-
Name
2,4,6-trinitrobenzene-1,3,5-triamine
- EINECS
- CAS No. 3058-38-6
- Article Data33
- CAS DataBase
- Density 1.958g/cm3
- Solubility
- Melting Point 324 °C (decomp)
- Formula C6H6 N6 O6
- Boiling Point 669.5°Cat760mmHg
- Molecular Weight 258.15
- Flash Point 358.7°C
- Transport Information
- Appearance
- Safety Low toxicity by ingestion and inhalation. A mild eye irritant. When heated to decomposition it emits toxic vapors of NOx.
- Risk Codes
-
Molecular Structure
- Hazard Symbols Low toxicity by ingestion and inhalation. A mild eye irritant.
- Synonyms 1,3,5-Triamino-2,4,6-trinitrobenzene;2,4,6-Trinitro-1,3,5-benzenetriamine; EDC 35; NSC 243156; TATB;s-Triaminotrinitrobenzene; sym-Triaminotrinitrobenzene
- PSA 215.52000
- LogP 3.47100
Synthetic route

| Conditions | Yield |
|---|---|
| With 4-amino-1,2,4-triazole; sodium methylate In methanol at 25 - 60℃; for 21.5h; Product distribution / selectivity; | 99.8% |
| Stage #1: picramide With 4-amino-1,2,4-triazole; sodium methylate In methanol; toluene at 25 - 60℃; for 16.5h; Stage #2: With acetic acid In methanol; toluene Product distribution / selectivity; | 98% |
| With 4-amino-1,2,4-triazole; sodium methylate In dimethyl sulfoxide at 20℃; for 3h; | 94.1% |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water; dimethyl sulfoxide at 80℃; for 6h; pH=1; | 99.6% |
-
-
68959-41-1
1,3,5-triethoxy-2,4,6-trinitrobenzene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With ammonia In toluene at 90℃; under 3863.02 Torr; for 4h; Product distribution / selectivity; | 99.5% |
| With methanol; ammonia at -3 - 50℃; for 24h; Product distribution / selectivity; Sealed reactor; Industry scale; | 89.5% |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 65℃; for 8h; pH=13.5; | 99.5% |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 65℃; for 5h; pH=14; | 99.4% |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 60℃; for 5h; pH=14; | 99.4% |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 60℃; for 6h; pH=14; | 99.2% |

| Conditions | Yield |
|---|---|
| With ammonia In toluene for 10h; Heating; | 99% |
| With ammonia | 90% |
| With ammonia In toluene for 24h; Reflux; | 87% |
-
-
21985-87-5
2,3,4,5,6-pentanitroaniline 1,2-dichloroethane

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With ammonia In tetrachloromethane; toluene for 0.583333h; | 97% |
| With ammonia |

| Conditions | Yield |
|---|---|
| With sodium methylate In dimethyl sulfoxide at 20℃; for 3h; | 89.5% |
-
-
489-98-5
picramide

-
-
5470-11-1
hydroxylamine hydrochloride

-
A
-
439614-66-1
1,3,5-triamino-2,4-dinitro-6-nitrososbenzene

-
B
-
1630-08-6
1,3,5-trinitrobenzenediamine

-
C
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Stage #1: picramide; hydroxylamine hydrochloride With sodium methylate In methanol; dimethyl sulfoxide; toluene at 25 - 90℃; for 2.33333h; Stage #2: With acetic acid In dimethyl sulfoxide; toluene Product distribution / selectivity; | A n/a B n/a C 85% |

-
-
56140-58-0
1,3-diamino-5-hydroxytrinitrobenzene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With diammonium phosphate In sulfolane at 20 - 177℃; for 8h; | 80% |
-
-
172265-85-9
2,4,6-trinitro-1,3,5-triacetylaminobenzene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 100℃; for 0.5h; Temperature; Reagent/catalyst; | 77.5% |
| With ammonia In water; N,N-dimethyl-formamide at 20 - 130℃; under 8791.75 Torr; for 0.666667 - 2.5h; Product distribution / selectivity; | 68% |
| With ammonia In water at 123℃; for 1h; | 58% |
-
-
83430-12-0
1,3,5-tribromo-2,4,6-trinitrobenzene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With ammonia In toluene | 72% |

-
-
88106-06-3
N,N'-Diacetyl-2,4-diamino-1,3,5-trinitrobenzene

-
-
172265-85-9
2,4,6-trinitro-1,3,5-triacetylaminobenzene

-
A
-
439614-66-1
1,3,5-triamino-2,4-dinitro-6-nitrososbenzene

-
B
-
1630-08-6
1,3,5-trinitrobenzenediamine

-
C
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With ammonia In water at 123℃; for 1h; | A n/a B n/a C 62% |

-
-
26243-62-9
N,N',N”-triacetyl-1,3,5-triaminobenzene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Stage #1: N,N',N”-triacetyl-1,3,5-triaminobenzene With nitric acid; acetic anhydride at -5℃; for 8h; Stage #2: With nitric acid; acetic anhydride In water at 20 - 105℃; for 17h; Reagent/catalyst; Temperature; Solvent; | 56% |
| Multi-step reaction with 2 steps 1: nitric acid; sulfuric acid / 0.25 h / -5 - 25 °C 2: hydrogenchloride / water / 0.5 h / 100 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: nitric acid; sulfuric acid / 4 h / 50 °C / Cooling with ice 2.1: water / 6 h / 95 °C / Cooling with ice 3.1: acetic anhydride / 2 h / Cooling with ice 3.2: 8 h / 40 °C View Scheme |
-
-
1423-11-6
1,3,5-trifluoro-2,4,6-trinitrobenzene

-
A
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

-
B
-
73333-89-8
1-amino-3,5-difluorotrinitrobenzene

-
C
-
78925-50-5
3,5-diamino-1-fluoro-2,4,6-trinitrobenzene

| Conditions | Yield |
|---|---|
| With ammonia In dichloromethane at -73℃; for 1h; Yields of byproduct given; | A 0.22 g B n/a C n/a |
| With ammonia In dichloromethane at -73℃; for 1h; Yield given. Yields of byproduct given; |

-
-
1423-11-6
1,3,5-trifluoro-2,4,6-trinitrobenzene

-
A
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

-
B
-
78925-50-5
3,5-diamino-1-fluoro-2,4,6-trinitrobenzene

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate; tert-butylamine; trifluoroacetic acid 1.) CH2Cl2, -30 deg C, 2.) room temperature, 15h, 3.) 20 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With ethanol; ammonia in der Siedehitze; |
-
-
83430-12-0
1,3,5-tribromo-2,4,6-trinitrobenzene

-
-
7664-41-7
ammonia

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene
-
-
21985-87-5
2,3,4,5,6-pentanitroaniline 1,2-dichloroethane

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene
-
-
35572-78-2
2-amino-4,6-dinitrotoluene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 80percent aq. H2SO4, 90percent aq. nitric acid / 24 h / Ambient temperature 2: 78 percent / 96percent aq. H2SO4, anisole / 0.5 h / Ambient temperature 3: 40 percent / 90percent HNO3, 100percent H2SO4 / 1.) RT, 60 min, 2.) 75 deg C, 90 min 4: 97 percent / anh. ammonia / toluene; CCl4 / 0.58 h View Scheme |
-
-
19406-51-0
4-Amino-2,6-dinitrotoluene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 78 percent / 96percent H2SO4, 90percent HNO3 / acetic acid / 9 h / 0 °C 2: 35 percent / 96percent aq. H2SO4 / 60 h / 0 °C 3: 40 percent / 90percent HNO3, 100percent H2SO4 / 1.) RT, 60 min, 2.) 75 deg C, 90 min 4: 97 percent / anh. ammonia / toluene; CCl4 / 0.58 h View Scheme | |
| Multi-step reaction with 4 steps 1: 100 percent / 96percent aq. H2SO4, 90percent aq. HNO3 / 16 h / 0 - 5 °C 2: 78 percent / 96percent aq. H2SO4, anisole / 0.5 h / Ambient temperature 3: 40 percent / 90percent HNO3, 100percent H2SO4 / 1.) RT, 60 min, 2.) 75 deg C, 90 min 4: 97 percent / anh. ammonia / toluene; CCl4 / 0.58 h View Scheme |
-
-
84432-53-1
4-Amino-2,3,5,6-tetranitrotoluene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 40 percent / 90percent HNO3, 100percent H2SO4 / 1.) RT, 60 min, 2.) 75 deg C, 90 min 2: 97 percent / anh. ammonia / toluene; CCl4 / 0.58 h View Scheme |
-
-
84432-52-0
4-amino-N,2,3,6-tetranitrotoluene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 35 percent / 96percent aq. H2SO4 / 60 h / 0 °C 2: 40 percent / 90percent HNO3, 100percent H2SO4 / 1.) RT, 60 min, 2.) 75 deg C, 90 min 3: 97 percent / anh. ammonia / toluene; CCl4 / 0.58 h View Scheme |
-
-
84432-54-2
4-amino-N,2,3,5,6-pentanitrotoluene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 78 percent / 96percent aq. H2SO4, anisole / 0.5 h / Ambient temperature 2: 40 percent / 90percent HNO3, 100percent H2SO4 / 1.) RT, 60 min, 2.) 75 deg C, 90 min 3: 97 percent / anh. ammonia / toluene; CCl4 / 0.58 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sulfuric acid <100 percent >; nitric acid / 75 °C 2: ammonia View Scheme |
-
-
18523-16-5
1,3,5-Trimethoxy-2,4,6-trinitrobenzene

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With ammonia In diethyl ether | |
| With ammonia In ethyl acetate under 760.051 Torr; for 6h; |

| Conditions | Yield |
|---|---|
| With ammonia; sodium In methanol Heating; | 95% |
| With hydrogen; palladium on activated charcoal In ethyl acetate | 80% |
| With phenylhydrazine 1.) 120 degC, 2.5 h; 2.) to 140 degC in 2.5 h; 3.) 140 degC, 1h; | 68% |
-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Stage #1: 2,4,6-triamino-1,3,5-trinitrobenzene With palladium 10% on activated carbon; hydrogen In ethyl acetate under 3150.32 Torr; for 72h; Stage #2: With hydrogenchloride | 93.2% |
| Stage #1: 2,4,6-triamino-1,3,5-trinitrobenzene With palladium 10% on activated carbon; hydrogen In ethyl acetate under 3150.32 Torr; for 72h; Stage #2: With hydrogenchloride; palladium 10% on activated carbon; hydrogen In water; ethyl acetate under 3150.32 Torr; for 5h; | 92% |
| Stage #1: 2,4,6-triamino-1,3,5-trinitrobenzene With palladium 10% on activated carbon; hydrogen In ethyl acetate under 3150.32 Torr; for 72h; Stage #2: With hydrogenchloride; hydrogen In water; ethyl acetate for 5h; | 92% |
| With hydrogenchloride; tin(II) chloride dihdyrate In ethyl acetate for 5h; Reflux; | 75% |
-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

-
-
108-24-7
acetic anhydride

-
-
172265-85-9
2,4,6-trinitro-1,3,5-triacetylaminobenzene

| Conditions | Yield |
|---|---|
| sulfuric acid at 20 - 98℃; for 5h; Product distribution / selectivity; | 93% |
| With sulfuric acid at 115 - 120℃; for 28h; | 85.3% |
-
-
67-56-1
methanol

-
-
3153-26-2
bis(acetylacetonate)oxovanadium

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| at 160℃; for 48h; Autoclave; | 80% |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

-
-
135285-90-4
2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurzitane

| Conditions | Yield |
|---|---|
| In water for 1.5h; | 75.5% |
-
-
67-56-1
methanol

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| at 160℃; for 48h; Autoclave; | 65% |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin(ll) chloride In water at 20℃; for 48h; Inert atmosphere; Schlenk technique; | 64% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin |

| Conditions | Yield |
|---|---|
| at 140 - 200℃; | |
| at 140℃; dann bei 200grad; |
-
-
4443-63-4
icosane-10,11-dione

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

-
A
-
120829-14-3
5-Amino-6-nitro-2,3,8,9-tetranonylpyrazino<2,3-f>quinoxaline

-
B
-
96355-62-3
2,3,6,7,10,11-hexanonyldipyrazino<2,3-f;2',3'-h>quinoxaline

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal 1.) ethyl acetate, 2.) ethanol, glacial acetic acid, H2O, 50 deg C, 1.5 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
3058-38-6
2,4,6-triamino-1,3,5-trinitrobenzene

-
A
-
4444-26-2
benzenehexamine

-
B
-
120829-15-4
nitrobenzenepentamine

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethyl acetate |
1,3,5-Triamino-2,4,6-trinitrobenzene Chemical Properties
Empirical Formula of 1,3,5-Triamino-2,4,6-trinitrobenzene (CAS NO.3058-38-6): C6H6N6O6
Molecular Weight: 258.15 g/mol
EINECS: 221-297-5
Index of Refraction: 1.844
Density: 1.958 g/cm3
Flash Point: 358.7 °C
Enthalpy of Vaporization: 98.39 kJ/mol
Boiling Point: 669.5 °C at 760 mmHg
Vapour Pressure: 8.67E-18 mmHg at 25 °C
Structure of 1,3,5-Triamino-2,4,6-trinitrobenzene (CAS NO.3058-38-6):
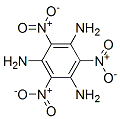
IUPAC Name of 1,3,5-Triamino-2,4,6-trinitrobenzene (CAS NO.3058-38-6): 2,4,6-Trinitrobenzene-1,3,5-triamine
1,3,5-Triamino-2,4,6-trinitrobenzene Toxicity Data With Reference
| 1. | eye-rbt 100 mg MLD | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) OTS0537049 . | ||
| 2. | orl-rat LD :>5 g/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) OTS0537049 . | ||
| 3. | ihl-rat LC :>212 mg/m3/1H | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) OTS0537049 . | ||
| 4. | orl-mus LD :>5 g/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) UCRL-13701 . |
1,3,5-Triamino-2,4,6-trinitrobenzene Consensus Reports
Reported in EPA TSCA Inventory.
1,3,5-Triamino-2,4,6-trinitrobenzene Safety Profile
Low toxicity by ingestion and inhalation. A mild eye irritant. When heated to decomposition it emits toxic vapors of NOx.
1,3,5-Triamino-2,4,6-trinitrobenzene Specification
1,3,5-Triamino-2,4,6-trinitrobenzene ,its cas register number is 3058-38-6. It also can be called 1,3,5-Benzenetriamine, 2,4,6-trinitro- .
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 3058-39-7
- 305-84-0
- 3058-47-7
- 305-85-1
- 305851-04-1
- 305858-63-3
- 30586-13-1
- 30586-93-7
- 3058-98-8
- 3059-67-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View