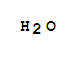Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:7789-20-0
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
D2O Deuterium oxide CAS 7789-20-0 Heavy water CAS no 7789-20-0 Water-d2 Deuterium oxide Manufacturer High quality Best price In stock factory CAS 7789-20-0 Deuterium oxide COA TDS price MSDS highly quality and immedaite delivery DEUTERI
Cas:7789-20-0
Min.Order:5 Kiloliter
FOB Price: $3.5 / 5.0
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:7789-20-0
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:7789-20-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
DEUTERIUM OXIDE CAS:7789-20-0 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermedia
Cas:7789-20-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Shandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Hubei Jiutian Bio-medical Technology Co., Ltd
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highly qualifie
Cas:7789-20-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:7789-20-0
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colourless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediat
Hebei Kunsui Technology Co., Ltd.
1. A strong scientific research team . 2. Stable quality (with a complete scientific research center and testing center to ensure the quality stability of each batch of products). 3. Rich export experience (with practical experience in exporting to m
Cas:7789-20-0
Min.Order:1 Liter
FOB Price: $4.5 / 5.0
Type:Trading Company
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:7789-20-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
HANGZHOU TIANYE CHEMICALS CO., LTD.
We product this chemical more than 10 years . We are very experience to export it to many countries, Our superior & stable quality , competitive price gain warm reception from our customers. Product Name:DEUTERIUM OXIDE CAS No:7789-20-0 Synon
Henan Kanbei Chemical Co.,LTD
High quality, competitive price, fast delivery and first-class service we possesses have won the trust and praise of customers. Standard: BP/USP/EP The purity is equal or greater than 99%. As a supplier, we can provide high-quality products. Cle
Cas:7789-20-0
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
DEUTERIUM OXIDE Basic information Product Name: DEUTERIUM OXIDE Synonyms: WATER-D2;WATER, HEAVY;DEUTERIUM OXIDE, "100" (MIN. 99.96 ATOM% D);Deuterium oxide "100%" >99.98 Atom % D;Deuterium oxide >99.92 Atom % D;Heavy
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:7789-20-0
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryHunan chemfish Pharmaceutical co.,Ltd
7789-20-0 Appearance:95%+ Package:R&D,Pilot run Application:7789-20-0 Transportation:per client require Port:Express ,Air, Sea
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:7789-20-0
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Golden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Henan Tianfu Chemical Co., Ltd.
Our advantage:Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientif
Cas:7789-20-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryWin-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:7789-20-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Synthetic route
-
-
13587-54-7
deuteroxyl

-
-
13587-52-5
nitric acid-d1

-
A
-
7789-20-0
water-d2

-
B
-
12033-49-7, 807306-99-6
nitrate radical

| Conditions | Yield |
|---|---|
| In gaseous matrix Kinetics; Irradiation (UV/VIS); photolysis in Pyrex vessel, gas flow velocity: 5-10 cms**-1, T: 239.7-, 370.1 K, p: 20-199.7 Torr, buffer gas: He, SF6; | A n/a B 0.95% |
| In neat (no solvent) Kinetics; 263-446 K, 1-20 Torr; | |
| With sulfur(VI) hexafluoride In neat (no solvent) Kinetics; 263-446 K, 1-107 Torr; |


| Conditions | Yield |
|---|---|
| In not given 34.2°C;; |

| Conditions | Yield |
|---|---|
| In not given byproducts: H(2)H; equilibrium constant discussed;; | |
| In not given byproducts: (2)HH; equilibrium constant discussed;; | |
| In not given byproducts: (2)HH; equilibrium discussed;; | |
| In not given byproducts: (2)HH; equilibrium constant discussed;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent, gas phase) hydrogen-isotope-exchange reaction studied in electrochem. double cells; |


| Conditions | Yield |
|---|---|
| In not given byproducts: H2; thermodynamic data discussed;; | |
| With catalyst: Pd on activated charcoal; In neat (no solvent) | |
| In not given byproducts: H; equilibrium constant discussed;; |

| Conditions | Yield |
|---|---|
| production of D2O by H/D exchange; | |
| production of D2O by H/D exchange; |

| Conditions | Yield |
|---|---|
| In not given Kinetics; | |
| In not given Kinetics; |

| Conditions | Yield |
|---|---|
| rutile In neat (no solvent) Kinetics; byproducts: formaldehyde, CO2, (2)H2; other Radiation; decompn. on stoch. and defective surfaces of TiO2(110) rutile single crystal at 400-613 K; yields of CO and (2)H2O were about 60 %; (2)H2CO and CO2 - about 10 %; (2)H2 -about 2 %; detd. by TPRS; |

| Conditions | Yield |
|---|---|
| In not given byproducts: Fe; equilibrium constant discussed;; |

| Conditions | Yield |
|---|---|
| In solid pressurizing (77 K, 15 kbar), releasing pressure (liq. N2), transferring(aluminium container), evacuating (20 mbar), cooling (5 or 15 K); |

| Conditions | Yield |
|---|---|
| In acetone Sonication; sonicating (30 min), D2-atmosphere (1 h), air atmosphere (overnight); detd. by (2)D NMR spectroscopy; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; react. of D and O coadsorbed on Rh(100) surface at 90 K, heated in UHV between 140 and 240 K, formation of OD intermediate, heating higher than 240 K, H2O formed and evolved from surface; react. is monitored by electron energy loss spectroscopy and low energy electron diffraction; |

| Conditions | Yield |
|---|---|
| platinum In neat (no solvent) other Radiation; 200 Torr, microwave discharge, 440-913 K; |
-
-
13537-07-0
silane-d4

-
-
7440-21-3
monosilane

-
A
-
7789-20-0
water-d2

-
B
-
14940-63-7
water

-
D
-
16873-17-9
deuterium

| Conditions | Yield |
|---|---|
| In gaseous matrix Kinetics; react. in a discharge flow system with MS spect. detection (O-atom generation by titration react. of N/NO), carrier gas Ar, room temp., pressure 400 Pa, coated reactor; product distribution; products not isolated, detd. by MS, GC; |

-
-
16873-17-9
deuterium

-
-
7789-20-0
water-d2

| Conditions | Yield |
|---|---|
| With αO in FeZSM-5 zeolite In neat (no solvent) Kinetics; -100 - +20 °C; | |
| In neat (no solvent) zeolite treated at 550°C, surface α-oxygen formed by decomposing N2O at 250°C, interaction of deuterium with α-oxygen at 100°C, condensed products freezed in a trap at -196°C for 30 min; | |
| With VO(x) on Pd(111) In neat (no solvent) oxidation of D2 on Pd(111) surface coated by VO(x) prepared by electron beam evaporation of V onto Pd at 523 K in O2 atmosphere of 2*10**-7 mbar; detd. by MAS; |
-
-
1333-74-0
hydrogen

-
-
10102-43-9
nitrogen(II) oxide

-
-
16873-17-9
deuterium

-
A
-
7727-37-9
nitrogen

-
B
-
7732-18-5
water

-
C
-
7789-20-0
water-d2

-
D
-
14940-63-7
water

| Conditions | Yield |
|---|---|
| With catalyst: Pd/Ce-Zr-La-γ-alumina In neat (no solvent) Kinetics; byproducts: N2O, NH3, NH2(2)H; Ar; at 155+/-3°C, at atm. pressure; further by-products: NH(2)H2,N(2)H3; MS; |

-
-
1333-74-0
hydrogen

-
-
80937-33-3
oxygen

-
-
16873-17-9
deuterium

-
A
-
7732-18-5
water

-
B
-
7789-20-0
water-d2

-
C
-
14940-63-7
water

| Conditions | Yield |
|---|---|
| platinum In gaseous matrix 200 mTorr; not isolated; IR spectroscopy; |


| Conditions | Yield |
|---|---|
| Kinetics; byproducts: CH4; up to various time on Ni/SiO2 catalyst; D2O detd. by trapping at 77 K, followed analysis using gas chromy.; | |
| rhodium In neat (no solvent) byproducts: CD4; methanation on Rh surface at 10**-8Torr and 150K using pulsed-laser image atom-probe; studying atomic steps of react.;; detn. react. intermediates by time-of-flight mass spectrum;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; react. of H2-D2 over PMo12 at 573 K; initial pressures of H2: 12 Torr, D2 20 Torr; detn. of pressure decrease; evolved water detd. volumetrically; |


| Conditions | Yield |
|---|---|
| platinum 300 K; |


| Conditions | Yield |
|---|---|
| With air; hydrogen; LaNi5(2)H6.7 room temp.; | A 0% B n/a |


| Conditions | Yield |
|---|---|
| copper(II) oxide In not given Kinetics; | |
| nickel(II) oxide In not given Kinetics; | |
| platinum Kinetics; water formation on Pt; |
-
-
34557-54-5
methane

-
-
124-38-9
carbon dioxide

-
-
16873-17-9
deuterium

-
A
-
201230-82-2
carbon monoxide

-
B
-
7732-18-5
water

-
C
-
1333-74-0
hydrogen

-
D
-
7789-20-0
water-d2

-
E
-
14940-63-7
water

| Conditions | Yield |
|---|---|
| With catalyst:7 wtpercentNi/MgO In neat (no solvent) Kinetics; byproducts: HDO; 7 wt% Ni/MgO as catalyst, CH4:CO2:D2=1:1:0.2, 973 K, 16.7 kPa CH4 and CO2, resp., 3.3 kPa D2, 100 kPa total pressure, Ar as balance, HDO was also formed; monitored by MS; |


| Conditions | Yield |
|---|---|
| nickel Kinetics; at 124-192 °C,total pressure:15 mmHg; | |
| With catalysts: Ir/Al2O3 Kinetics; at 393-473°C; | |
| With catalysts: Pt/Al2O3 Kinetics; at 393-473°C; |


| Conditions | Yield |
|---|---|
| In not given byproducts: Sn; equilibrium constant discussed;; | |
| In not given byproducts: Sn; | |
| In not given byproducts: Sn; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; |

-
-
124-38-9
carbon dioxide

-
-
16873-17-9
deuterium

-
A
-
7732-18-5
water

-
B
-
7789-20-0
water-d2

-
C
-
14940-63-7
water

| Conditions | Yield |
|---|---|
| With methane In neat (no solvent) reaction of CH4/CO2/D2/Ar mixt. at 873 K on catalyst Ru/Al2O3 studied; MS; | |
| With methane In gas react. of CH4/CO2/(2)H2 mixt. on Pt/(ZrO2-CeO2) at 873 K led to production of water isotopomers; MS; |

-
-
34557-54-5
methane

-
-
558-20-3
methane

-
-
124-38-9
carbon dioxide

-
A
-
201230-82-2
carbon monoxide

-
B
-
7732-18-5
water

-
C
-
7789-20-0
water-d2

-
D
-
7440-44-0
pyrographite

-
E
-
14940-63-7
water

| Conditions | Yield |
|---|---|
| With catalyst:7 wtpercentNi/MgO In neat (no solvent) Kinetics; byproducts: CHD3, CH2D2, CH3D; 7 wt% Ni/MgO as catalyst, CH4:CD4:H2O=1:1:2, 873-973 K, 12.5 kPa CH4 andCD4, resp., 25 kPa CO2, 100 kPa total pressure, Ar as balance; monitored by MS; |

-
-
558-20-3
methane

-
-
124-38-9
carbon dioxide

-
A
-
201230-82-2
carbon monoxide

-
B
-
7789-20-0
water-d2

-
C
-
7440-44-0
pyrographite

-
D
-
16873-17-9
deuterium

| Conditions | Yield |
|---|---|
| With catalyst:7 wtpercentNi/MgO In neat (no solvent) Kinetics; 7 wt% Ni/MgO as catalyst, 20 kPa CD4 and 25 kPa CO2 pressure, resp., He as balance, 100-1500 kPa total pressure, 873 K; |


| Conditions | Yield |
|---|---|
| rutile In neat (no solvent) react. at oxygen vacancy defects on surface of rutile TiO2(110); detd. by TPD; |

| Conditions | Yield |
|---|---|
| With sulfuric acid In hydrogenchloride dissolving the cerium oxide in dil. HCl adding the equiv. amt. of 0.1 NH2SO4 to yield a quant. amt. of sulfate which was pptd. as small needles by the addn. of an excess of ethyl alcohol; the crystals were filteredoff; washed with water/alcohol, purifn. by crystn., dehydration under vac. at 380°C; after sufficient dehydration the compound was dissolved in D2O at about 4°C; | 100% |

| Conditions | Yield |
|---|---|
| With sodium methoxide In tetrahydrofuran; deuteromethanol MeONa added to soln. of ((C5H4)2)Mo2(CO)5(C(CH2)3O) in THF and CH3OD, reacted for 5 min; filtered through short column of alumina deactivated with D2O, eluted with THF, rotary evapd.; | 100% |

-
-
188759-75-3
((CH3)5C5)W(NO)(OH)(η(2)-HN=C(Me)CH=CPh)

-
-
7789-20-0
water-d2

-
-
202531-09-7
((CH3)5C5)W(NO)(OD)(η(2)-DN=C(Me)CH=CPh)

| Conditions | Yield |
|---|---|
| In [D3]acetonitrile N2 or Ar-atmosphere, NMR tube; 45°C (overnight); not isolated, detd. by NMR spectroscopy; | 100% |
-
-
188759-74-2
((CH3)5C5)W(NO)(η(3)-HNC(Me)=NC(=CH2)CH=CPh)

-
-
7789-20-0
water-d2

-
-
202531-10-0
((CH3)5C5)W(NO)(η(3)-DNC(CD3)=NC(=CD2)CH=CPh)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran-d8 N2 or Ar-atmosphere; 55°C (48 h); not isolated, detd. by NMR spectroscopy; | 100% |
-
-
13587-54-7
deuteroxyl

-
-
13587-52-5
nitric acid-d1

-
A
-
7789-20-0
water-d2

-
B
-
12033-49-7, 807306-99-6
nitrate radical

| Conditions | Yield |
|---|---|
| In gaseous matrix Kinetics; Irradiation (UV/VIS); photolysis in Pyrex vessel, gas flow velocity: 5-10 cms**-1, T: 239.7-, 370.1 K, p: 20-199.7 Torr, buffer gas: He, SF6; | A n/a B 0.95% |
| In neat (no solvent) Kinetics; 263-446 K, 1-20 Torr; | |
| With sulfur(VI) hexafluoride In neat (no solvent) Kinetics; 263-446 K, 1-107 Torr; |


| Conditions | Yield |
|---|---|
| In not given 34.2°C;; |

| Conditions | Yield |
|---|---|
| In not given byproducts: H(2)H; equilibrium constant discussed;; | |
| In not given byproducts: (2)HH; equilibrium constant discussed;; | |
| In not given byproducts: (2)HH; equilibrium discussed;; | |
| In not given byproducts: (2)HH; equilibrium constant discussed;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent, gas phase) hydrogen-isotope-exchange reaction studied in electrochem. double cells; |


| Conditions | Yield |
|---|---|
| In not given byproducts: H2; thermodynamic data discussed;; | |
| With catalyst: Pd on activated charcoal; In neat (no solvent) | |
| In not given byproducts: H; equilibrium constant discussed;; |

| Conditions | Yield |
|---|---|
| production of D2O by H/D exchange; | |
| production of D2O by H/D exchange; |

| Conditions | Yield |
|---|---|
| In not given Kinetics; | |
| In not given Kinetics; |

| Conditions | Yield |
|---|---|
| rutile In neat (no solvent) Kinetics; byproducts: formaldehyde, CO2, (2)H2; other Radiation; decompn. on stoch. and defective surfaces of TiO2(110) rutile single crystal at 400-613 K; yields of CO and (2)H2O were about 60 %; (2)H2CO and CO2 - about 10 %; (2)H2 -about 2 %; detd. by TPRS; |

| Conditions | Yield |
|---|---|
| In not given byproducts: Fe; equilibrium constant discussed;; |

| Conditions | Yield |
|---|---|
| In solid pressurizing (77 K, 15 kbar), releasing pressure (liq. N2), transferring(aluminium container), evacuating (20 mbar), cooling (5 or 15 K); |

| Conditions | Yield |
|---|---|
| In acetone Sonication; sonicating (30 min), D2-atmosphere (1 h), air atmosphere (overnight); detd. by (2)D NMR spectroscopy; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; react. of D and O coadsorbed on Rh(100) surface at 90 K, heated in UHV between 140 and 240 K, formation of OD intermediate, heating higher than 240 K, H2O formed and evolved from surface; react. is monitored by electron energy loss spectroscopy and low energy electron diffraction; |

| Conditions | Yield |
|---|---|
| platinum In neat (no solvent) other Radiation; 200 Torr, microwave discharge, 440-913 K; |
-
-
13537-07-0
silane-d4

-
-
7440-21-3
monosilane

-
A
-
7789-20-0
water-d2

-
B
-
14940-63-7
water

-
D
-
16873-17-9
deuterium

| Conditions | Yield |
|---|---|
| In gaseous matrix Kinetics; react. in a discharge flow system with MS spect. detection (O-atom generation by titration react. of N/NO), carrier gas Ar, room temp., pressure 400 Pa, coated reactor; product distribution; products not isolated, detd. by MS, GC; |

-
-
16873-17-9
deuterium

-
-
7789-20-0
water-d2

| Conditions | Yield |
|---|---|
| With αO in FeZSM-5 zeolite In neat (no solvent) Kinetics; -100 - +20 °C; | |
| In neat (no solvent) zeolite treated at 550°C, surface α-oxygen formed by decomposing N2O at 250°C, interaction of deuterium with α-oxygen at 100°C, condensed products freezed in a trap at -196°C for 30 min; | |
| With VO(x) on Pd(111) In neat (no solvent) oxidation of D2 on Pd(111) surface coated by VO(x) prepared by electron beam evaporation of V onto Pd at 523 K in O2 atmosphere of 2*10**-7 mbar; detd. by MAS; |
-
-
1333-74-0
hydrogen

-
-
10102-43-9
nitrogen(II) oxide

-
-
16873-17-9
deuterium

-
A
-
7727-37-9
nitrogen

-
B
-
7732-18-5
water

-
C
-
7789-20-0
water-d2

-
D
-
14940-63-7
water

| Conditions | Yield |
|---|---|
| With catalyst: Pd/Ce-Zr-La-γ-alumina In neat (no solvent) Kinetics; byproducts: N2O, NH3, NH2(2)H; Ar; at 155+/-3°C, at atm. pressure; further by-products: NH(2)H2,N(2)H3; MS; |

-
-
1333-74-0
hydrogen

-
-
80937-33-3
oxygen

-
-
16873-17-9
deuterium

-
A
-
7732-18-5
water

-
B
-
7789-20-0
water-d2

-
C
-
14940-63-7
water

| Conditions | Yield |
|---|---|
| platinum In gaseous matrix 200 mTorr; not isolated; IR spectroscopy; |


| Conditions | Yield |
|---|---|
| Kinetics; byproducts: CH4; up to various time on Ni/SiO2 catalyst; D2O detd. by trapping at 77 K, followed analysis using gas chromy.; | |
| rhodium In neat (no solvent) byproducts: CD4; methanation on Rh surface at 10**-8Torr and 150K using pulsed-laser image atom-probe; studying atomic steps of react.;; detn. react. intermediates by time-of-flight mass spectrum;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; react. of H2-D2 over PMo12 at 573 K; initial pressures of H2: 12 Torr, D2 20 Torr; detn. of pressure decrease; evolved water detd. volumetrically; |


| Conditions | Yield |
|---|---|
| platinum 300 K; |


| Conditions | Yield |
|---|---|
| With air; hydrogen; LaNi5(2)H6.7 room temp.; | A 0% B n/a |


| Conditions | Yield |
|---|---|
| copper(II) oxide In not given Kinetics; | |
| nickel(II) oxide In not given Kinetics; | |
| platinum Kinetics; water formation on Pt; |
-
-
34557-54-5
methane

-
-
124-38-9
carbon dioxide

-
-
16873-17-9
deuterium

-
A
-
201230-82-2
carbon monoxide

-
B
-
7732-18-5
water

-
C
-
1333-74-0
hydrogen

-
D
-
7789-20-0
water-d2

-
E
-
14940-63-7
water

| Conditions | Yield |
|---|---|
| With catalyst:7 wtpercentNi/MgO In neat (no solvent) Kinetics; byproducts: HDO; 7 wt% Ni/MgO as catalyst, CH4:CO2:D2=1:1:0.2, 973 K, 16.7 kPa CH4 and CO2, resp., 3.3 kPa D2, 100 kPa total pressure, Ar as balance, HDO was also formed; monitored by MS; |


| Conditions | Yield |
|---|---|
| nickel Kinetics; at 124-192 °C,total pressure:15 mmHg; | |
| With catalysts: Ir/Al2O3 Kinetics; at 393-473°C; | |
| With catalysts: Pt/Al2O3 Kinetics; at 393-473°C; |


| Conditions | Yield |
|---|---|
| In not given byproducts: Sn; equilibrium constant discussed;; | |
| In not given byproducts: Sn; | |
| In not given byproducts: Sn; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; |

Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View