-
Name
1,3-Benzenedithiol
- EINECS 210-925-3
- CAS No. 626-04-0
- Article Data14
- CAS DataBase
- Density 1.24 g/cm3
- Solubility
- Melting Point 24-25 °C
- Formula C6H6S2
- Boiling Point 244.3 °C at 760 mmHg
- Molecular Weight 142.246
- Flash Point 112.7 °C
- Transport Information
- Appearance colorless liquid or white solid
- Safety 26-39
- Risk Codes 37/38-41
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms m-Benzenedithiol(6CI,7CI,8CI);1,3-Dimercaptobenzene;Dithioresorcinol;Thioresorcinol;
- PSA 77.60000
- LogP 2.26400
Synthetic route
-
-
50667-85-1
1,3-Bis-(dimethylcarbamoylthio)-benzol

-
-
626-04-0
3-mercaptothiophenol

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethylene glycol for 1h; Heating; | 98.5% |
| With potassium hydroxide In methanol Heating; |

| Conditions | Yield |
|---|---|
| Reaktion ueber mehrere Stufen; |
-
-
861601-41-4
1,3-bis-trichloromethylsulfanyl-benzene

-
-
62-53-3
aniline

-
A
-
626-04-0
3-mercaptothiophenol

-
B
-
101-01-9
N,N',N''-triphenylguanidine


| Conditions | Yield |
|---|---|
| With sodium isopropanethiolate; sodium 1.) HMPA, 2.) 100 deg C; Yield given. Multistep reaction; |

-
-
626-04-0
3-mercaptothiophenol

-
-
4101-68-2
1,10-dibromodecane

-
-
75703-91-2
2,4-Benzo-1,5-dithiacyclopentadec-2-ene

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 45 - 50℃; | 95% |
| With caesium carbonate In N,N-dimethyl-formamide at 50℃; | 95% |
-
-
626-04-0
3-mercaptothiophenol

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; N,N-dimethyl-formamide stirring under N2 atmosphere for 16 h at room temperature; filtn. through a sintered glass crucible into a HCl soln., evapn. (reduced pressure), addn. of aq. NH4PF6, filtn., drying (vac.), washing (ether), drying; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-mercaptothiophenol With sodium hydride In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; Schlenk technique; Glovebox; Stage #2: chloro-diphenylphosphine In tetrahydrofuran Reflux; Inert atmosphere; Schlenk technique; Glovebox; | 95% |

| Conditions | Yield |
|---|---|
| In methanol exclusion of light; addn. of soln. of dithiol in abs. methanol to a suspension of Pb compd. in abs. methanol (preparing suspension with ice cooling); sepn. of a yellow compd.;; filtration of ppt. after 1h; washing with methanol or acetone; drying in vac.; elem. anal.;; | 93% |
| In acetone exclusion of light; addn. of soln. of dithiol in acetone to a suspension of Pb compd. in acetone (preparing suspension with ice cooling); sepn. of a yellow compd.;; filtration of ppt. after 1h; washing with methanol or acetone; drying in vac.; elem. anal.;; | 93% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-mercaptothiophenol With sodium hydride In tetrahydrofuran at 20℃; for 1h; Glovebox; Schlenk technique; Inert atmosphere; Stage #2: Chlorodiisopropylphosphane In tetrahydrofuran at 20℃; for 1h; Glovebox; Schlenk technique; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol for 3h; Heating; | 92% |
| With potassium carbonate In acetone at 50℃; | 88% |
-
-
13755-29-8
sodium tetrafluoroborate

-
-
626-04-0
3-mercaptothiophenol

-
-
53317-87-6
tris[triphenylphosphinegold(I)]oxonium tetrafluoroborate

-
-
448280-28-2
phenylene-1-bis[(triphenylphosphine)gold(I)]sulfonio-3-[(triphenylphosphine)gold(I)]thiolate tetrafluoroborate

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: H2O; a soln. of complex in CH2Cl2 was treated with a soln. of dithiol in CH2Cl2 and a small quantity of NaBF4 at 20°C for 1 h (N2); ppt. was filtered off, crystd. upon addn. of pentane and cooling to -20°C, crystals were dried in vac.; elem. anal.; | 92% |

-
-
626-04-0
3-mercaptothiophenol

-
-
107-13-1
acrylonitrile

-
-
16079-08-6
3,3'-(m-phenylenedithio)dipropionitrile

| Conditions | Yield |
|---|---|
| With N-benzyl-trimethylammonium hydroxide In tetrahydrofuran; methanol at -78 - 20℃; Inert atmosphere; | 90% |
| With trimethylamine |
-
-
626-04-0
3-mercaptothiophenol

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl acetamide at 100℃; | 90% |
-
-
626-04-0
3-mercaptothiophenol

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; N,N-dimethyl-formamide stirring under N2 atmosphere for 16 h at room temperature; filtn. through a sintered glass crucible into a HCl soln., evapn. (reduced pressure), addn. of aq. NH4PF6, filtn., drying (vac.), washing (ether), drying; | 90% |
-
-
626-04-0
3-mercaptothiophenol

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; N,N-dimethyl-formamide stirring under N2 atmosphere for 16 h at room temperature; filtn. through a sintered glass crucible into a HCl soln., evapn. (reduced pressure), addn. of aq. NH4PF6, filtn., drying (vac.), washing (ether), drying; | 90% |
1,3-Benzenedithiol Specification
The 1,3-Benzenedithiol, with the CAS registry number 626-04-0, is also known as 1,3-Dimercaptobenzene. Its EINECS number is 210-925-3. This chemical's molecular formula is C6H6S2 and molecular weight is 142.24. What's more, its IUPAC name is benzene-1,2-dithiol. The product should be sealed and stored in containers with dry inert gas. What's more, it should be protected from oxidants and air.
Physical properties of 1,3-Benzenedithiol are: (1)ACD/LogP: 2.92; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.76; (4)ACD/LogD (pH 7.4): 1.14 ; (5)#Freely Rotating Bonds: 2; (6)Polar Surface Area: 77.6 Å2; (7)Index of Refraction: 1.665; (8)Molar Refractivity: 42.6 cm3; (9)Molar Volume: 114.6 cm3; (10)Polarizability: 16.88×10-24cm3; (11)Surface Tension: 49.6 dyne/cm; (12)Density: 1.24 g/cm3; (13)Flash Point: 112.7 °C; (14)Enthalpy of Vaporization: 46.19 kJ/mol; (15)Boiling Point: 244.3 °C at 760 mmHg; (16)Vapour Pressure: 0.0477 mmHg at 25°C.
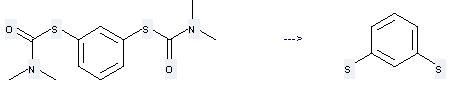
Preparation of 1,3-Benzenedithiol: this chemical can be prepared by 1,3-Bis-(dimethylcarbamoylthio)-benzol by heating. This reaction will need reagent aq. KOH and solvent ethane-1,2-diol with the reaction time of 1 hour. The yield is about 98.5%.
Uses of 1,3-Benzenedithiol: it can be used to produce 1,3-bis(-styrylthio)-benzol by heating. It will need reagent sodium and solvent methanol with the reaction time of 20 hours. The yield is about 82%.
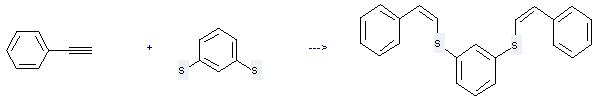
When you are using this chemical, please be cautious about it as the following:
It is irritating to respiratory system and skin. It has a limited evidence of a carcinogenic effect. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear eye/face protection.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: C1=CC=C(C(=C1)S)S
(2)InChI: InChI=1S/C6H6S2/c7-5-3-1-2-4-6(5)8/h1-4,7-8H
(3)InChIKey: JRNVQLOKVMWBFR-UHFFFAOYSA-N
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 626-05-1
- 62605-98-5
- 626-06-2
- 62607-26-5
- 626-07-3
- 62607-69-6
- 62608-15-5
- 626-08-4
- 62609-86-3
- 62610-39-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View