-
Name
10-(2,5-Dihydroxyphenyl)-10H-9-oxa-10-phospha-phenantbrene-10-oxide
- EINECS 619-409-6
- CAS No. 99208-50-1
- Article Data9
- CAS DataBase
- Density 1.498 g/cm3
- Solubility 1.15mg/L at 20℃
- Melting Point 245-255 °C
- Formula C18H13O4P
- Boiling Point 592.882 °C at 760 mmHg
- Molecular Weight 324.273
- Flash Point 312.364 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 1,4-Benzenediol,2-(6H-dibenz[c,e][1,2]oxaphosphorin-6-yl)-, P-oxide;6H-Dibenz[c,e][1,2]oxaphosphorin,1,4-benzenediol deriv.;2-(6-Oxido-6H-dibenz[c,e][1,2]oxaphosphorin-6-yl)-1,4-benzenediol;HCA-HQ;PHQ;Sanko HCA-HQ;DOPO-HQ;
- PSA 76.57000
- LogP 3.38610
Synthetic route
-
-
106-51-4
p-benzoquinone

-
-
35948-25-5
9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| In 2-ethoxy-ethanol at 125℃; for 4h; Inert atmosphere; | 93% |
| In toluene at 110℃; for 3.5h; Temperature; Inert atmosphere; | 92% |
| In toluene at 120℃; for 5h; Inert atmosphere; | 91.2% |
-
-
23030-43-5
2,5-dihydroxy-1-iodobenzene

-
-
35948-25-5
9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium acetate In 1,4-dioxane at 100℃; for 12h; Inert atmosphere; | 65% |
-
-
583-69-7
2-bromobenzene-1,4-diol

-
-
35948-25-5
9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium acetate In 1,4-dioxane at 100℃; for 24h; Inert atmosphere; | 25% |
-
-
2574-25-6
Diisopropyl chlorophosphate

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; for 8h; Reflux; | 93% |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
-
819-43-2
dibutyl chlorophosphate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; for 8h; Reflux; | 89.2% |
-
-
814-49-3
diethyl chlorophosphate

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; for 8h; Reflux; | 82.6% |
-
-
2510-89-6
dipropyl chlorophosphate

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; for 8h; Reflux; | 80.5% |
-
-
813-77-4
Dimethyl chlorophosphate

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; for 8h; Reflux; | 78% |
-
-
54757-38-9
di(sec-butyl) phosphoryl chloride

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; for 8h; Reflux; | 76.2% |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
-
506-77-4
cyanogen chloride

-
-
1018986-69-0
C20H11N2O4P

| Conditions | Yield |
|---|---|
| With triethylamine In acetone at -9 - -6℃; for 2.75h; | 75% |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
-
1384457-22-0
2-(6-oxido-6H-dibenzo[c,e][1,2]oxaphosphinine-6-yl)cyclohexa-2,5-diene-1,4-dione

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide In acetone at 20℃; for 1h; | 68.8% |
-
-
350-46-9
4-Fluoronitrobenzene

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl acetamide at 160℃; for 12h; Inert atmosphere; | 67% |
| With cesium fluoride In N,N-dimethyl acetamide at 160℃; for 10h; | 55.55% |
-
-
1592-20-7
4-Vinylbenzyl chloride

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| Stage #1: 10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide With sodium hydroxide In toluene at 70℃; for 0.5h; Stage #2: 4-Vinylbenzyl chloride With tetrabutylammomium bromide In toluene at 70℃; for 10h; | 65% |
| Stage #1: 10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide With sodium hydroxide In toluene at 70℃; for 0.5h; Stage #2: 4-Vinylbenzyl chloride With tetrabutylammomium bromide In toluene for 10h; | 65% |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In tetrahydrofuran | 58% |
-
-
59748-18-4
4'-octyloxy(1,1'-biphenyl)-4-carboxylic acid

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In tetrahydrofuran for 6h; | 28% |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
-
823222-06-6
4-{4-[2-(2-methoxyethoxy)ethoxy]phenyl} benzoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In tetrahydrofuran | 27% |
-
-
106-89-8
epichlorohydrin

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With sodium methylate Etherification; | |
| With N-benzyl-N,N,N-triethylammonium chloride at 95℃; for 4h; Reagent/catalyst; Temperature; Inert atmosphere; |
-
-
59748-16-2
4'-n-hexyloxybiphenyl-4-carboxylic acid

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In tetrahydrofuran |

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide
-
-
50-00-0
formaldehyd

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
-
3863-11-4
3,4-difluoroaniline

-
-
1151509-52-2
C34H23F4N2O4P

| Conditions | Yield |
|---|---|
| at 90℃; |

-
-
50-00-0
formaldehyd

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| Heating; |

-
-
350-46-9
4-Fluoronitrobenzene

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
B
-
20638-32-8
1,4'-bis(4-nitrophenoxy)benzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl acetamide at 160℃; for 12h; Inert atmosphere; |

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
-
1384457-24-2
6-(2-(diphenylphosphoryl)-3,6-dihydroxyphenyl)-6H-dibenzo[c,e][1,2]oxaphosphinine 6-oxide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: manganese(IV) oxide / acetone / 1 h / 20 °C 2: toluene / 3 h / Inert atmosphere; Reflux View Scheme |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
-
1384457-25-3
6-(2-(5,5-dimethyl-2-oxido-1,3,2-dioxaphosphinan-2-yl)-3,6-dihydroxyphenyl)-6H-dibenzo[c,e][1,2]oxaphosphinine 6-oxide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: manganese(IV) oxide / acetone / 1 h / 20 °C 2: toluene / 3 h / Inert atmosphere; Reflux View Scheme |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

-
-
1384457-27-5
6-(4-hydroxy-3-(6-oxido-6H-dibenzo[c,e][1,2]-oxaphosphinine-6-yl)phenoxy)-6H-dibenzo[c,e][1,2]-oxaphosphinine 6-oxide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: manganese(IV) oxide / acetone / 1 h / 20 °C 2: toluene / 3 h / Inert atmosphere; Reflux 3: triethylamine / toluene / 120 h / Reflux View Scheme |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: manganese(IV) oxide / acetone / 1 h / 20 °C 2: toluene / 3 h / Inert atmosphere; Reflux View Scheme |
-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: manganese(IV) oxide / acetone / 1 h / 20 °C 2: toluene / 3 h / Inert atmosphere; Reflux View Scheme |
-
-
2451-62-9
teroxirone

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| at 150℃; for 5h; |
-
-
557-40-4
Allyl ether

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

| Conditions | Yield |
|---|---|
| With magnesium sulfate; 3-chloro-benzenecarboperoxoic acid In chloroform at 25℃; for 8h; Inert atmosphere; |
-
-
523-44-4
α-Naphthol Orange

-
-
82684-68-2
1,4-bis[4-(carbonochloridoyl)phenyl] benzene-1,4-dicarboxylate

-
-
99208-50-1
10‑(2,5‑dihydroxyphenyl)‑10H-9‑oxa‑10‑phosphaphenanthrene‑10‑oxide

10-(2,5-Dihydroxyphenyl)-10H-9-oxa-10-phospha-phenantbrene-10-oxide Chemical Properties
Empirical Formula: C18H13O4P
Molecular Weight: 324.2672g/mol
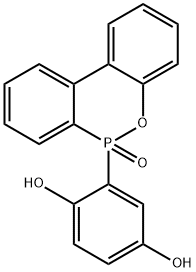
Structure of 1,4-Benzenediol,2-(6-oxido-6H-dibenz[c,e][1,2]oxaphosphorin-6-yl)- (CAS NO.99208-50-1):
Index of Refraction: 1.722
Molar Refractivity: 85.657 cm3
Molar Volume: 216.443 cm3
Polarizability: 33.957×10-24cm3
Surface Tension: 76.6 dyne/cm
Density: 1.498 g/cm3
Flash Point: 312.364 °C
Enthalpy of Vaporization: 91.669 kJ/mol
Meliting Point: 245-255 °C
Boiling Point: 592.882 °C at 760 mmHg
Vapour Pressure: 0 mmHg at 25°C
Product Categories: Industrial/Fine Chemicals
Systematic Name: 2-(6-Oxido-6H-dibenzo[c,e][1,2]oxaphosphinin-6-yl)benzene-1,4-diol
SMILES: Oc1cc(c(O)cc1)P3(=O)Oc4ccccc4c2ccccc23
InChI: InChI=1/C18H13O4P/c19-12-9-10-15(20)18(11-12)23(21)17-8-4-2-6-14(17)13-5-1-3-7-16(13)22-23/h1-11,19-20H
InChIKey: KMRIWYPVRWEWRG-UHFFFAOYAE
10-(2,5-Dihydroxyphenyl)-10H-9-oxa-10-phospha-phenantbrene-10-oxide Specification
1,4-Benzenediol,2-(6-oxido-6H-dibenz[c,e][1,2]oxaphosphorin-6-yl)- , its cas register number is 99208-50-1. It also can be called 2-(6-Oxido-6H-dibenzo[c,e][1,2]oxaphosphinin-6-yl)benzene-1,4-diol .
Related Products
- 10-(2-(1-Methyl-2-piperidyl)ethyl)-2-methylsulfinyl phenothiazine
- 10-(2-(4-METHYL-1-PIPERAZINYL)ETHYL)-PHENOTHIAZINE
- 10-(2-(DIETHYLAMINO)PROPYL)-10H-PYRIDO(3,2-B)(1,4)BENZOTHIAZINE
- 10-(2-(DIMETHYLAMINO)ETHYL)ISOALLOXAZINE SULFATE
- 10-(2-(DIMETHYLAMINO)PROPYL)PHENO-THIAZIN-2-YL MORPHOLINOMETHYL KETONE
- 10-(2,5-Dihydroxyphenyl)-10H-9-oxa-10-phospha-phenantbrene-10-oxide
- 10-(2-Benzothiazolyl)-2,3,6,7-tetrahydro-1,1,7,7-tetramethyl-1H,5H,11H-(1)benzopyropyrano(6,7-8-I,j)quinolizin-11-one
- 10-(2-DIETHYLAMINOPROPYL)PHENOTHIAZINE
- 10-(2-Naphthyl)anthracene-9-boronic acid
- 99210-65-8
- 99210-90-9
- 99212-30-3
- 99-21-8
- 992-21-2
- 99-24-1
- 992-59-6
- 99260-66-9
- 99-26-3
- 99270-16-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View