-
Name
1-Amino-8-hydroxynaphthalene-3,6-disulphonic acid
- EINECS 201-975-7
- CAS No. 90-20-0
- Article Data15
- CAS DataBase
- Density 1.88 g/cm3
- Solubility
- Melting Point >300 °C(lit.)
- Formula C10H9NO7S2
- Boiling Point
- Molecular Weight 319.316
- Flash Point
- Transport Information
- Appearance White to gray-yellow powder
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 1-Naphthol-3,6-disulfonicacid, 8-amino- (3CI);1-Amino-3,6-disulfo-8-naphthol;1-Hydroxy-8-amino-3,6-naphthalenedisulfonic acid;1-Naphthol-8-amino-3,6-disulfonicacid;3,6-Disulfo-8-hydroxy-1-naphthylamine;4-Amino-5-hydroxy-2,7-naphthalenedisulfonic acid;
- PSA 171.75000
- LogP 3.36380
Synthetic route

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; water at 90℃; for 5h; | 65.86% |
-
-
117-42-0
8-amino-naphthalene-1,3,6-trisulfonic acid

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; water at 180 - 190℃; im offenen Gefaess; | |
| With sodium hydroxide at 210℃; im geschlossenen Gefaess; | |
| durch Alkalischmelze; | |
| Stage #1: 8-amino-naphthalene-1,3,6-trisulfonic acid With sodium hydroxide under 6750.68 Torr; for 6h; Stage #2: With sulfuric acid In water for 2h; pH=2; |
-
-
6362-11-4
1,8-diamino-3,6-naphthalenedisulfonic acid

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| at 200 - 210℃; |
-
-
6362-11-4
1,8-diamino-3,6-naphthalenedisulfonic acid

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid at 110 - 120℃; | |
| With diluted alkali at 200 - 210℃; Kochen der erhaltenen Produkte nut verd. Schwefelsaeure oder Salzsaeure; |
-
-
7664-93-9
sulfuric acid

-
-
6362-23-8
1H-naphtho[1,8-de][1,2,3]triazine-5,8-disulfonic acid

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| at 170℃; |
-
-
7732-18-5
water

-
-
408511-00-2
1,1-dioxo-2H-1λ6-naphth[1,8-cd]isothiazole-4,7-disulfonic acid

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| at 180℃; beim Schmelzen; |

-
-
784103-83-9
4-amino-5-chloro-naphthalene-2,7-disulfonic acid

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| beim Verschmelzen; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: durch Nitrierung und Reduktion der entstandenen Dinitroverbindung 2: diluted alkali / 200 - 210 °C / Kochen der erhaltenen Produkte nut verd. Schwefelsaeure oder Salzsaeure View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: sulfuric acid / 100 - 165 °C 1.2: 10.75 h / 30 - 165 °C 2.1: nitric acid / 10 °C 3.1: iron / 60 °C 3.2: 80 - 90 °C 4.1: sodium hydroxide / 6 h / 6750.68 Torr 4.2: 2 h / pH 2 View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: nitric acid / 10 °C 2.1: iron / 60 °C 2.2: 80 - 90 °C 3.1: sodium hydroxide / 6 h / 6750.68 Torr 3.2: 2 h / pH 2 View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: 1-aminonaphthalene-3,6,8-trisulphonic acid trisodium salt With sodium hydroxide In methanol at 180 - 200℃; under 21002.1 Torr; Stage #2: With sulfuric acid In methanol |

-
-
91-20-3
naphthalene

-
A
-
148-25-4
chromatotropic acid

-
B
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: sulfur trioxide; sulfuric acid / 65 - 150 °C 1.2: 40 °C 2.1: hydrogen / 170 °C / 150015 Torr 3.1: sodium hydroxide / methanol / 180 - 200 °C / 21002.1 Torr View Scheme |

-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
2944-29-8
1,4-bis((4-aminophenyl)amino)anthrancene-9,10-dione

-
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichloro-2,4,6-triazine; 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid With sodium carbonate In water at 0 - 5℃; for 1h; pH=4 - 5; Stage #2: aniline In water at 25 - 30℃; for 3h; pH=5 - 6; Stage #3: 1,4-bis((4-aminophenyl)amino)anthrancene-9,10-dione Further stages; | 91.3% |


| Conditions | Yield |
|---|---|
| Stage #1: C12H16N2O6S2 With hydrogenchloride; sodium nitrite In water Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid With sodium hydrogencarbonate In water for 3h; pH=3.5; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichloro-2,4,6-triazine; 1-amino-2-sulfonic acid-4-(3-amino-2,4,6-trimethyl-5-sulfoamidophenyl)anthraquinone monosodium salt In water at 0 - 5℃; for 4h; pH=4- 6.5; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid In water at 28 - 35℃; pH=3.5 - 4; Stage #3: sodium 7-aminonaphthalene-1,3,6-trisulphonate Further stages; | 86.4% |


| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichloro-2,4,6-triazine; 1-amino-2-sulfonic acid-4-(3-amino-2,4,6-trimethyl-5-sulfoamidophenyl)anthraquinone monosodium salt In water at 0 - 5℃; for 4h; pH=4- 6.5; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid In water at 28 - 35℃; pH=3.5 - 4; Stage #3: sodium 3‐aminonaphthalene‐1,5‐disulfonate Further stages; | 86.4% |

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
246253-72-5
10-[(4'-bromomethyl)benzil]phenothiazine

-
-
246253-74-7
4-hydroxy-5-(4-phenothiazin-10-ylmethyl-benzylamino)-naphthalene-2,7-disulfonic acid

| Conditions | Yield |
|---|---|
| With sodium acetate In water at 5 - 25℃; for 8h; Condensation; | 85% |
-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
88-21-1
2-amino-1-benzenesulfonic acid

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichloro-2,4,6-triazine; 1-amino-2-sulfonic acid-4-(3-amino-2,4,6-trimethyl-5-sulfoamidophenyl)anthraquinone monosodium salt In water at 0 - 5℃; for 4h; pH=4- 6.5; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid In water at 28 - 35℃; pH=3.5 - 4; Stage #3: 2-amino-1-benzenesulfonic acid Further stages; | 84.23% |

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| Stage #1: bis(2-hydroxyethyl) 2-aminoterephthalate With hydrogenchloride; cis-nitrous acid In water at 5℃; for 5h; pH=8.5; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid With sodium carbonate for 1h; pH=8.5; | 83.3% |
-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid


| Conditions | Yield |
|---|---|
| With sodium carbonate; sodium chloride In water 1.) 0-5 deg C, 3 h; 2.) 60 deg C, 1 h; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-aminobenzoyl fluoride With hydrogenchloride; sodium nitrite In water at 0 - 5℃; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid With ammonium chloride In water at 0 - 5℃; for 0.666667h; pH=5.5 - 6.5; | 81% |
-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
10312-55-7
3-aminoterephthalic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-aminoterephthalic acid With hydrogenchloride; cis-nitrous acid In water at 10℃; for 5h; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid With sodium carbonate pH=7.5; | 80.2% |
-
-
2494-89-5
2-(p-aminophenylsulfonyl)ethyl hydrogen sulfate

-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
88-21-1
2-amino-1-benzenesulfonic acid

-
-
87-02-5
1-hydroxy-6-amino-3-naphthalenesulfonic acid

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
88-63-1
2,4-Diaminobenzenesulfonic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichloro-2,4,6-triazine; 7-amino-4-hydroxy-2-naphthalenesulfonic acid With sodium hydroxide In water at 0 - 5℃; pH=5.6 - 6.5; Stage #2: With sodium carbonate In water at 3 - 5℃; for 5h; Stage #3: 2-(p-aminophenylsulfonyl)ethyl hydrogen sulfate; 2-amino-1-benzenesulfonic acid; 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid; 2,4-Diaminobenzenesulfonic acid Further stages; | 77.1% |

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
6860-97-5
N-(p-tosyl)-H-acid

| Conditions | Yield |
|---|---|
| With sodium acetate; sodium hydroxide In water at 70℃; pH=3 - 6; Darkness; | 77% |
-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
26878-11-5
1-(4'-sulfophenyl)-3-methylpyrazol-5-one

-
-
88-64-2
2-amino-4-acetylaminobenzene sulfonic acid

-
-
24605-36-5
2-amino-1,4-benzenedisulfonic acid monosodium Salt

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichloro-2,4,6-triazine; 2-amino-1,4-benzenedisulfonic acid monosodium Salt In water at 0℃; for 3h; pH=6; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid In water at 35℃; for 4h; pH=4 - 5; Stage #3: 1-(4'-sulfophenyl)-3-methylpyrazol-5-one; 2-amino-4-acetylaminobenzene sulfonic acid Further stages; | 58.7% |

-
-
675-14-9
trifluoro-[1,3,5]triazine

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
26878-11-5
1-(4'-sulfophenyl)-3-methylpyrazol-5-one

-
-
88-64-2
2-amino-4-acetylaminobenzene sulfonic acid

-
-
24605-36-5
2-amino-1,4-benzenedisulfonic acid monosodium Salt

| Conditions | Yield |
|---|---|
| Stage #1: trifluoro-[1,3,5]triazine; 2-amino-1,4-benzenedisulfonic acid monosodium Salt In water at 0 - 5℃; pH=3 - 4; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid In water at 25℃; pH=4 - 5; Stage #3: 1-(4'-sulfophenyl)-3-methylpyrazol-5-one; 2-amino-4-acetylaminobenzene sulfonic acid Further stages; | 52.1% |


-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid In water for 0.5h; | 49% |
-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| pH 5; | 44% |
-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4,4'-Diaminostilbene dihydrochloride diazotization; Stage #2: 4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid | 38.5% |
-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| pH 9; | 38% |
-
-
75-44-5
phosgene

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
97629-54-4
2-oxo-2,3-dihydro-naphth[1,8-de][1,3]oxazine-5,8-disulfonic acid

| Conditions | Yield |
|---|---|
| With alkali |
-
-
38707-70-9
8-formylquinoline

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| With ethanol; water |
-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
7538-88-7
4-(4,6-dichloro-[1,3,5]triazin-2-ylamino)-5-hydroxy-naphthalene-2,7-disulfonic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide | |
| With sodium hydroxide In water at 0 - 5℃; for 1.5h; | |
| With sodium hydroxide; disodium hydrogenphosphate In water at 0℃; for 2h; pH=3; |
-
-
53855-64-4
3-acetoxy-2-naphthoyl chloride

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| With alkali Verseifung der entstandenen Acetylverbindung mit verd. Natronlauge; |
-
-
17356-93-3
2-methoxy-benzenediazonium

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
57573-52-1
4-methylbenzene diazonium

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
84077-76-9
5-amino-4-hydroxy-3-p-tolylazo-naphthalene-2,7-disulfonic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
17333-86-7
o-Carboxybenzenediazonium

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

-
-
36429-86-4
2-(8-amino-1-hydroxy-3,6-disulfo-[2]naphthylazo)-benzoic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
24892-65-7
diazotiertes o-Phenetidin

-
-
90-20-0
4-amino-5-hydroxy-2,7-naphthalenedisulfonic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate |
1-Amino-8-hydroxynaphthalene-3,6-disulphonic acid Consensus Reports
Reported in EPA TSCA Inventory.
1-Amino-8-hydroxynaphthalene-3,6-disulphonic acid Specification
The IUPAC name of this chemical is 4-Amino-5-hydroxy-2,7-naphthalene disulfonic acid. With the CAS registry number 90-20-0 and EINECS registry number 201-975-7, it is also named as 2,7-Naphthalenedisulfonicacid, 4-amino-5-hydroxy-. In addition, the molecular formula is C10H9NO7S2 and the molecular weight is 319.31.
Physical properties about this chemical are: (1)ACD/LogP: -3.45; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): -7.95; (4)ACD/LogD (pH 7.4): -7.98; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 8; (10)#H bond donors: 5; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 115.97 Å2; (13)Index of Refraction: 1.764; (14)Molar Refractivity: 70.15 cm3; (15)Molar Volume: 169.8 cm3; (16)Polarizability: 27.81 ×10-24cm3; (17)Surface Tension: 104.7 dyne/cm; (18)Density: 1.88 g/cm3.
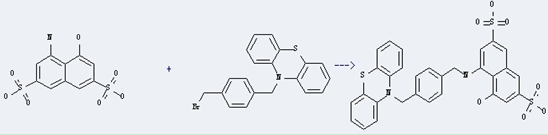
Uses of 4-Amino-5-hydroxy-2,7-naphthalene disulfonic acid: it can be used in organic synthesisas and used as intermediates of dyes and pigments. And it can react with 10-(4-bromomethyl-benzyl)-10H-phenothiazine to get 4-hydroxy-5-(4-phenothiazin-10-ylmethyl-benzylamino)-naphthalene-2,7-disulfonic acid. This reaction will need reagent AcONa and solvent H2O. The reaction time is 8 hours at reaction temperature of 5-250 °C. The yield is about 85%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. When you are using it, wear suitable protective clothing. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: O=S(=O)(O)c1cc(N)c2c(c1)cc(cc2O)S(=O)(=O)O(2)InChI: InChI=1/C10H9NO7S2/c11-8-3-6(19(13,14)15)1-5-2-7(20(16,17)18)4-9(12)10(5)8/h1-4,12H,11H2,(H,13,14,15)(H,16,17,18)
(3)InChIKey: APRRQJCCBSJQOQ-UHFFFAOYAZ
Related Products
- 1-Amino-8-hydroxynaphthalene-3,6-disulphonic acid
- 90203-05-7
- 902-04-5
- 90207-55-9
- 90210-34-7
- 902133-66-8
- 902135-89-1
- 902135-90-4
- 90213-66-4
- 902154-51-2
- 90217-02-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View