-
Name
1H-Pyrazole-4-carboxaldehyde
- EINECS 636-879-8
- CAS No. 35344-95-7
- Article Data13
- CAS DataBase
- Density 1.322 g/cm3
- Solubility
- Melting Point 82-83 °C(Solv: cyclohexane (110-82-7))
- Formula C4H4N2O
- Boiling Point 300 °C at 760 mmHg
- Molecular Weight 96.0886
- Flash Point 139.7 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 4-Formylpyrazole;4-Pyrazolecarboxaldehyde;
- PSA 45.75000
- LogP 0.22220
Synthetic route
-
-
37622-90-5
4-ethoxycarbonylpyrazole

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With n-butyllithium; diisobutylaluminium hydride; tert-butyl alcohol In tetrahydrofuran; hexane at 0℃; | 97% |
-
-
18655-47-5
triformylmethane

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In methanol at 20℃; for 5h; | 87% |
| With hydrogenchloride; hydrazine hydrate In methanol for 22h; Ambient temperature; | 52% |
| With hydrogenchloride; hydrazine hydrate In methanol for 22h; Ambient temperature; other hydrazines and amidines; | 52% |
| With hydrogenchloride; hydrazine In methanol at 20℃; for 22h; |
-
-
25222-43-9
(1H-pyrazol-4-yl)-methanol

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide In acetone at 60℃; for 4h; | 52.33% |
| With manganese(IV) oxide In acetone at 20 - 60℃; |
-
-
2075-45-8
4-bromo-1H-pyrazole

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1H-pyrazole With tert.-butyl lithium In tetrahydrofuran; hexane at -78 - 20℃; for 3.5h; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran; hexane at 20℃; | 41% |

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 1,4-dioxane at 60℃; | 0.4 g |
-
-
153687-35-5
1-(4-methoxybenzyl)-1H-pyrazole-4-carbaldehyde

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid for 2h; Heating / reflux; |
-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
591-86-6
acetaldehyde semicarbazone

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃; for 1h; Stage #2: acetaldehyde semicarbazone at 80℃; for 1.5h; |

-
A
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
B
-
112253-17-5
6-nitro-<1,2,5>oxadiazolo<3,4-b>pyridine 1-oxide

| Conditions | Yield |
|---|---|
| In dimethylsulfoxide-d6 |
-
-
1197943-43-3
1-(tetrahydro-2H-pyran-2-yl)-1H-pyrazole-4-carboxaldehyde

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 20℃; pH=< 1; | 6 g |
-
-
373-88-6
2,2,2-trifluroethylamine hydrochloride

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
1424799-64-3
(1H-pyrazol-4-ylmethylene)(2,2,2-trifluoroethyl)amine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; toluene at 20℃; Cooling with ice; | 100% |
-
-
14704-31-5
3-(bromomethyl)-1,1'-biphenyl

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; | 98% |
-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
540-51-2
2-bromoethanol

-
-
1082065-98-2
1-(2-hydroxyethyl)-1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 150℃; for 0.333333h; Microwave irradiation; | 97% |
| With potassium carbonate In acetonitrile at 150℃; for 0.333333h; Microwave irradiation; | 97% |
| With potassium carbonate In acetonitrile at 150℃; for 0.666667h; Sealed tube; Microwave irradiation; | 89% |
| With potassium carbonate In acetonitrile at 150℃; for 0.5h; Microwave irradiation; | 69% |
-
-
824-94-2
p-methoxybenzyl chloride

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
153687-35-5
1-(4-methoxybenzyl)-1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 1h; Reflux; | 93.3% |
-
-
586-78-7
para-nitrophenyl bromide

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
37921-21-4
1-(4-nitrophenyl)-1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With quebrachitol; copper; caesium carbonate In water; dimethyl sulfoxide at 100℃; Ullmann Condensation; Inert atmosphere; Green chemistry; | 93% |
| With D-galacturonic acid; potassium carbonate; copper(I) bromide In water; dimethyl sulfoxide at 80 - 100℃; Inert atmosphere; Green chemistry; | 93% |
| With copper; caesium carbonate; methyl-alpha-D-glucopyranoside In water; dimethyl sulfoxide at 100℃; Sealed tube; Green chemistry; | 90% |
-
-
110-87-2
3,4-dihydro-2H-pyran

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
1197943-43-3
1-(tetrahydro-2H-pyran-2-yl)-1H-pyrazole-4-carboxaldehyde

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In tetrahydrofuran at 90℃; for 4h; | 92% |
| With toluene-4-sulfonic acid In dichloromethane at 20 - 60℃; for 10.5h; | 86% |

| Conditions | Yield |
|---|---|
| With Porcine Pancreas Lipase In neat (no solvent) at 45℃; for 4h; Catalytic behavior; Reagent/catalyst; Temperature; Solvent; Enzymatic reaction; | 92% |

-
-
19788-37-5
4-chloromethyl-3,5-dimethylisoxazole

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
1082065-96-0
1-(3,5-dimethylisoxazol-4-ylmethyl)-1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1H-pyrazole-4-carbaldehyde With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.333333h; Stage #2: 4-chloromethyl-3,5-dimethylisoxazole In N,N-dimethyl-formamide at 20℃; for 18h; | 91% |
-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
132945-75-6
(S)-tert-butyl 3-(methylsulfonyloxy)pyrrolidine-1-carboxylate

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 100℃; for 18h; | 91% |
-
-
2567-29-5
p-phenylbenzyl bromide

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1H-pyrazole-4-carbaldehyde With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0℃; for 0.25h; Stage #2: p-phenylbenzyl bromide In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 1.58333h; | 90.3% |
-
-
540-36-3
para-difluorobenzene

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With tetrabutylammonium tetrafluoroborate; acetic acid; 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 20℃; for 36h; Sealed tube; Inert atmosphere; Irradiation; Electrolysis; | 90% |
-
-
109-99-9
tetrahydrofuran

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With C24H42N3(1+)*ClO4(1-); lithium perchlorate; acetic acid In acetonitrile at 20℃; for 36h; Irradiation; Electrochemical reaction; Inert atmosphere; | 89% |
-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
74-88-4
methyl iodide

-
-
25016-11-9
1-methylpyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 60℃; | 87% |
| With caesium carbonate In N,N-dimethyl-formamide at 60℃; for 1h; | 80% |

| Conditions | Yield |
|---|---|
| In toluene at 90℃; for 2h; | 87% |
-
-
1239320-11-6
tert-butyl 6-((methylsulfonyl)oxy)-2-azaspiro[3.3]heptane-2-carboxylate

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide for 16h; Heating; | 84.01% |
-
-
7051-34-5
cyclopropylcarbinyl bromide

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
1082065-99-3
1-(cyclopropylmethyl)-1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1H-pyrazole-4-carbaldehyde With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 0.5h; Stage #2: cyclopropylcarbinyl bromide In N,N-dimethyl-formamide; mineral oil at 20℃; for 18h; | 84% |
| Stage #1: 1H-pyrazole-4-carbaldehyde With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.333333h; Stage #2: cyclopropylcarbinyl bromide In N,N-dimethyl-formamide at 0 - 20℃; for 18h; | 56% |
-
-
107-13-1
acrylonitrile

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; choline chloride; glycerol In water at 20℃; for 1.33333h; Morita-Baylis-Hillman Alkylation; Green chemistry; | 84% |
-
-
98-59-9
p-toluenesulfonyl chloride

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
916257-05-1
1-(p-tolylsulfonyl)pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 20℃; for 19h; | 84% |
-
-
55666-43-8
tert-butyl 3-bromopropionate

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1H-pyrazole-4-carbaldehyde With caesium carbonate In N,N-dimethyl-formamide at 0℃; for 0.25h; Inert atmosphere; Stage #2: tert-butyl 3-bromopropionate In N,N-dimethyl-formamide at 100℃; for 16h; Inert atmosphere; | 83% |

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride In dichloromethane at 20℃; for 18h; | 80.7% |
-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
137049-02-6
1,2-dimethylimidazole-4-sulphonyl chloride

-
-
1082066-05-4
1-(1,2-dimethyl-1H-imidazole-4-sulfonyl)-1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 18h; | 80% |
-
-
109-04-6
2-bromo-pyridine

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With copper; caesium carbonate; methyl-alpha-D-glucopyranoside In water; dimethyl sulfoxide at 100℃; Sealed tube; Green chemistry; | 80% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
821767-61-7
tert-butyl 4-formyl-1H-pyrazole-1-carboxylate

| Conditions | Yield |
|---|---|
| dmap In acetonitrile at 20℃; for 0.5h; | 76% |
| With dmap In acetonitrile at 20℃; | 71% |
-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
254454-54-1
tert-butyl-3-iodoazetidine-1-carboxylate

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide Heating; | 75.71% |
-
-
501-53-1
benzyl chloroformate

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 4℃; for 1h; Inert atmosphere; | 74% |
-
-
19686-73-8
1-bromo-2-propanol

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
1082066-01-0
1-(2-hydroxy-propyl)-1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 150℃; for 0.333333h; Microwave irradiation; | 73% |
-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
1099-45-2
ethyl (triphenylphosphoranylidene)acetate

-
-
1396722-63-6
(E)-ethyl 3-(1H-pyrazol-4-yl)acrylate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 70℃; for 14h; | 73% |
| In tetrahydrofuran at 70℃; for 8h; Inert atmosphere; | 60% |
| In tetrahydrofuran at 20 - 70℃; for 8h; Inert atmosphere; | 60% |
-
-
352-33-0
1-Chloro-4-fluorobenzene

-
-
35344-95-7
1H-pyrazole-4-carbaldehyde

-
-
63874-99-7
1-(4-chlorophenyl)-1H-pyrazole-4-carbaldehyde

| Conditions | Yield |
|---|---|
| With tetrabutylammonium tetrafluoroborate; acetic acid; 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 20℃; for 36h; Catalytic behavior; Reagent/catalyst; Sealed tube; Inert atmosphere; Irradiation; Electrolysis; Large scale; | 73% |
1H-Pyrazole-4-carboxaldehyde Chemical Properties
Molecular Structure:
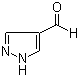
Molecular Formula: C4H4N2O
Molecular Weight: 96.0874
IUPAC Name: 1H-Pyrazole-4-carbaldehyde
Synonyms of 1H-Pyrazole-4-carboxaldehyde (CAS NO.35344-95-7): 1H-Pyrazole-4-carbaldehyde ; Akos bb-9811
CAS NO: 35344-95-7
Product Categories: Aldehydes ; Pyrazoles & Triazoles ; Pyrazoles & Triazoles
Index of Refraction: 1.62
Molar Refractivity: 25.53 cm3
Molar Volume: 72.6 cm3
Surface Tension: 64.7 dyne/cm
Density: 1.322 g/cm3
Flash Point: 139.7 °C
Enthalpy of Vaporization: 54 kJ/mol
Boiling Point: 300 °C at 760 mmHg
Vapour Pressure of 1H-Pyrazole-4-carboxaldehyde (CAS NO.35344-95-7): 0.00115 mmHg at 25°C
1H-Pyrazole-4-carboxaldehyde Safety Profile
Hazard Codes of 1H-Pyrazole-4-carboxaldehyde (CAS NO.35344-95-7):  Xi
Xi
HazardClass: IRRITANT
Related Products
- 1H-Pyrazole-4-carboxaldehyde
- 1H-Pyrazole-4-carboxaldehyde, 3-(2-thienyl)-
- 1H-Pyrazole-4-carboxaldehyde, 5-chloro-1-methyl-
- 1H-Pyrazole-4-carboxaldehyde,1-ethyl-3,5-dimethyl-
- 1H-Pyrazole-4-carboxaldehyde,1-methyl-3-phenyl-
- 1H-Pyrazole-4-carboxaldehyde,1-methyl-5-(trifluoromethyl)-
- 1H-Pyrazole-4-carboxaldehyde,1-methyl-5-phenyl-
- 1H-Pyrazole-4-carboxaldehyde,3-(4-fluorophenyl)-
- 1H-Pyrazole-4-carboxaldehyde,3,5-dimethyl-1-(phenylmethyl)-
- 1H-Pyrazole-4-carboxaldehyde,3,5-dimethyl-1-phenyl-
- 353-49-1
- 353492-16-7
- 353-50-4
- 353504-54-8
- 35351-35-0
- 35351-45-2
- 3535-30-6
- 3535-32-8
- 3535-33-9
- 3535-34-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View