-
Name
(+)-Pilocarpine hydrochloride
- EINECS 200-212-5
- CAS No. 54-71-7
- Article Data5
- CAS DataBase
- Density 1.22 g/cm3
- Solubility 100 mg/mL in water
- Melting Point 199-204 °C
- Formula C11H16N2O2.HCl
- Boiling Point 431.8 °C at 760 mmHg
- Molecular Weight 244.721
- Flash Point 215 °C
- Transport Information UN 1544 6.1/PG 3
- Appearance white crystalline solid
- Safety 25-45
- Risk Codes 26/28
-
Molecular Structure
-
Hazard Symbols
 T+
T+
- Synonyms 2(3H)-Furanone,3-ethyldihydro-4-[(1-methyl-1H-imidazol-5-yl)methyl]-, monohydrochloride,(3S,4R)- (9CI);2(3H)-Furanone,3-ethyldihydro-4-[(1-methyl-1H-imidazol-5-yl)methyl]-, monohydrochloride,(3S-cis)-;Pilocarpine, monohydrochloride (8CI);(+)-Pilocarpine hydrochloride;Adsorbocarpine;Almocarpine;Isopto Carpine;NSC 5746;Pilocar;Pilocar SMP;Pilocarpal;Pilocarpine chloride;Pilocarpine hydrochloride;Pilocarpinemuriate;Pilocarpinium chloride;Pilocel;Pilovisc;Salagen;Sanpilo;Vistacarpin N;
- PSA 44.12000
- LogP 1.96380
Synthetic route

-
-
54-71-7
pilocarpine hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium tetrahydroborate 1a.) iPrOH, -5 deg C, 1 h, 1b.) RT, 21 h, 2.) MeOH, water, 2 h; Yield given. Multistep reaction; |

-
-
54-71-7
pilocarpine hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 100 percent / H2 / Pd/C (5percent) / methanol / 60 h / 3102.9 Torr 2: 1.) NaBH4, 2.) conc. aq. HCl / 1a.) iPrOH, -5 deg C, 1 h, 1b.) RT, 21 h, 2.) MeOH, water, 2 h View Scheme |

-
-
54-71-7
pilocarpine hydrochloride

| Conditions | Yield |
|---|---|
| With lithium borohydride; sodium hydrogencarbonate In hydrogenchloride; methanol; water; isopropyl alcohol |
-
-
1192231-13-2
(R)-3-ethylidene-4-[(1-methylimidazol-5-yl)methyl]tetrahydrofuran-2-one

-
-
54-71-7
pilocarpine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: (R)-3-ethylidene-4-[(1-methylimidazol-5-yl)methyl]tetrahydrofuran-2-one With platinum(IV) oxide; hydrogen In methanol at 20℃; for 24h; Stage #2: With hydrogenchloride In ethanol; water at 0℃; Cooling; |

-
-
54-71-7
pilocarpine hydrochloride

| Conditions | Yield |
|---|---|
| With Na2CO3 In ethanol; water; ethyl acetate organic ligand in water alkalinized with Na2CO3 to pH=9; extracted with ethyl acetate; overlayered with 0.5 equivalent CoCl2*6H2O in EtOH; allowed to evaporate slowly at room temp. for 2 days; elem. anal.; | 97% |
-
-
10417-94-4
all cis-5,8,11,14,17-eicosapentaenoic acid

-
-
54-71-7
pilocarpine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: pilocarpine hydrochloride With sodium hydrogencarbonate In methanol for 0.0833333h; Stage #2: all cis-5,8,11,14,17-eicosapentaenoic acid In methanol at 20℃; for 16h; | 96.8% |

| Conditions | Yield |
|---|---|
| Stage #1: pilocarpine hydrochloride With sodium hydrogencarbonate In acetone at 20℃; for 0.0833333h; Stage #2: (R)-1,2-dithiolane-3-pentanoic acid In acetone at 20℃; for 16h; | 91.58% |

| Conditions | Yield |
|---|---|
| With Na2CO3 In chloroform; water; butan-1-ol organic ligand in water alkalinized with Na2CO3 to pH=9; extracted with CHCl3; removed in vacuo; dissolved in butanol; 0.5 equivalent of CuCl2 in butanol added; heated for a few minutes; sepd. by filtration; crystals grown from acetone; elem. anal.; | 14% |
-
-
54-71-7
pilocarpine hydrochloride

-
-
117639-12-0
3-ethyl-4,5-dihydro-4-<(1-methyl-1H-imidazol-5-yl)methyl>-2(3H)-thiophenethione

| Conditions | Yield |
|---|---|
| With Lawessons reagent In xylene at 135℃; for 7h; | 4.63 g |
-
-
54-71-7
pilocarpine hydrochloride

-
A
-
101769-87-3
sodium isopilocarpate

-
B
-
92598-79-3
sodium pilocarpate

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 4℃; for 1h; Yield given; |
-
-
54-71-7
pilocarpine hydrochloride

-
A
-
531-35-1
isopilocarpine

-
B
-
28406-15-7
Pilocarpinsaeure

-
C
-
34350-99-7
Isopilocarpinsaeure

| Conditions | Yield |
|---|---|
| With potassium chloride In water for 1h; Kinetics; Thermodynamic data; Mechanism; Heating; var. pH-value, temp., times; epimerisation; |

-
-
54-71-7
pilocarpine hydrochloride

| Conditions | Yield |
|---|---|
| Erhitzen auf Siedetemperatur; |
-
-
54-71-7
pilocarpine hydrochloride

-
-
62-53-3
aniline

-
-
92-13-7, 531-35-1, 4354-74-9, 35594-23-1, 48148-73-8, 54163-69-8, 102282-25-7
(+-)-isopilocarpine

| Conditions | Yield |
|---|---|
| at 205 - 210℃; |


| Conditions | Yield |
|---|---|
| for 40h; Benzylation; Heating; |


| Conditions | Yield |
|---|---|
| Stage #1: pilocarpine hydrochloride With sodium hydroxide Stage #2: With hydrogenchloride |
-
-
54-71-7
pilocarpine hydrochloride

-
-
28406-15-7
Pilocarpinsaeure

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 90℃; for 2h; |
-
-
54-71-7
pilocarpine hydrochloride

-
-
34350-99-7
Isopilocarpinsaeure

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: aq. NaOH 1.2: aq. HCl 2.1: aq. NH3 / 2 h / 90 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: aq. NH3 / 2 h / 90 °C 2.1: aq. NaOH 2.2: aq. HCl View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
137034-76-5
(3R,4R)-3-ethyl-4-[(1-methyl-1H-imidazol-5-yl)-methyl]-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 40 h / Heating 2: Li; PrNH2; H2NCH2CH2NH2 / 2-methyl-propan-2-ol / 4 h / 0 °C View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
96914-10-2
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid ethyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 27 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
96914-11-3
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid hexyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 36 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
92598-80-6
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 30 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
92598-82-8
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid benzyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 35 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
92598-86-2
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid phenethyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 22 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
92598-93-1
pilocarpic acid 4-methylbenzyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 40 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
92598-81-7
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid 1-phenyl-ethyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 35 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
92598-92-0
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid 2-methyl-benzyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 35 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
92598-89-5
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid 4-chloro-benzyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 43 percent / dimethylformamide / Ambient temperature View Scheme |
-
-
54-71-7
pilocarpine hydrochloride

-
-
92598-99-7
(2S,3R)-2-Ethyl-3-hydroxymethyl-4-(3-methyl-3H-imidazol-4-yl)-butyric acid 4-tert-butyl-benzyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NaOH / H2O / 1 h / 0 - 4 °C 2: 18 percent / dimethylformamide / Ambient temperature View Scheme |
(+)-Pilocarpine hydrochloride Chemical Properties
The Molecular Weight of PILOCARPINE MONOHYDROCHLORIDE(54-71-7):244.72
The Molecular Structure of PILOCARPINE MONOHYDROCHLORIDE(54-71-7) is:
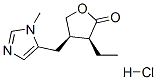
Melting point:202-205 °C(lit.)
Boiling point:431.8 °C at 760 mmHg
Flash point:215 °C
Solubility in water:100 mg/mL
Sensitive:Hygroscopic
Merck:14,7424
Enthalpy of Vaporization:68.75 kJ/mol
Vapour Pressure:1.16E-07 mmHg at 25°C
Appearance:crystalline Solid
IUPAC Name:(3S,4R)-3-ethyl-4-[(3-methylimidazol-4-yl)methyl]oxolan-2-one hydrochloride
Synonyms:ALMOCARPINE HYDROCHLORIDE;3-ETHYL-4-[(1-METHYL-5-IMIDAZOLYL)METHYL]TETRAHYDROFURAN-2-ONE HYDROCHLORIDE;(3S,4R)-4,5-DIHYDRO-3-ETHYL-4-(1-METHYL-1H-IMIDAZOL-5-YLMETHYL)-2(3H)-FURANONE HYDROCHLORIDE;(3S-CIS)-3-ETHYLDIHYDRO-4-[(1-METHYL-1H-IMIDAZOL-5-YL)METHYL]-2(3H)-FURANONE HYDROCHLORIDE;PILOCARPINE HCL;(+)-PILOCARPINE HYDROCHLORIDE;PILOCARPINE HYDROCHLORIDE;(3s-cis)-ochlorid
(+)-Pilocarpine hydrochloride Uses
(+)-Pilocarpine hydrochloride Toxicity Data With Reference
| 1. | ocu-man TDLo:200 µg/kg/7H-I:CVS | AIMDAP Archives of Internal Medicine. 147 (1987),586. | ||
| 2. | orl-mus LD50:200 mg/kg | NIIRDN “Drugs in Japan. Ethical Drugs, 6th Edition 1982“ Edited by Japan Pharmaceutical Information Center. 6 (1982),646. | ||
| 3. | ipr-mus LD50:155 mg/kg | ATXKA8 Archiv fuer Toxikologie. 29 (1972),39. | ||
| 4. | ivn-pgn LDLo:353 mg/kg | HBAMAK “Abdernalden’s Handbuch der Biologischen Arbeitsmethoden.“ 4 (1935),1289. |
(+)-Pilocarpine hydrochloride Consensus Reports
(+)-Pilocarpine hydrochloride Safety Profile
Poison by ingestion, intraperitoneal, and intravenous routes. Experimental teratogenic and reproductive effects. Human systemic effects: cardiac changes. When heated to decomposition it emits very toxic fumes of HCl and NOx. See also PILOCARPINE
Hazard Codes:T+
T+ :Very Toxic
Risk Statements
R26/28:Very Toxic by inhalation and if swallowed .
Safety Statements
S25:Avoid contact with eyes .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) .
RIDADR:UN 1544 6.1/PG 3
WGK Germany:3
RTECS:TK1450000
F:3-8-10
F3:Hygroscopic.
F8:Photosensitive.
F10:Keep under argon.
HazardClass:6.1
PackingGroup:III
(+)-Pilocarpine hydrochloride Specification
Incompatibilities with Other Materials Strong oxidizing agents.
Conditions to Avoid: Incompatible materials, light, exposure to moist air or water, heat
Hazardous Decomposition Products Hydrogen chloride, nitrogen oxides, carbon monoxide, carbon dioxide, oxides of chlorine.
Hazardous Polymerization Will not occur.
Related Products
- (+)-Pilocarpine hydrochloride
- 547-17-1
- 54717-17-8
- 5471-77-2
- 5471-81-8
- 5471-82-9
- 54719-15-2
- 5471-97-6
- 5472-01-5
- 5472-06-0
- 5472-13-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View