HUNAN CHEMAPI BIOLOGICAL TECHNOLOGY CO.,LTD.
Hunan Kaimir Biotechnology Co., Ltd. is a professional technology company specializing in the research and production of raw materials and intermediates. In 2017, it was recognized as a high-tech enterprise and a small technological innovation giant
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:79617-96-2
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:79617-96-2
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:79617-96-2
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
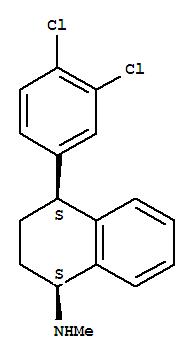
Sertraline Basic information Product Name: Sertraline Synonyms: (1s-cis)-1,2,3,4-tetrahydro-4-(3,4-dichlorophenyl)-n-methyl-1-naphthalenamin;1,2,3,4-tetrahydro-4-(3,4-dichlorophenyl)-n-m
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:79617-96-2
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:White Crystalline Powder Storage:Store in sealed conta
Cas:79617-96-2
Min.Order:10 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:79617-96-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Sertraline CAS:79617-96-2 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates,
Cas:79617-96-2
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:79617-96-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:79617-96-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
DB BIOTECH CO., LTD
Q1: Are you a manufacturer?Answer: Yes, we are factory founded on 2002. Q2: How to contact with us?Click "contact supplier" And then send us message the product you interest in, you will get reply within 12 hours. Q3:Which kind of payment terms do yo
Cas:79617-96-2
Min.Order:0
Negotiable
Type:Trading Company
inquiryAfine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Hefei Zhaobo Technology Co., Ltd.
High quality Sertraline cas 79617-96-2 Purity: 99% Min Application: Intermediates Appearance: Powder Package: Bag Delivery: 3-5days Our Advantage & Service 1.Top quality: Using high quality material and establishing a strict quality
Cas:79617-96-2
Min.Order:1 Kilogram
FOB Price: $10.0 / 50.0
Type:Trading Company
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Wuhan Xinhao Biotechnology Co., Ltd.
1. We can provide customers with "one-stop"packaging service,from research,development,production,export and so on 2. Powerful R&D strength let our technology in a leading level,forever,in turn,to provide customers with better service .
Cas:79617-96-2
Min.Order:100 Gram
FOB Price: $10.0 / 100.0
Type:Trading Company
inquiryHangzhou Lingrui Chemical Co.,Ltd.
our strengths: 1: Fast and guaranteed shipment (TNT;EMS;FEDEX;DHL;UPS;EUB, special line) 2: Various payment items accepted (Btc; MoneyGram; WU) 3: Valued package (Paraffin coating; Double aluminum foil bag; Vacuum packaging) 4: Efficient delivery
Cas:79617-96-2
Min.Order:1 Gram
Negotiable
Type:Other
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:79617-96-2
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryTaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Hangzhou Dawn Ray Pharmaceutical Co.,Ltd
Description: white crystalline powder, odorless Specific optical rotation :+37.5°~+42.5° Identification: Chemical iden…Appearance:white crystalline powder, odorless Storage:cool and dry Package:25kg/drum Application:Other
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Synthetic route
-
-
79559-97-0
sertraline hydrochloride

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; toluene for 0.25h; pH=> 10; | 93% |
| With sodium hydroxide In water; ethyl acetate | 77% |
| With ammonium hydroxide In water pH=10; | |
| With sodium hydroxide In water | |
| With potassium carbonate In water at 20℃; | 10.9 g |

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; ethyl acetate for 0.25h; pH=> 10; | 93% |
| With sodium hydroxide In water; ethyl acetate for 0.25h; pH=> 10; | 93% |
-
-
1346228-89-4
benzyl (1S,4S)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydronaphthalen-1-yl(methyl)carbamate

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With hydrogen In methanol; dichloromethane; water at 20℃; for 5h; Inert atmosphere; | 89% |
-
-
79560-19-3, 79836-44-5, 124379-29-9
(4S)-4-(3,4-dichlorophenyl)-1-tetralone

-
-
74-89-5
methylamine

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In tetrahydrofuran; diethyl ether; dichloromethane at -78 - 25℃; | 88% |
-
-
267884-85-5
cis-(1S,4S)-N-(tert-butoxycarbonyl)-N-methyl-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-1-naphthalenamine

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; for 1h; deacylation; | 85% |
-
-
234448-58-9
C17H16Cl2IN

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With tert.-butyl lithium In tetrahydrofuran; toluene at -78℃; | 69% |

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With cesium fluoride In N,N-dimethyl-formamide at 95℃; for 20h; diastereoselective reaction; | 65% |
-
-
4229-44-1
N-methylhydroxyamine hydrochloride

-
-
79560-19-3, 79836-44-5, 124379-29-9
(4S)-4-(3,4-dichlorophenyl)-1-tetralone

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Stage #1: N-methylhydroxyamine hydrochloride; (4S)-4-(3,4-dichlorophenyl)-1-tetralone With sodium acetate In ethanol for 15h; Reflux; Stage #2: With Lindlar's catalyst; hydrogen In methanol; dichloromethane at 20℃; under 760.051 Torr; for 20h; | 47% |
-
-
79617-95-1
cis-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthaleneamine

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With potassium carbonate; lithium (hydro)trimethoxyaluminate 1) MeCN, 2) THF, reflux, 40 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With lithium (hydro)trimethoxyaluminate In tetrahydrofuran for 40h; Reduction; Methylation; Heating; | 10 mg |
-
-
261776-41-4
(4-(3,4-dichlorophenyl)-3,4-dihydro-2H-naphthalen-1-ylidene)methylamine

-
A
-
91797-60-3
trans-(1S,4R)-N-methyl-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-1-naphthalenamine

-
B
-
79617-96-2
Sertraline

-
C
-
155748-61-1
(4R)-(3,4-Dichlorophenyl)-3,4-dihydro-1(2H)-naphthalenone

| Conditions | Yield |
|---|---|
| With (S,S)-(ethylenebis(tetrahydroindenyl))TiF2; phenylsilane In tetrahydrofuran at 20℃; for 24h; kinetic resolution; |

-
-
6154-14-9
N-chloromethylamine

-
-
439134-50-6
1-(3,4-dichlorophenyl)-1,2-dihydronaphthalene

-
A
-
79951-46-5
trans-(1R,4S)-N-methyl-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-1-naphthalenamine

-
B
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Stage #1: 1-(3,4-dichlorophenyl)-1,2-dihydronaphthalene With benzo[1,3,2]dioxaborole; (S)-1-(2-(diphenylphosphanyl)naphthalen-1-yl)isoquinoline In dichloromethane at 20℃; for 12h; Stage #2: With diethylzinc In toluene at 20℃; for 2h; Stage #3: N-chloromethylamine In diethyl ether; water at 0℃; for 0.0833333h; Further stages.; |

-
-
312620-93-2
(S)-N-(4-(3,4-dichlorophenyl)-3,4-dihydronaphthalen-1(2H)-ylidene)methanamine

-
A
-
79951-46-5
trans-(1R,4S)-N-methyl-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-1-naphthalenamine

-
B
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With hydrogen; nickel In methanol | |
| With C32H12BF24(1-)*C36H44IrNOP(1+); hydrogen at 20℃; under 15201 Torr; for 12h; optical yield given as %ee; enantioselective reaction; |

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: magnesium / tetrahydrofuran 1.2: CuBr-SMe2 / 13 / -30 - 0 °C 2.1: sodium tetrahydroborate / tetrahydrofuran; water 3.1: triethylamine 4.1: sodium iodide / acetone / Heating 5.1: tetrahydrofuran 6.1: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: magnesium / tetrahydrofuran 1.2: CuBr-SMe2 / 13 / -30 - 0 °C 2.1: sodium tetrahydroborate / tetrahydrofuran; water 3.1: 1H-imidazole; iodine; triphenylphosphine 4.1: hydrogenchloride 5.1: tetrahydrofuran 6.1: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme |

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: sodium tetrahydroborate / tetrahydrofuran; water 2: triethylamine 3: sodium iodide / acetone / Heating 4: tetrahydrofuran 5: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme | |
| Multi-step reaction with 5 steps 1: sodium tetrahydroborate / tetrahydrofuran; water 2: 1H-imidazole; iodine; triphenylphosphine 3: hydrogenchloride 4: tetrahydrofuran 5: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme |
-
-
234448-56-7
C18H20Cl2O3

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 1H-imidazole; iodine; triphenylphosphine 2: hydrogenchloride 3: tetrahydrofuran 4: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme | |
| Multi-step reaction with 4 steps 1: triethylamine 2: sodium iodide / acetone / Heating 3: tetrahydrofuran 4: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme |

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: hydrogenchloride 2: tetrahydrofuran 3: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme |

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: NaI / acetone / Heating 2: 100 percent / tetrahydrofuran 3: 69 percent / t-BuLi / tetrahydrofuran; toluene / -78 °C View Scheme |
-
-
234448-57-8
C16H13Cl2IO

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrahydrofuran 2: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme |
-
-
35849-09-3
o-bromobenzaldehyde dimethyl acetal

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: magnesium / tetrahydrofuran 1.2: CuBr-SMe2 / 13 / -30 - 0 °C 2.1: sodium tetrahydroborate / tetrahydrofuran; water 3.1: triethylamine 4.1: sodium iodide / acetone / Heating 5.1: tetrahydrofuran 6.1: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: magnesium / tetrahydrofuran 1.2: CuBr-SMe2 / 13 / -30 - 0 °C 2.1: sodium tetrahydroborate / tetrahydrofuran; water 3.1: 1H-imidazole; iodine; triphenylphosphine 4.1: hydrogenchloride 5.1: tetrahydrofuran 6.1: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme |
-
-
7312-27-8
3,4-Dichlorocinnamic acid

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: pivaloyl chloride; triethylamine; lithium chloride 2.1: magnesium / tetrahydrofuran 2.2: CuBr-SMe2 / 13 / -30 - 0 °C 3.1: sodium tetrahydroborate / tetrahydrofuran; water 4.1: triethylamine 5.1: sodium iodide / acetone / Heating 6.1: tetrahydrofuran 7.1: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: pivaloyl chloride; triethylamine; lithium chloride 2.1: magnesium / tetrahydrofuran 2.2: CuBr-SMe2 / 13 / -30 - 0 °C 3.1: sodium tetrahydroborate / tetrahydrofuran; water 4.1: 1H-imidazole; iodine; triphenylphosphine 5.1: hydrogenchloride 6.1: tetrahydrofuran 7.1: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme |
-
-
6630-33-7
ortho-bromobenzaldehyde

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: toluene-4-sulfonic acid / methanol 2.1: magnesium / tetrahydrofuran 2.2: CuBr-SMe2 / 13 / -30 - 0 °C 3.1: sodium tetrahydroborate / tetrahydrofuran; water 4.1: triethylamine 5.1: sodium iodide / acetone / Heating 6.1: tetrahydrofuran 7.1: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: toluene-4-sulfonic acid / methanol 2.1: magnesium / tetrahydrofuran 2.2: CuBr-SMe2 / 13 / -30 - 0 °C 3.1: sodium tetrahydroborate / tetrahydrofuran; water 4.1: 1H-imidazole; iodine; triphenylphosphine 5.1: hydrogenchloride 6.1: tetrahydrofuran 7.1: tert.-butyl lithium / tetrahydrofuran; toluene / -78 °C View Scheme |
-
-
79560-19-3
4-(3,4-dichlorophenyl)-3,4-dihydro-2H-naphthalen-1-one

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: BH3*Me2S / (S)-prolinol core-containing second-generation dendrimer / tetrahydrofuran / 0.8 h 2: PCC / CH2Cl2 / 0 - 20 °C 3: TiCl4 / diethyl ether / -78 - 20 °C 4: H2; Raney-Ni / methanol View Scheme | |
| Multi-step reaction with 2 steps 1: 91 percent / TiCl4 / diethyl ether / -35 - 20 °C 2: (S,S)-(ethylenebis(tetrahydroindenyl))TiF2; PhSiH3 / tetrahydrofuran / 24 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: TiCl4 / toluene / 17 h / Ambient temperature 2: sodium borohydride / methanol / 1.5 h / Ambient temperature View Scheme |
-
-
79560-19-3, 79836-44-5, 124379-29-9
(4S)-4-(3,4-dichlorophenyl)-1-tetralone

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TiCl4 / diethyl ether / -78 - 20 °C 2: H2; Raney-Ni / methanol View Scheme | |
| Multi-step reaction with 4 steps 1: NaBH4 / ethanol 2: 88 percent / dppa, DBU / tetrahydrofuran 3: H2 / Pd-C / ethanol 4: 1) K2CO3, 2) LiAlH(OMe)3 / 1) MeCN, 2) THF, reflux, 40 h View Scheme | |
| Multi-step reaction with 2 steps 1: methanol / 6 h / Reflux 2: sodium tetrahydroborate; methanol View Scheme |
-
-
167026-40-6
(1R,4S)-4-(3,4-dichloro-phenyl)-1,2,3,4-tetrahydro-naphthalen-1-ol

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: PCC / CH2Cl2 / 0 - 20 °C 2: TiCl4 / diethyl ether / -78 - 20 °C 3: H2; Raney-Ni / methanol View Scheme | |
| Multi-step reaction with 4 steps 1: 88 percent / diphenylphosphorylazide; DBU / tetrahydrofuran / 0 - 20 °C 2: H2 / Pd/C / ethanol / 1 h 3: K2CO3 / acetonitrile / 2 h / Heating 4: 10 mg / LiAlH(OMe)3 / tetrahydrofuran / 40 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: 88 percent / dppa, DBU / tetrahydrofuran 2: H2 / Pd-C / ethanol 3: 1) K2CO3, 2) LiAlH(OMe)3 / 1) MeCN, 2) THF, reflux, 40 h View Scheme |
-
-
439134-46-0
1-(3,4-dichlorophenyl)-1,2-dihydronaphthalene

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: catecholborane / (S)-QUINAP-Rh / toluene / 2 h / 20 °C 1.2: aq. H2O2; flash silica / hexane / 1 h / 0 °C 2.1: catecholborane / (S)-QUINAP-Rh / CH2Cl2 / 12 h / 20 °C 2.2: ZnEt2 / toluene / 2 h / 20 °C 2.3: H2O; diethyl ether / 0.08 h / 0 °C View Scheme |
-
-
174023-92-8
D-dibenzylaminophenylglycinal

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 2.62 g / benzene; CH2Cl2 / 15 h / 20 °C 2: 83 percent / Mg / 4 h 3: 76 percent / LiAlH4 / tetrahydrofuran / 0 - 20 °C 4: 0.85 g / pyridinium dichromate / CH2Cl2 / 5 h / 20 °C 5: 1.0 g / tetrahydrofuran / 5 h 6: 78 percent / AlCl3 / CH2Cl2 / 1 h / 20 °C 7: H2 / Pd(OH)2 / methanol / 3 h 8: 0.162 g / methanol / 3 h 9: 84 percent / NaH / tetrahydrofuran / 6 h 10: 85 percent / TFA / CH2Cl2 / 1 h / 20 °C View Scheme |
-
-
193476-02-7
(+)-(2R)-2-[bis(phenylmethyl)amino]-2-phenylethanol

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1: DMSO; oxalyl chloride; Et3N / CH2Cl2 / -78 - 0 °C 2: 2.62 g / benzene; CH2Cl2 / 15 h / 20 °C 3: 83 percent / Mg / 4 h 4: 76 percent / LiAlH4 / tetrahydrofuran / 0 - 20 °C 5: 0.85 g / pyridinium dichromate / CH2Cl2 / 5 h / 20 °C 6: 1.0 g / tetrahydrofuran / 5 h 7: 78 percent / AlCl3 / CH2Cl2 / 1 h / 20 °C 8: H2 / Pd(OH)2 / methanol / 3 h 9: 0.162 g / methanol / 3 h 10: 84 percent / NaH / tetrahydrofuran / 6 h 11: 85 percent / TFA / CH2Cl2 / 1 h / 20 °C View Scheme |
-
-
87857-41-8
norsertraline

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 0.162 g / methanol / 3 h 2: 84 percent / NaH / tetrahydrofuran / 6 h 3: 85 percent / TFA / CH2Cl2 / 1 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: K2CO3 / acetonitrile / 2 h / Heating 2: 10 mg / LiAlH(OMe)3 / tetrahydrofuran / 40 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| In ethyl acetate at 20℃; | 99% |
| In tetrahydrofuran |
-
-
87-69-4, 133-37-9, 147-71-7, 147-73-9, 526-83-0
L-tartaric acid

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| In ethyl acetate | 98% |

| Conditions | Yield |
|---|---|
| In ethanol; ethyl acetate at 25 - 30℃; for 5h; | 98% |
-
-
79617-96-2
Sertraline

-
-
107-13-1
acrylonitrile

| Conditions | Yield |
|---|---|
| With sodium In methanol at 55℃; for 12h; Inert atmosphere; | 98% |
-
-
79617-96-2
Sertraline

-
-
77-92-9
citric acid

-
-
836597-67-2
(1S-cis)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphtalenamine citrate

| Conditions | Yield |
|---|---|
| In water; ethyl acetate at 0 - 60℃; for 1h; Product distribution / selectivity; | 97% |
| In propan-1-ol; isopropyl alcohol at 0 - 40℃; for 2h; Product distribution / selectivity; | 96% |

| Conditions | Yield |
|---|---|
| In ethyl acetate for 48h; | 96% |

| Conditions | Yield |
|---|---|
| In ethyl acetate at 20℃; for 4h; | 96% |
-
-
79617-96-2
Sertraline

-
-
79559-97-0
sertraline hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In propan-1-ol at 30℃; for 1h; Product distribution / selectivity; | 96% |
| With hydrogenchloride In acetonitrile for 1h; Product distribution / selectivity; | 95% |
| With hydrogenchloride In octanol for 1h; Product distribution / selectivity; | 95% |
-
-
611-71-2
(R)-Mandelic Acid

-
-
79617-96-2
Sertraline

-
-
244223-89-0, 79617-97-3
(cis-1S)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthalenamine (R)-mandelate

| Conditions | Yield |
|---|---|
| In ethanol Reflux; | 94% |

| Conditions | Yield |
|---|---|
| In ethanol; ethyl acetate at 25 - 30℃; for 5h; | 92% |
-
-
17199-29-0
(S)-Mandelic acid

-
-
79617-96-2
Sertraline

-
-
1357380-69-8
(1S,4S)-sertraline*(S)-mandelic acid

| Conditions | Yield |
|---|---|
| In ethanol; hexane Reflux; | 91% |

| Conditions | Yield |
|---|---|
| In ethyl acetate at 20℃; | 88% |
| In ethanol |
-
-
455-24-3
4-trifluoromethylbenzoic acid

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With potassium phosphate; 18-crown-6 ether; phenylsilane In tetrahydrofuran at 80℃; for 8h; Glovebox; Molecular sieve; Schlenk technique; | 88% |

| Conditions | Yield |
|---|---|
| In ethyl acetate at 25 - 30℃; for 5h; | 86% |
-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With tris(pentafluorophenyl)borate; phenylsilane at 120℃; for 24h; Inert atmosphere; Sealed tube; Glovebox; | 85% |

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| Stage #1: tert-butyl ((1H-pyrazol-1-yl)(2,2,2-trifluoroacetamido)methylene)carbamate; Sertraline In tetrahydrofuran at 20℃; Inert atmosphere; Stage #2: With potassium hydrogencarbonate In tetrahydrofuran; methanol Inert atmosphere; | 83% |
-
-
79617-96-2
Sertraline

-
-
75-05-8
acetonitrile

-
-
122873-20-5
(1S,4S)-4-(3,4-dichlorophenyl)-N,N-dimethyl-1,2,3,4-tetrahydro naphthalen-1-amine

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; carbon dioxide at 80℃; under 750.075 Torr; for 24h; Inert atmosphere; Schlenk technique; Glovebox; | 83% |
-
-
79617-96-2
Sertraline

-
-
76-05-1
trifluoroacetic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; 18-crown-6 ether; phenylsilane In tetrahydrofuran at 80℃; for 8h; Glovebox; Molecular sieve; Schlenk technique; | 81% |
-
-
124-38-9
carbon dioxide

-
-
79617-96-2
Sertraline

| Conditions | Yield |
|---|---|
| With cyclopentadienyl iron(II) dicarbonyl dimer; tributylphosphine; phenylsilane In acetonitrile at 30℃; under 760.051 Torr; for 36h; Schlenk technique; Sealed tube; | 80% |
| With phenylsilane; tetrabutyl ammonium fluoride at 20℃; for 4h; | 60% |
-
-
79617-96-2
Sertraline

-
-
291306-20-2
(1S-cis )-4-(3,4-dichlorophenyl)-7-iodo-1,2,3,4-tetrahydro-N-methyl-1-naphthalenamine

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; trifluorormethanesulfonic acid In dichloromethane for 23h; | 79% |

| Conditions | Yield |
|---|---|
| In ethanol; ethyl acetate at 25 - 30℃; for 5h; | 78% |
-
-
124-38-9
carbon dioxide

-
-
79617-96-2
Sertraline

-
-
122873-20-5
(1S,4S)-4-(3,4-dichlorophenyl)-N,N-dimethyl-1,2,3,4-tetrahydro naphthalen-1-amine

| Conditions | Yield |
|---|---|
| With cyclopentadienyl iron(II) dicarbonyl dimer; phenylsilane; triphenylphosphine In acetonitrile at 100℃; under 760.051 Torr; for 24h; Schlenk technique; Sealed tube; | 78% |
| With diphenylsilane; [1,3-bis(2,4,6-trimethylphenyl)imidazol]-2-ylidene In N,N-dimethyl-formamide at 50℃; under 760.051 Torr; chemoselective reaction; | 75% |

| Conditions | Yield |
|---|---|
| In ethanol; ethyl acetate at 25 - 30℃; for 5h; | 76% |

| Conditions | Yield |
|---|---|
| In ethyl acetate at 25 - 30℃; for 5h; | 67% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 Xi
Xi