-
Name
(+)-Diisopropyl L-tartrate
- EINECS 218-709-0
- CAS No. 2217-15-4
- Article Data4
- CAS DataBase
- Density 1.188 g/cm3
- Solubility
- Melting Point
- Formula C10H18O6
- Boiling Point 268.1 °C at 760 mmHg
- Molecular Weight 234.249
- Flash Point 109.4 °C
- Transport Information
- Appearance clear, colorless liquid
- Safety 24/25-36-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Butanedioicacid, 2,3-dihydroxy- (2R,3R)-, bis(1-methylethyl) ester (9CI);Butanedioic acid,2,3-dihydroxy-[R-(R*,R*)]-, bis(1-methylethyl) ester;Tartaric acid,diisopropyl ester (5CI);Tartaric acid, diisopropyl ester, (+)- (8CI);(+)-DIPT;(+)-Tartaric acid diisopropyl ester;Diisopropyl(+)-L-tartrate;Diisopropyl (2R,3R)-tartrate;Diisopropyl L-tartrate;Diisopropyl tartrate;L-(+)-Tartaric acid diisopropylester;
- PSA 93.06000
- LogP -0.38860
Synthetic route

| Conditions | Yield |
|---|---|
| With indium; iodine for 7h; transesterification; Heating; | 87% |

| Conditions | Yield |
|---|---|
| With calcium chloride |
-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
18680-27-8
pinanediol

-
A
-
2217-15-4
L-(+)-diisopropyl tartrate

-
B
-
76110-78-6
(3aS,4S,6S,7aR)-3a,5,5-trimethyl-2-phenylhexahydro-4,6-methanobenzo[d][1,3,2]dioxaborole

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.25h; Title compound not separated from byproducts.; | A n/a B 99 % Spectr. |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
504-63-2
trimethyleneglycol

-
A
-
4406-77-3
2-Phenyl-1,3,2-dioxaborinane

-
B
-
2217-15-4
L-(+)-diisopropyl tartrate

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.1h; Title compound not separated from byproducts.; | A 99 % Spectr. B n/a |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
126-30-7
2,2-Dimethyl-1,3-propanediol

-
A
-
2217-15-4
L-(+)-diisopropyl tartrate

-
B
-
5123-13-7
5,5-dimethyl-2-phenyl-1,3,2-dioxaborinane

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.1h; Title compound not separated from byproducts.; | A n/a B 99 % Spectr. |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
24892-49-7, 139687-48-2
2,4-dimethylpentane-2,4-diol

-
A
-
2217-15-4
L-(+)-diisopropyl tartrate

-
B
-
95843-97-3
2,4-dimethyl-2,4-pentanediol phenylboronic ester

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 3h; Title compound not separated from byproducts.; | A n/a B 99 % Spectr. |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
111-42-2
2,2'-iminobis[ethanol]

-
A
-
2217-15-4
L-(+)-diisopropyl tartrate

-
B
-
4406-73-9
diethanolamine phenylboronic ester

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.1h; Title compound not separated from byproducts.; | A n/a B 99 % Spectr. |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
2160-93-2
N-tert-butyldiethanolamine

-
A
-
2217-15-4
L-(+)-diisopropyl tartrate

-
B
-
73029-08-0
N-tert-butyldiethanolamine phenylboronic ester

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.1h; Title compound not separated from byproducts.; | A n/a B 99 % Spectr. |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
1195-59-1
2.6-bis(hydroxymethyl)pyridine

-
B
-
2217-15-4
L-(+)-diisopropyl tartrate

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.1h; Title compound not separated from byproducts.; | A 90 % Spectr. B n/a |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
19132-06-0
(2S,3S)-butane-2,3-diol

-
B
-
2217-15-4
L-(+)-diisopropyl tartrate

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.1h; Title compound not separated from byproducts.; | A 98 % Spectr. B n/a |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
A
-
2217-15-4
L-(+)-diisopropyl tartrate

-
B
-
24388-23-6
2-phenyl-4,4,5,5-tetramethyl-1,3,2-dioxoborole

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 48h; Title compound not separated from byproducts.; | A n/a B 99 % Spectr. |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
1792-81-0
cis-1,2-cyclohexane

-
A
-
2217-15-4
L-(+)-diisopropyl tartrate

-
B
-
6638-70-6
(3aR,7aS)-2-phenyl-hexahydro-2H-1,3,2-benzodioxaborole

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 20h; Title compound not separated from byproducts.; | A n/a B 92 % Spectr. |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
5057-98-7
cis-1,2-cyclopentanediol

-
A
-
2217-15-4
L-(+)-diisopropyl tartrate

-
B
-
7462-37-5
(3aR,6aS)-2-phenyl-hexahydrocyclopenta[d][1,3,2]dioxaborole

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.25h; Title compound not separated from byproducts.; | A n/a B 99 % Spectr. |

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

-
-
107-21-1
ethylene glycol

-
A
-
4406-72-8
2-phenyl[1,3,2]dioxaborolane

-
B
-
2217-15-4
L-(+)-diisopropyl tartrate

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 0.1h; Title compound not separated from byproducts.; | A 99 % Spectr. B n/a |


| Conditions | Yield |
|---|---|
| With thionyl chloride at 20℃; |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
136029-38-4
(4R,5R)-2-Oxo-2λ4-[1,3,2]dioxathiolane-4,5-dicarboxylic acid diisopropyl ester

| Conditions | Yield |
|---|---|
| With thionyl chloride; N,N-dimethyl-formamide at 50℃; for 1h; | 100% |
| With thionyl chloride In tetrachloromethane for 1h; Heating; | 96% |
| With pyridine; thionyl chloride In dichloromethane at 0℃; |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
147488-89-9
(2R,3R)-2,3-Bis-(tert-butyl-dimethyl-silanyloxy)-succinic acid diisopropyl ester

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 60℃; for 12h; | 100% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
83540-97-0
(2R)-diisopropyl 2-hydroxybutanedioate

| Conditions | Yield |
|---|---|
| With samarium diiodide; ethylene glycol In tetrahydrofuran for 1h; Ambient temperature; | 99% |

-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
99687-40-8, 106357-20-4, 106357-33-9, 99745-86-5
(R,R)-[(E)-2-butenyl]diisopropyl tartrate boronate

| Conditions | Yield |
|---|---|
| With sodium chloride In diethyl ether stirred under nitrogen for 5 min; extraction of aqueous layer with Et2O; organic extracts combined; dried over MgSO4; filtration; concn.; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: L-(+)-diisopropyl tartrate With sodium periodate In water at 0℃; for 2h; Stage #2: benzoic acid hydrazide In ethanol for 12h; | 99% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
77-76-9
2,2-dimethoxy-propane

-
-
81327-47-1
diisopropyl (2S,3S)-2,3-O-isopropylidenetartrate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene for 3h; Heating; | 97% |
| In various solvent(s) Heating; | 82% |

| Conditions | Yield |
|---|---|
| Stage #1: L-(+)-diisopropyl tartrate With sodium hydride In tetrahydrofuran at 0 - 25℃; for 1h; Metallation; Stage #2: allyl bromide With 18-crown-6 ether; tetra-(n-butyl)ammonium iodide In tetrahydrofuran at 0 - 25℃; for 4h; Etherification; | 97% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
130678-42-1
Di(1-methylethyl)-(2R,3R)-2,3-bis<(trimethylsilyl)oxy>butandioat

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane a) 0 deg C, 10 min, b) r.t., 5h; | 96% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
78-39-7
Triethyl orthoacetate

-
-
130258-01-4
diisopropyl (4R,5R)-2-ethoxy-2-methyl-1,3-dioxolan-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With sulfuric acid at 130℃; for 3h; | 96% |

| Conditions | Yield |
|---|---|
| With sodium periodate In diethyl ether; water at 0℃; for 2.33333h; | 95% |
| With sodium periodate In water at 0℃; for 2h; Inert atmosphere; | 60% |
| With sodium periodate at 0℃; for 2h; |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
25226-98-6
trans-cinnamaldehyde diethylacetal

-
-
99267-72-8
(4R,5R)-4,5-Bis((2-methylethoxy)carbonyl)-2-(trans-2-phenylethenyl)-1,3-dioxolane

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In benzene for 1h; Heating; | 93% |
-
-
12636-72-5
bis(cyclopentadienyl)dimethylzirconium(IV)

-
-
2217-15-4
L-(+)-diisopropyl tartrate

| Conditions | Yield |
|---|---|
| In dichloromethane evapd., twice recrystd. from toluene;; | 93% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
100-39-0
benzyl bromide

-
-
139631-40-6
*,R*)>-bis(1-methylethyl) 2,3-bis(phenylmethoxy)butanedioate

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; | 92% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
188691-14-7
dihydroxyacetic acid isopropyl ester

| Conditions | Yield |
|---|---|
| With sodium periodate; silica gel In dichloromethane for 1.5h; Ambient temperature; | 90% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 130℃; for 3h; | 88% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
104-54-1
3-Phenylpropenol

-
-
98819-68-2
3-phenyl-(2R,3R)-oxiran-2-ylmethanol

| Conditions | Yield |
|---|---|
| Stage #1: L-(+)-diisopropyl tartrate With titanium(IV) isopropylate In dichloromethane at 300℃; for 0.0833333h; Inert atmosphere; Molecular sieve; Stage #2: 3-Phenylpropenol In dichloromethane for 1h; Inert atmosphere; | 88% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; | 86% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; | 85% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
106357-20-4
(R,R)-diisopropyl tartrate (Z)-crotylboronate

| Conditions | Yield |
|---|---|
| With sodium chloride In diethyl ether; water | 85% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
99687-40-8, 106357-20-4, 106357-33-9, 99745-86-5
(R,R)-[(E)-2-butenyl]diisopropyl tartrate boronate

| Conditions | Yield |
|---|---|
| With sodium chloride In diethyl ether; water | 85% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
79-04-9
chloroacetyl chloride

-
-
226710-52-7
(2R,3R)-2,3-Bis-(2-chloro-acetoxy)-succinic acid diisopropyl ester

| Conditions | Yield |
|---|---|
| With pyridine In chloroform for 5h; Ambient temperature; | 84% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
532-55-8
Benzoyl isothiocyanate

-
-
253350-02-6
(4R,5R)-4,5-di(isopropyloxycarbonyl)-3-benzoyloxazolidin-2-one

| Conditions | Yield |
|---|---|
| Stage #1: L-(+)-diisopropyl tartrate With di(n-butyl)tin oxide In 1,2-dichloro-ethane for 4h; Heating; Stage #2: Benzoyl isothiocyanate With triethylamine In 1,2-dichloro-ethane for 1h; Heating; Stage #3: With tetrabutylammomium bromide In 1,2-dichloro-ethane for 2h; Heating; Further stages.; | 84% |

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; | 83% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
129423-29-6
[(1E)-3-phenylprop-1-en-1-yl]boronic acid

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 83% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
105-88-4
α.ε.ι-trimethyl-undecylaldehyde

-
-
130678-57-8, 130793-54-3, 130793-55-4, 130793-59-8
Di(1-methylethyl)-(4R,5R)-2-<(1RS,5RS)-1,5,9-trimethyldecyl>-1,3-dioxolan-4,5-dicarboxylat

| Conditions | Yield |
|---|---|
| toluene-4-sulfonic acid In toluene Heating; | 82% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
149-73-5
trimethyl orthoformate

-
-
649721-37-9
(4R,5R)-di-isopropyl-2-methoxy-1,3-dioxolane-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With sulfuric acid In toluene Heating; | 81% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
98-80-6
phenylboronic acid

-
-
1061180-99-1
(4R,5R)-diisopropyl-2-phenyl-1,3,2-dioxaborolane-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| In toluene byproducts: H2O; addn. of diisopropyl tartrate to toluene soln. of boronic acid deriv., refluxing for 24 h in Dean-Stark app.; cooling to room temp., drying, filtration, evapn. in vac., recrystn. (hexane), elem. anal.; | 81% |
| In pentane at 20℃; |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 130℃; for 3h; | 80% |
-
-
2217-15-4
L-(+)-diisopropyl tartrate

-
-
80522-42-5
triisopropylsilyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With 2,5-dimethylpyridine In dichloromethane at 21℃; | 80% |
(+)-Diisopropyl L-tartrate Chemical Properties
Molecular structure of Tartaric acid, diisopropyl ester (CAS NO.2217-15-4) is:
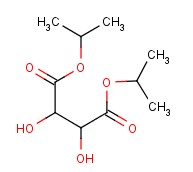
Product Name: Tartaric acid, diisopropyl ester
CAS Registry Number: 2217-15-4
IUPAC Name: dipropan-2-yl 2,3-dihydroxybutanedioate
Molecular Weight: 234.24632 [g/mol]
Molecular Formula: C10H18O6
XLogP3: 0.6
H-Bond Donor: 2
H-Bond Acceptor: 6
EINECS: 218-709-0
Sensitive: Hygroscopic
Refractive index: 1.438-1.44
Surface Tension: 42.2 dyne/cm
Density: 1.188 g/cm3
Flash Point: 109.4 °C
Enthalpy of Vaporization: 58.77 kJ/mol
Boiling Point: 268.1 °C at 760 mmHg
Vapour Pressure: 0.00106 mmHg at 25°C
Product Categories: chiral;API intermediates;Asymmetric Synthesis;Chiral Building Blocks;Esters (Chiral);Synthetic Organic Chemistry;Chiral Chemicals;Hydroxy Acids & Deriv.;Chiral Compound
(+)-Diisopropyl L-tartrate Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 6300uL/kg (6.3mL/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 93, Pg. 26, 1948. | |
| mouse | LD50 | skin | > 10mL/kg (10mL/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 93, Pg. 26, 1948. | |
| rat | LD50 | oral | 6mL/kg (6mL/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 93, Pg. 26, 1948. |
(+)-Diisopropyl L-tartrate Safety Profile
Safty information about Tartaric acid, diisopropyl ester (CAS NO.2217-15-4) is:
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 24/25-36-26
S24/25:Avoid contact with skin and eyes.
S36:Wear suitable protective clothing.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
WGK Germany: 3
RTECS: WW8100000
HS Code: 29181300
(+)-Diisopropyl L-tartrate Specification
Tartaric acid, diisopropyl ester , its cas register number is 2217-15-4. It also can be called Diisopropyl tartrate ; Diisopropyl L-(+)-tartarate ; (+)-Diisopropyl L-tartrate .It is a clear, colorless liquid.
Related Products
- (+)-Diisopropyl L-tartrate
- 22172-17-4
- 2217-32-5
- 2217-33-6
- 2217-35-8
- 2217-40-5
- 2217-41-6
- 22174-29-4
- 2217-43-8
- 22174-70-5
- 22177-92-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View