quality: Quality is life. Quality is the most important element for all goods. We have a lab doing research in Wuhan China and produce sarms in bulk quantity. We have 8 years experience making all kinds of sarms. And all our old customers
Henan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:566-48-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryTriumph International Development Limilted
Main Service 1.Own made fine chemical products 2.Out sourcing and quality controlling service in China 3. COM for Chemical synthesis 4.Lab custom synthesis of API and intermadiates Quality Assurance 1. NMR,HPLC and COA can be supplied 2. Fr
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Wuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:566-48-3
Min.Order:0 Metric Ton
Negotiable
Type:Other
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:566-48-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:Needles Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum;200kg/drum as per your request Application:An antitumor drug. An aromatase inhibi
Cas:566-48-3
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryTaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
HANWAYS CHEMPHARM CO.,LIMITED
Hanways Chempharm Co., Limited, the former is Hubei Hanways Pharchem CO.,Limited, set up in 2009 in Wuhan, China. We specialize in sourcing and supplying APIs, pharmaceutical intermediates, and fine chemicals for worldwide markets. The founder ha
Siwei Development Group Ltd.
Product name: Fomestane CAS No.:566-48-3 Molecule Formula:C20H28O2 Molecule Weight:300.44 Purity: 99.0% Package: 25kg/drum Description:White or off-white powder Manufacture Standards:Enterprise Standard TESTING ITEMS
Cas:566-48-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:566-48-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:566-48-3
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Synthetic route
-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid; silica gel; acetic acid at 70℃; for 0.05h; | 86% |
| With boron trifluoride diethyl etherate In benzene for 19h; Ambient temperature; | 47% |
| With formic acid for 0.75h; Heating; | 45% |
| Multi-step reaction with 2 steps 1: 62 percent / aq. sodium hydroxide / 3 h / Heating 2: 53 percent / hydrochloric acid / dioxane / 24 h / Heating View Scheme |
-
-
180303-17-7
3-hydroxy-5α-androst-2-ene-4,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With sodium In methanol for 1h; Ambient temperature; | 80% |
| With sodium In methanol at 20℃; for 1h; | 80% |
| With sodium methylate In methanol |

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With sodium In methanol for 1h; Ambient temperature; | 80% |
-
-
434939-05-6
3β,4α-dihydroxy-5β-androstan-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Stage #1: 3β,4α-dihydroxy-5β-androstan-17-one With dimethyl sulfoxide; triethylamine; trifluoroacetic anhydride In dichloromethane at -60℃; for 3h; Stage #2: With sodium In methanol at 20℃; for 3h; | 80% |
| Stage #1: 3β,4α-dihydroxy-5β-androstan-17-one With dimethyl sulfoxide; triethylamine; trifluoroacetic anhydride at -60℃; for 3.25h; Swern oxidation; Stage #2: With sodium methylate In methanol | |
| Multi-step reaction with 2 steps 1: TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: 80 percent / Na / methanol / 1 h / 20 °C View Scheme |
-
-
434939-09-0
3-hydroxy-5β-androst-2-ene-4,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With sodium In methanol at 20℃; for 1h; | 80% |
-
-
501433-34-7
5α/β-androst-3-en-17-one

-
A
-
63-05-8
Androstenedione

-
B
-
1229-12-5
androstane-3,17-dione

-
C
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multistep reaction; | A n/a B n/a C 54% |

-
-
20986-46-3
4-methoxy-4-androstene-3,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 1,4-dioxane for 24h; Heating; | 53% |
-
-
64-19-7
acetic acid

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
2241-85-2
2α-acetoxyandrost-4-ene-3,17-dione

-
B
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
C
-
571-17-5
2alpha-Hydroxyandrost-4-ene-3,17-dione

-
D
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid a) RT, 4 h, b) 4 deg C, 12 h; Further byproducts given; | A 23% B 52% C 0.58 g D 0.7 g |

-
-
64-19-7
acetic acid

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
B
-
571-17-5
2alpha-Hydroxyandrost-4-ene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140223-19-4
4α-acetoxy-5α-hydroxyandrostane-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid a) RT, 4h,b) 4 deg C, 12 h; Further byproducts given; | A 52% B 0.58 g C 0.7 g D 3% |

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
2241-85-2
2α-acetoxyandrost-4-ene-3,17-dione

-
B
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
C
-
571-17-5
2alpha-Hydroxyandrost-4-ene-3,17-dione

-
D
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid a) RT, 4 h, b) 4 deg C, 12 h; Further byproducts given; | A 23% B 52% C 0.58 g D 0.7 g |

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
2241-85-2
2α-acetoxyandrost-4-ene-3,17-dione

-
B
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140223-19-4
4α-acetoxy-5α-hydroxyandrostane-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid a) RT, 4h , b) 4 deg C, 12 h; Further byproducts given; | A 23% B 52% C 0.7 g D 3% |

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
B
-
571-17-5
2alpha-Hydroxyandrost-4-ene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140223-19-4
4α-acetoxy-5α-hydroxyandrostane-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid a> RT, 4 h, b) 4 deg C, 12 h; Further byproducts given; | A 52% B 0.58 g C 0.7 g D 3% |


| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid for 18h; Ambient temperature; | 50% |
| With pyridine; hydrogen fluoride at 55℃; |
-
-
64-19-7
acetic acid

-
-
88509-24-4
4,5-epoxyandrost-9(11)-ene-3,17-dione

-
A
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
B
-
140111-67-7
2α-hydroxyandrost-4,9(11)-diene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140111-66-6
2α-acetoxyandrost-4,9(11)-diene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid a) RT, 4h, b) 4 deg C, 12 h; | A 13.6% B n/a C 390 mg D 9% |

-
-
88509-24-4
4,5-epoxyandrost-9(11)-ene-3,17-dione

-
A
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
B
-
140111-67-7
2α-hydroxyandrost-4,9(11)-diene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140111-66-6
2α-acetoxyandrost-4,9(11)-diene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid a) RT, 4 h, b) 4 deg C, 12h; | A 13.6% B n/a C 390 mg D 9% |
| With sulfuric acid In acetic acid a) RT, 4 h, b) 4 deg C, 12 h; | A 13.6% B n/a C 390 mg D 9% |
| With sulfuric acid In acetic acid a) RT, 4h, b) 4 deg C, 12 h; | A 13.6% B n/a C 390 mg D 9% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; sulfuric acid; dihydrogen peroxide 1a) 0 to 4 deg C, 30 min, MeOH, 1b) 5 to 7 deg C, 22 h, MeOH, 2) HOAc, RT, 45 min; Yield given. Multistep reaction; | |
| Multi-step reaction with 4 steps 1: 60 percent / Zn; AcOH / 0.25 h / 118 °C 2: 96 percent / aq. H2O2; HCO2H / 1 h / 20 °C 3: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 4: NaOMe / methanol View Scheme | |
| Multi-step reaction with 2 steps 1: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme | |
| Multi-step reaction with 2 steps 1: osmium tetroxide, 35percent H2O2 / 2-methyl-propan-2-ol / 72 h / Ambient temperature 2: potassium hydroxide / methanol / 0.17 h / 55 °C View Scheme | |
| Multi-step reaction with 2 steps 1: osmium(VIII) oxide; dihydrogen peroxide / tert-butyl alcohol / 72 h / 20 °C 2: potassium hydroxide / methanol / 0.17 h / 55 °C View Scheme |
-
-
110267-65-7
4ξ,5-dihydroxy-5ξ-androstane-3,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol at 55℃; for 0.166667h; Yield given; | |
| With potassium hydroxide In methanol at 55℃; for 0.166667h; | 0.452 g |
-
-
14935-81-0
5α-androst-3-en-17β-ol

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 96 percent / aq. H2O2; HCO2H / 1 h / 20 °C 2: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 3: NaOMe / methanol View Scheme | |
| Multi-step reaction with 3 steps 1: 96 percent / H2O2; formic acid / CH2Cl2; H2O / 1 h / 20 °C 2: 98 percent / TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 3: 80 percent / Na / methanol / 1 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) 30percent H2O2, 90percent aq. HCOOH 2.) aq. NaOH / 1.) r.t., 1 h, 2.) MeOH, 2: 1.) DMSO, TFAA 2.) NEt3 / 1.) CH2Cl2, -60 deg C, 10 min, 2.) -60 deg C, 15 min; -60 deg C to 5 deg C 3: 80 percent / Na / methanol / 1 h / Ambient temperature View Scheme |
-
-
501433-34-7
5α/β-androst-3-en-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: aq. H2O2; HCO2H / CH2Cl2 / 6 h / 20 °C 2.1: 48 percent / HCO2H / 0.5 h / 20 °C 3.1: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 3.2: NaOMe / methanol View Scheme |
-
-
37716-99-7
CGP 79318

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 2: NaOMe / methanol View Scheme | |
| Multi-step reaction with 2 steps 1: 98 percent / TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: 80 percent / Na / methanol / 1 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 1.) DMSO, TFAA 2.) NEt3 / 1.) CH2Cl2, -60 deg C, 10 min, 2.) -60 deg C, 15 min; -60 deg C to 5 deg C 2: 80 percent / Na / methanol / 1 h / Ambient temperature View Scheme |
-
-
192192-77-1
3β,4β-epoxy-5β-androstan-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: 48 percent / HCO2H / 0.5 h / 20 °C 2.1: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 2.2: NaOMe / methanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Na / methanol / 1 h / 20 °C 2: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme |
-
-
33386-50-4
3β,5β-dihydroxyandrostan-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme | |
| Multi-step reaction with 4 steps 1: TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: Na / methanol / 1 h / 20 °C 3: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme |
-
-
434939-07-8
3α,4β-dihydroxy-5β-androstane-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: 80 percent / Na / methanol / 1 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 2: 1) DMSO, trifluoroacetic anhydride, 2) Et3N / 1) CH2Cl2, -60 deg C, 3 h, 2) CH3Cl2, -60 deg C, 15 min, -60 deg C to 5 deg C 3: 80 percent / Na / methanol / 1 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1) DMSO, trifluoroacetic anhydride, 2) Et3N / 1) CH2Cl2, -60 deg C, 3 h, 2) CH3Cl2, -60 deg C, 15 min, -60 deg C to 5 deg C 2: 80 percent / Na / methanol / 1 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Jones reagent / acetone / -20 °C 2: osmium(VIII) oxide; dihydrogen peroxide / tert-butyl alcohol / 72 h / 20 °C 3: potassium hydroxide / methanol / 0.17 h / 55 °C View Scheme |
-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
-
75-36-5
acetyl chloride

-
-
61630-32-8
3,17-dioxoandrost-4-en-4-yl acetate

| Conditions | Yield |
|---|---|
| In pyridine for 3h; cooling; | 98% |
| With pyridine at 0 - 20℃; for 21.5h; | 72% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 2h; Substitution; Heating; | 85% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In dichloromethane; water for 1h; Ambient temperature; | 79% |
-
-
108-24-7
acetic anhydride

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
-
61630-32-8
3,17-dioxoandrost-4-en-4-yl acetate

| Conditions | Yield |
|---|---|
| With pyridine Ambient temperature; | 67% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In dichloromethane; water for 0.166667h; Ambient temperature; | 66% |
-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
-
134797-42-5
4-hydroxy-16-oximino-4-androsten-3,17-dione

| Conditions | Yield |
|---|---|
| With n-Amyl nitrite; potassium tert-butylate In tert-butyl alcohol for 2h; | 55% |
-
-
201230-82-2
carbon monoxide

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With di(rhodium)tetracarbonyl dichloride In 1,2-dichloro-benzene at 130℃; under 760.051 Torr; for 96h; Glovebox; | 55% |
| With di(rhodium)tetracarbonyl dichloride; tris(pentafluorophenyl)phosphine In 1,2-dichloro-benzene at 130℃; for 96h; | 55% |

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
-
98-88-4
benzoyl chloride

-
-
76942-05-7
Benzoic acid (8R,9S,10R,13S,14S)-10,13-dimethyl-3,17-dioxo-2,3,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-4-yl ester

| Conditions | Yield |
|---|---|
| With pyridine Ambient temperature; | 48% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene for 48h; Reflux; Dean-Stark; | 47% |

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 20℃; for 1h; Inert atmosphere; | 40% |
-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid; silica gel; acetic acid at 70℃; for 0.05h; | 86% |
| With boron trifluoride diethyl etherate In benzene for 19h; Ambient temperature; | 47% |
| With formic acid for 0.75h; Heating; | 45% |
| Multi-step reaction with 2 steps 1: 62 percent / aq. sodium hydroxide / 3 h / Heating 2: 53 percent / hydrochloric acid / dioxane / 24 h / Heating View Scheme |
-
-
180303-17-7
3-hydroxy-5α-androst-2-ene-4,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With sodium In methanol for 1h; Ambient temperature; | 80% |
| With sodium In methanol at 20℃; for 1h; | 80% |
| With sodium methylate In methanol |

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With sodium In methanol for 1h; Ambient temperature; | 80% |
-
-
434939-05-6
3β,4α-dihydroxy-5β-androstan-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Stage #1: 3β,4α-dihydroxy-5β-androstan-17-one With dimethyl sulfoxide; triethylamine; trifluoroacetic anhydride In dichloromethane at -60℃; for 3h; Stage #2: With sodium In methanol at 20℃; for 3h; | 80% |
| Stage #1: 3β,4α-dihydroxy-5β-androstan-17-one With dimethyl sulfoxide; triethylamine; trifluoroacetic anhydride at -60℃; for 3.25h; Swern oxidation; Stage #2: With sodium methylate In methanol | |
| Multi-step reaction with 2 steps 1: TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: 80 percent / Na / methanol / 1 h / 20 °C View Scheme |
-
-
434939-09-0
3-hydroxy-5β-androst-2-ene-4,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With sodium In methanol at 20℃; for 1h; | 80% |
-
-
501433-34-7
5α/β-androst-3-en-17-one

-
A
-
63-05-8
Androstenedione

-
B
-
1229-12-5
androstane-3,17-dione

-
C
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multistep reaction; | A n/a B n/a C 54% |

-
-
20986-46-3
4-methoxy-4-androstene-3,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 1,4-dioxane for 24h; Heating; | 53% |
-
-
64-19-7
acetic acid

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
2241-85-2
2α-acetoxyandrost-4-ene-3,17-dione

-
B
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
C
-
571-17-5
2alpha-Hydroxyandrost-4-ene-3,17-dione

-
D
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid a) RT, 4 h, b) 4 deg C, 12 h; Further byproducts given; | A 23% B 52% C 0.58 g D 0.7 g |

-
-
64-19-7
acetic acid

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
B
-
571-17-5
2alpha-Hydroxyandrost-4-ene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140223-19-4
4α-acetoxy-5α-hydroxyandrostane-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid a) RT, 4h,b) 4 deg C, 12 h; Further byproducts given; | A 52% B 0.58 g C 0.7 g D 3% |

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
2241-85-2
2α-acetoxyandrost-4-ene-3,17-dione

-
B
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
C
-
571-17-5
2alpha-Hydroxyandrost-4-ene-3,17-dione

-
D
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid a) RT, 4 h, b) 4 deg C, 12 h; Further byproducts given; | A 23% B 52% C 0.58 g D 0.7 g |

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
2241-85-2
2α-acetoxyandrost-4-ene-3,17-dione

-
B
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140223-19-4
4α-acetoxy-5α-hydroxyandrostane-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid a) RT, 4h , b) 4 deg C, 12 h; Further byproducts given; | A 23% B 52% C 0.7 g D 3% |

-
-
77057-73-9
4,5-epoxyandrostane-3,17-dione

-
A
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
B
-
571-17-5
2alpha-Hydroxyandrost-4-ene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140223-19-4
4α-acetoxy-5α-hydroxyandrostane-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid a> RT, 4 h, b) 4 deg C, 12 h; Further byproducts given; | A 52% B 0.58 g C 0.7 g D 3% |


| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid for 18h; Ambient temperature; | 50% |
| With pyridine; hydrogen fluoride at 55℃; |
-
-
64-19-7
acetic acid

-
-
88509-24-4
4,5-epoxyandrost-9(11)-ene-3,17-dione

-
A
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
B
-
140111-67-7
2α-hydroxyandrost-4,9(11)-diene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140111-66-6
2α-acetoxyandrost-4,9(11)-diene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid a) RT, 4h, b) 4 deg C, 12 h; | A 13.6% B n/a C 390 mg D 9% |

-
-
88509-24-4
4,5-epoxyandrost-9(11)-ene-3,17-dione

-
A
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

-
B
-
140111-67-7
2α-hydroxyandrost-4,9(11)-diene-3,17-dione

-
C
-
88509-26-6
4-hydroxyandrost-4,9(11)-diene-3,17-dione

-
D
-
140111-66-6
2α-acetoxyandrost-4,9(11)-diene-3,17-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid a) RT, 4 h, b) 4 deg C, 12h; | A 13.6% B n/a C 390 mg D 9% |
| With sulfuric acid In acetic acid a) RT, 4 h, b) 4 deg C, 12 h; | A 13.6% B n/a C 390 mg D 9% |
| With sulfuric acid In acetic acid a) RT, 4h, b) 4 deg C, 12 h; | A 13.6% B n/a C 390 mg D 9% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; sulfuric acid; dihydrogen peroxide 1a) 0 to 4 deg C, 30 min, MeOH, 1b) 5 to 7 deg C, 22 h, MeOH, 2) HOAc, RT, 45 min; Yield given. Multistep reaction; | |
| Multi-step reaction with 4 steps 1: 60 percent / Zn; AcOH / 0.25 h / 118 °C 2: 96 percent / aq. H2O2; HCO2H / 1 h / 20 °C 3: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 4: NaOMe / methanol View Scheme | |
| Multi-step reaction with 2 steps 1: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme | |
| Multi-step reaction with 2 steps 1: osmium tetroxide, 35percent H2O2 / 2-methyl-propan-2-ol / 72 h / Ambient temperature 2: potassium hydroxide / methanol / 0.17 h / 55 °C View Scheme | |
| Multi-step reaction with 2 steps 1: osmium(VIII) oxide; dihydrogen peroxide / tert-butyl alcohol / 72 h / 20 °C 2: potassium hydroxide / methanol / 0.17 h / 55 °C View Scheme |
-
-
110267-65-7
4ξ,5-dihydroxy-5ξ-androstane-3,17-dione

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol at 55℃; for 0.166667h; Yield given; | |
| With potassium hydroxide In methanol at 55℃; for 0.166667h; | 0.452 g |
-
-
14935-81-0
5α-androst-3-en-17β-ol

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 96 percent / aq. H2O2; HCO2H / 1 h / 20 °C 2: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 3: NaOMe / methanol View Scheme | |
| Multi-step reaction with 3 steps 1: 96 percent / H2O2; formic acid / CH2Cl2; H2O / 1 h / 20 °C 2: 98 percent / TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 3: 80 percent / Na / methanol / 1 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) 30percent H2O2, 90percent aq. HCOOH 2.) aq. NaOH / 1.) r.t., 1 h, 2.) MeOH, 2: 1.) DMSO, TFAA 2.) NEt3 / 1.) CH2Cl2, -60 deg C, 10 min, 2.) -60 deg C, 15 min; -60 deg C to 5 deg C 3: 80 percent / Na / methanol / 1 h / Ambient temperature View Scheme |
-
-
501433-34-7
5α/β-androst-3-en-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: aq. H2O2; HCO2H / CH2Cl2 / 6 h / 20 °C 2.1: 48 percent / HCO2H / 0.5 h / 20 °C 3.1: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 3.2: NaOMe / methanol View Scheme |
-
-
37716-99-7
CGP 79318

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 2: NaOMe / methanol View Scheme | |
| Multi-step reaction with 2 steps 1: 98 percent / TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: 80 percent / Na / methanol / 1 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 1.) DMSO, TFAA 2.) NEt3 / 1.) CH2Cl2, -60 deg C, 10 min, 2.) -60 deg C, 15 min; -60 deg C to 5 deg C 2: 80 percent / Na / methanol / 1 h / Ambient temperature View Scheme |
-
-
192192-77-1
3β,4β-epoxy-5β-androstan-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: 48 percent / HCO2H / 0.5 h / 20 °C 2.1: dimethyl sulfoxide; (CF3CO)2O; Et3N / 3.25 h / -60 °C 2.2: NaOMe / methanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Na / methanol / 1 h / 20 °C 2: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme |
-
-
33386-50-4
3β,5β-dihydroxyandrostan-17-one

-
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme | |
| Multi-step reaction with 4 steps 1: TFAA; Et3N; DMSO / CH2Cl2 / 3 h / -60 °C 2: Na / methanol / 1 h / 20 °C 3: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme |
Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View






 T
T