-
Name
1,1-Cyclobutanedicarboxylic acid
- EINECS 226-651-2
- CAS No. 5445-51-2
- Article Data11
- CAS DataBase
- Density 1.534 g/cm3
- Solubility Soluble in water, ethyl ether, chloroform and benzene
- Melting Point 158 °C(lit.)
- Formula C6H8O4
- Boiling Point 366.4 °C at 760 mmHg
- Molecular Weight 144.127
- Flash Point 189.6 °C
- Transport Information UN 3261 8/PG 2
- Appearance white fine crystalline powder
- Safety 26-36/37/39-45
- Risk Codes 34
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms 1,1-Dicarboxycyclobutane;1-Carboxycyclobutanecarboxylic acid;NSC 22073;Cyclobutane-1,1-dicarboxylic acid;
- PSA 74.60000
- LogP 0.32590
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride for 2h; Ambient temperature; | 23% |
-
-
28246-87-9
ethyl 1-cyano-1-cyclobutanecarboxylate

-
A
-
141-82-2
malonic acid

-
B
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| Hydrolysis; |

| Conditions | Yield |
|---|---|
| With potassium carbonate | |
| With oxonium | |
| With sodium hydroxide; water at 20℃; for 48h; Hydrolysis; |
-
-
30491-91-9
1-cyanocyclobutane-1-carboxylic acid

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
109-64-8
1,3-dibromo-propane

-
-
105-53-3
diethyl malonate

-
A
-
3721-95-7
Cyclobutanecarboxylic acid

-
B
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride for 1h; Ambient temperature; other subst. 1,3-dibromo compounds, other phase transfer catalysts; | |
| With sodium hydroxide; tetrabutyl-ammonium chloride for 1h; Ambient temperature; Yield given. Yields of byproduct given; |

-
-
300687-20-1
(3-iodo-propyl)-malonic acid diethyl ester

-
-
108-88-3
toluene

-
A
-
103-82-2
phenylacetic acid

-
B
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
60-29-7
diethyl ether

-
-
300687-20-1
(3-iodo-propyl)-malonic acid diethyl ester

-
A
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| Hydrolyse mit alkoh. KOH und Decarboxylierung; |


-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide pH=7; Inert atmosphere; Schlenk technique; | 100% |
| With potassium hydroxide In water | |
| With potassium hydroxide In water |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 5h; | 99.6% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 5h; | 99.6% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 5h; | 99.4% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 5h; | 99.3% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 5h; | 99.2% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 5h; | 99.2% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 5h; | 99.1% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 5h; | 99% |
-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
1308671-66-0
5,5-difluoro-10-mesityl-1,3,7,9-tetramethyl-5H-4λ4,5λ4-dipyrrolo[1,2-c:2',1'-f][1,3,2]diazaborinine

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In acetonitrile at 120℃; for 0.5h; Microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at -20 - 20℃; for 4h; Inert atmosphere; | 95% |
| In tetrahydrofuran | 92% |
| With lithium aluminium tetrahydride In tetrahydrofuran for 12h; Heating; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: cyclobutane-1,1'-dicarboxylic acid; zinc(II) oxide In water at 20℃; for 0.5h; Stage #2: ammonia In water | 95% |


| Conditions | Yield |
|---|---|
| In water at 5℃; for 20h; Temperature; Time; | 93.2% |
| In water at 30℃; for 0.15h; Temperature; Microwave irradiation; | 90% |
| In water at 20℃; |
-
-
14871-92-2
dichloro(2,2'-bipyridine)palladium(II)

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
214846-23-8
(2,2'-bipyridine-κ2N,N')(1,1-cyclobutanedicarboxylato-κ2O,O')palladium(II)

| Conditions | Yield |
|---|---|
| With silver nitrate In water byproducts: AgCl; stirring the Pd complex and AgNO3 in H2O at 60°C for 2, filtration, addn. of the org. acid, adjustment of pH to 5; vac. filtration, washing (H2O; EtOH; Et2O), drying (vac.); elem. anal.; | 93% |
-
-
75992-73-3, 25794-47-2, 30729-25-0, 15274-33-6, 22840-91-1, 14568-13-9
cis-dichlorobis(dimethylsulfoxide)platinum(II)

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
187224-67-5, 119759-73-8
{1,1-cyclobutanedicarboxylato(2-)-O,O'}bis{sulfinylbis{methane}-S-}platinum(II)

| Conditions | Yield |
|---|---|
| Stage #1: cyclobutane-1,1'-dicarboxylic acid With silver(l) oxide In water at 20℃; for 0.333333h; Darkness; Stage #2: cis-dichlorobis(dimethylsulfoxide)platinum(II) In water at 70℃; for 0.166667h; Darkness; Stage #3: In water at 20℃; for 24h; Darkness; | 90% |

| Conditions | Yield |
|---|---|
| With barium dihydroxide; silver sulfate In water byproducts: AgI, BaSO4; addn. of Ag2SO4 to slight excess of Pt-complex, stirring (4 h), filtration off of AgI, concn., addn. of soln. of ligand (neutralized with Ba(OH)2); filtration off of BaSO4, concn. (crystn.), washing (EtOH, Et2O), drying(vac.); elem. anal.; | 88% |
| Stage #1: cis-diaminediiodoplatinum(II) With silver sulfate In water for 4h; Stage #2: cyclobutane-1,1'-dicarboxylic acid With barium hydroxide octahydrate In water | 85% |
| Stage #1: cis-diaminediiodoplatinum(II) With silver nitrate In water Darkness; Stage #2: cyclobutane-1,1'-dicarboxylic acid With potassium hydroxide for 3h; pH=5; | 50.3% |
| With potassium hydroxide In water; N,N-dimethyl-formamide Pt-compd. dissolved in DMF with heating, addn. of the acid and aq. KOH, heated in an unstoppered flask at 60°C for 20 h; cooled, filtered, addn. of ether; | 40% |

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| In water (inert atmosphere); addn. of the Tl salt in hot H2O to the org. diacid in boiling H2O; pptn. by concn.; elem. anal.; | 86% |
-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

| Conditions | Yield |
|---|---|
| at 130℃; for 24h; Autoclave; Microwave irradiation; | 85% |

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
7646-79-9
cobalt(II) chloride

| Conditions | Yield |
|---|---|
| at 130℃; for 24h; Autoclave; | 85% |
| at 130℃; for 24h; Autoclave; Microwave irradiation; | 85% |

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: cyclobutane-1,1'-dicarboxylic acid With sodium hydroxide In water Stage #2: silver nitrate In water at 20℃; for 1h; Darkness; | 82% |
| With NaOH In water aq. soln. of NaOH added to dicarboxylic acid in water, AgNO3 added in dark, stirred for 15-30 min; ppt. filtered, washed with water, dried in air; | |
| With sodium hydroxide In water |
-
-
67-56-1
methanol

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
10224-72-3
dimethyl cyclobutane-1,1-dicarboxylate

| Conditions | Yield |
|---|---|
| With sulfuric acid In dichloromethane for 15h; Heating; | 81% |
-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: [Pd(1,4-bis(2-hydroxyethyl)piperazine)Cl2] With silver nitrate In water for 24h; Darkness; Stage #2: cyclobutane-1,1'-dicarboxylic acid With silver(I) chloride; sodium hydroxide In water at 60℃; for 2h; pH=5-6; | 81% |
-
-
15663-27-1
cisplatin

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; N,N-dimethyl-formamide Pt-compd. dissolved in DMF with heating, addn. of the acid and aq. KOH, heated in an unstoppered flask at 60°C for 20 h; cooled, filtered, concn. in vac.; | 80% |
-
-
15020-99-2
cis-dichloro(ethylenediamine)palladium(II)

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
214846-19-2
(1,2-ethylenediamine)(1,1-cyclobutanedicaboxylato)palladium(II) monohydrate

| Conditions | Yield |
|---|---|
| With sodium hydroxide; silver nitrate In water byproducts: AgCl; stirring the Pd complex and AgNO3 in H2O at 20°C in darkness overnight, filtration, addn. of the org. acid, adjustment of pH to 5 with NaOH; vac. filtration, washing (H2O; EtOH; Et2O), drying (vac.); elem. anal.; | 80% |
| With AgNO3; NaHCO3 In water byproducts: AgCl, CO2; AgNO3 was added to a suspn. of complex in water in the dark with stirring and heating at 60°C for 1 h, refrigerated, AlCl was filtered, dicarboxylic acid was added, NaHCO3 was added dropwise with heating and stirring (CO2), left at room temp.; solid was filtered, washed with water, air-dried; elem. anal.; | 78% |

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
7732-18-5
water

-
-
151840-51-6, 879282-32-3
{(C4H6(COO)2) copper(II)}*H2O

| Conditions | Yield |
|---|---|
| In water soln. of ligand in H2O added dropwise to warm aq. suspn. of Cu salt under stirring and heating below 70°C (water bath); heated for 2 h; filtered; filtrate evapd. for a few d; filtered; ppt. washed with EtOH and ether; air dried; elem. anal.; | 80% |

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
34867-87-3
spirocyclobutyl-malonyl peroxide

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; urea hydrogen peroxide adduct at 22℃; for 18h; | 80% |
| With methanesulfonic acid; urea hydrogen peroxide adduct at 20℃; for 18h; | 80% |
-
-
1202-34-2
di(pyridin-2-yl)amine

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
7732-18-5
water

-
-
7646-79-9
cobalt(II) chloride

| Conditions | Yield |
|---|---|
| With sodium carbonate for 2h; Thermodynamic data; Heating; | 80% |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In tetrahydrofuran; benzene at 80℃; for 4h; Dean-Stark; | 80% |

-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

| Conditions | Yield |
|---|---|
| In formamide High Pressure; Tb(NO3)3*6H2O, cyclobutane-1,1'-dicarboxylic acid dissolved in formamideplaced in autoclave, heated at 130°C for 24 h, slowly cooled ove rnight; collected, washed with EtOH, petroleum ether, dried under N2 at room temp.; elem. anal.; | 77% |


-
-
5445-51-2
cyclobutane-1,1'-dicarboxylic acid

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

| Conditions | Yield |
|---|---|
| In formamide High Pressure; Gd(NO3)3*6H2O, cyclobutane-1,1'-dicarboxylic acid dissolved in formamideplaced in autoclave, heated at 130°C for 24 h, slowly cooled ove rnight; collected, washed with EtOH, petroleum ether, dried under N2 at room temp.; elem. anal.; | 77% |

1,1-Cyclobutanedicarboxylic acid Specification
The 1,1-Cyclobutanedicarboxylic acid, with the CAS registry number 5445-51-2, is also known as 1-Carboxycyclobutanecarboxylic acid. It belongs to the product categories of Carboxylic Acids; Organic acids; Cyclobutanes & Cyclobutenes; Simple 4-Membered Ring Compounds; Carboxylic Acids; Ring Systems; Cycloalkanes; C6; Carbonyl Compounds. Its EINECS number is 226-651-2. This chemical's molecular formula is C6H8O4 and molecular weight is 144.13. What's more, its systematic name is cyclobutane-1,1-dicarboxylic acid. It is stable at common pressure and temperature, and it should be sealed and stored in a cool and dry place. It is used as drug intermediates.
Physical properties of 1,1-Cyclobutanedicarboxylic acid are: (1)ACD/LogP: -0.24; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -2.6; (4)ACD/LogD (pH 7.4): -4.19; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 4; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 52.6 Å2; (13)Index of Refraction: 1.564; (14)Molar Refractivity: 30.56 cm3; (15)Molar Volume: 93.9 cm3; (16)Polarizability: 12.11×10-24cm3; (17)Surface Tension: 80.5 dyne/cm; (18)Density: 1.534 g/cm3; (19)Flash Point: 189.6 °C; (20)Enthalpy of Vaporization: 67.31 kJ/mol; (21)Boiling Point: 366.4 °C at 760 mmHg; (22)Vapour Pressure: 2.27E-06 mmHg at 25°C.
Preparation: this chemical can be prepared by 1-bromo-3-chloropropane and diethyl malonate with the existence of sodium ethoxide, then diethyl cyclobutane-1,1-dicarboxylate is produced, and finally the product is made through hydrolysis.
Uses of 1,1-Cyclobutanedicarboxylic acid: it can be used to produce cyclobutane-1,1-diyl-bis-methanol by heating. It will need reagent LiAlH4 and solvent tetrahydrofuran with the reaction time of 12 hours. The yield is about 88%.
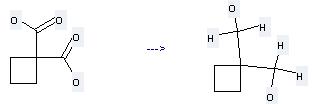
When you are using this chemical, please be cautious about it as the following:
This chemical can cause burns. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection. In case of accident or if you feel unwell, you must seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)C1(C(=O)O)CCC1
(2)Std. InChI: InChI=1S/C6H8O4/c7-4(8)6(5(9)10)2-1-3-6/h1-3H2,(H,7,8)(H,9,10)
(3)Std. InChIKey: CCQPAEQGAVNNIA-UHFFFAOYSA-N
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 5445-63-6
- 5445-67-0
- 544-57-0
- 54458-61-6
- 5446-02-6
- 54460-46-7
- 544-60-5
- 5446-10-6
- 5446-16-2
- 5446-18-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View