-
Name
2,7-Dihydroxy-9-fluorenone
- EINECS
- CAS No. 42523-29-5
- Article Data15
- CAS DataBase
- Density 1.497 g/cm3
- Solubility
- Melting Point 338 °C
- Formula C13H8O3
- Boiling Point 444.5 °C at 760 mmHg
- Molecular Weight 212.205
- Flash Point 236.7 °C
- Transport Information
- Appearance Brown red powder
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 2,7-Dihydroxy-9H-fluoren-9-one;2,7-Dihydroxyfluoren-9-one;RMI 15152;9H-Fluoren-9-one,2,7-dihydroxy-;
- PSA 57.53000
- LogP 2.30920
Synthetic route
-
-
53133-99-6
9-oxo-9H-fluorene-2,7-diyl diacetate

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With potassium hydroxide for 2h; Heating; | 97.6% |
-
-
765944-63-6
4,4'-dihydroxy-biphenyl-2-carboxylic acid methyl ester

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 110 - 120℃; for 2h; | 95% |
| Multi-step reaction with 2 steps 1: sodium hydroxide / ethanol / 78 °C 2: thionyl chloride / 79 °C View Scheme |
-
-
53197-57-2
4,4'-dihydroxy-[1,1'-biphenyl]-2-carboxylic acid

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 210℃; Neat (no solvent); | 90% |
| With zinc(II) chloride | |
| With zinc(II) chloride at 200℃; | |
| With thionyl chloride at 79℃; Friedel-Crafts acylation; | |
| Multi-step reaction with 2 steps 1: sulfuric acid / methanol / 2 h / Reflux 2: zinc(II) chloride / 2 h / 110 - 120 °C View Scheme |
-
-
42523-28-4
2,7-dimethoxy-9H-fluorenon-9-one

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid In water for 12h; Reflux; | 87% |
| With hydrogen bromide In acetic acid |
-
-
202865-84-7
4-bromo-3-iodo-phenol

-
-
100-83-4
meta-hydroxybenzaldehyde

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium hydrogencarbonate; DL-Pro-NHMe In tert-Amyl alcohol at 120℃; for 24h; Inert atmosphere; | 74% |
-
-
89450-82-8
2,7-dihydroxy-9-oxo-fluorene-1-carboxylic acid

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With calcium oxide melting; |

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With sulfuric acid |

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one
-
-
71597-85-8
(p-hydroxyphenyl)boronic acid

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sodium carbonate / tris(dibenzylideneacetone)dipalladium(0) / 1,2-dimethoxy-ethane; H2O / 85 °C 2: sodium hydroxide / ethanol / 78 °C 3: thionyl chloride / 79 °C View Scheme |
-
-
154607-00-8
methyl 2-bromo-5-hydroxybenzoate

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sodium carbonate / tris(dibenzylideneacetone)dipalladium(0) / 1,2-dimethoxy-ethane; H2O / 85 °C 2: sodium hydroxide / ethanol / 78 °C 3: thionyl chloride / 79 °C View Scheme |
-
-
84185-72-8
3-(p-anisyl)-6-hydroxyphthalic anhydride

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 87 percent / AlCl3 / benzene / Heating 2: CaO / melting View Scheme |
-
-
84185-84-2
4-Hydroxy-4'-methoxy-biphenyl-2,3-dicarboxylic acid

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 66 percent 2: 87 percent / AlCl3 / benzene / Heating 3: CaO / melting View Scheme |
-
-
31551-45-8
2,7-dinitro-9-fluorenone

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: (i) SnCl2, HCl, AcOH, (ii) NaOH 2: aq. HBF4, NaNO2 3: aq. H2SO4 View Scheme |
-
-
2915-84-6
2,7-diaminofluoren-9-one

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. HBF4, NaNO2 2: aq. H2SO4 View Scheme |
-
-
696-62-8
para-iodoanisole

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: (i) Cu, (ii) aq. NaOH, MeOH 2: polyphosphoric acid 3: aq. HBr / acetic acid View Scheme | |
| Multi-step reaction with 2 steps 1: silver trifluoroacetate; acetic acid; 1,1,1,3',3',3'-hexafluoro-propanol / 36 h / 120 °C / Sealed tube 2: acetic acid; hydrogen bromide / water / 12 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: anthranilic acid; palladium diacetate; silver trifluoroacetate; acetic acid; 1,1,1,3',3',3'-hexafluoro-propanol / 36 h / 120 °C / Sealed tube 2: acetic acid; hydrogen bromide / water / 12 h / Reflux View Scheme |
-
-
35450-36-3
methyl 2-bromo-5-methoxybenzoate

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: (i) Cu, (ii) aq. NaOH, MeOH 2: polyphosphoric acid 3: aq. HBr / acetic acid View Scheme |
-
-
42523-25-1
4,4'-dimethoxy-[1,1'-biphenyl]-2-carboxylic acid

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: polyphosphoric acid 2: aq. HBr / acetic acid View Scheme |
-
-
86-73-7
9H-fluorene

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Stage #1: 9H-fluorene With aluminum (III) chloride; acetic anhydride Stage #2: With 3-chloro-benzenecarboperoxoic acid; trifluoroacetic acid In chloroform Further stages; | |
| Multi-step reaction with 4 steps 1.1: sulfuric acid / 100 °C 1.2: 100 °C 2.1: potassium permanganate / water / Heating 3.1: sodium hydroxide 4.1: zinc(II) chloride / 210 °C / Neat (no solvent) View Scheme | |
| Multi-step reaction with 5 steps 1.1: sulfuric acid / water / 2 h / 110 - 115 °C 1.2: 0.5 h / 20 °C 2.1: potassium permanganate / water / 2 h / 20 - 30 °C 3.1: potassium hydroxide; sodium hydroxide / 1 h / 270 - 300 °C 4.1: sulfuric acid / methanol / 2 h / Reflux 5.1: zinc(II) chloride / 2 h / 110 - 120 °C View Scheme |
-
-
13354-15-9
9H-fluorene-2,7-disulfonic acid dipotassium salt

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: potassium permanganate / water / Heating 2: sodium hydroxide 3: zinc(II) chloride / 210 °C / Neat (no solvent) View Scheme | |
| Multi-step reaction with 4 steps 1: potassium permanganate / water / 2 h / 20 - 30 °C 2: potassium hydroxide; sodium hydroxide / 1 h / 270 - 300 °C 3: sulfuric acid / methanol / 2 h / Reflux 4: zinc(II) chloride / 2 h / 110 - 120 °C View Scheme |

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: potassium hydroxide; sodium hydroxide / 1 h / 270 - 300 °C 2: sulfuric acid / methanol / 2 h / Reflux 3: zinc(II) chloride / 2 h / 110 - 120 °C View Scheme |
-
-
591-31-1
3-methoxy-benzaldehyde

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: anthranilic acid; palladium diacetate; silver trifluoroacetate; acetic acid; 1,1,1,3',3',3'-hexafluoro-propanol / 36 h / 120 °C / Sealed tube 2: acetic acid; hydrogen bromide / water / 12 h / Reflux View Scheme | |
| Multi-step reaction with 3 steps 1: 1,1,1,3',3',3'-hexafluoro-propanol / 8 h / 110 °C / Inert atmosphere 2: silver trifluoroacetate; acetic acid; 1,1,1,3',3',3'-hexafluoro-propanol / 36 h / 120 °C / Sealed tube 3: acetic acid; hydrogen bromide / water / 12 h / Reflux View Scheme |

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: silver trifluoroacetate; acetic acid; 1,1,1,3',3',3'-hexafluoro-propanol / 36 h / 120 °C / Sealed tube 2: acetic acid; hydrogen bromide / water / 12 h / Reflux View Scheme |
-
-
14348-75-5
2,7-dibromofluorene-9-one

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With lithium hydroxide monohydrate; N1-(4-hydroxy-2,6-dimethylphenyl)-N2-(4-hydroxy-3,5-dimethylphenyl)oxalamide; copper(II) acetylacetonate In water; dimethyl sulfoxide at 120℃; for 4h; | 865 mg |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 12h; Reflux; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,7-dihydroxy-9H-fluoren-9-one With potassium carbonate In N,N-dimethyl-formamide at 120℃; for 1.5h; Stage #2: 1-bromo-octane In N,N-dimethyl-formamide at 120℃; for 24h; | 95% |
| Stage #1: 2,7-dihydroxy-9H-fluoren-9-one With potassium carbonate In N,N-dimethyl-formamide at 110℃; for 1h; Stage #2: 1-bromo-octane In N,N-dimethyl-formamide at 110℃; for 24h; | 3.92 g |
-
-
96-49-1
[1,3]-dioxolan-2-one

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
129375-33-3
2,7-bis(2-hydroxyethoxy)-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With potassium carbonate at 100 - 120℃; | 93% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
62-53-3
aniline

-
-
657862-72-1
9,9-bis(4-aminophenyl)-2,7-dihydroxyfluorenone

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 150℃; for 10h; Inert atmosphere; | 92.3% |
| With methanesulfonic acid at 150℃; for 10h; | 86% |
| With aniline hydrochloride at 180℃; for 1h; | 73.6% |
| With methanesulfonic acid at 150℃; for 10h; | |
| With methanesulfonic acid at 150℃; for 14h; |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
350-46-9
4-Fluoronitrobenzene

-
-
1332524-31-8
2,7-bis(4-nitrophenoxy)-9-fluorenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1-methyl-pyrrolidin-2-one at 90℃; for 3h; Inert atmosphere; | 92.2% |
| With potassium carbonate In 1-methyl-pyrrolidin-2-one at 90℃; for 3h; Inert atmosphere; | 92.2% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
84487-73-0
3,6-Dichloro-2,7-dihydroxyfluoren-9-one

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride In acetic acid at 42 - 45℃; regioselective reaction; | 90% |
| With sulfuryl dichloride; acetic acid at 42 - 45℃; for 0.666667h; Inert atmosphere; | 90.2% |
| With sulfuryl dichloride In acetic acid at 45℃; for 0.5h; | 82% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
75-03-6
ethyl iodide

-
-
303735-64-0
2,7-diethoxy-fluoren-9-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone Heating; | 90% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
74-88-4
methyl iodide

-
-
42523-28-4
2,7-dimethoxy-9H-fluorenon-9-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 4h; Heating; | 90% |
| With potassium carbonate In N,N-dimethyl-formamide at 55℃; for 6h; | 80% |
| With sodium hydroxide In dimethyl sulfoxide for 3h; | 65% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
107-06-2
1,2-dichloro-ethane

-
-
28686-75-1
2,7-bis(2-chloroethoxy)fluoren-9-one

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride; sodium hydroxide In water at 75℃; for 24h; | 87% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| With 3-mercaptopropionic acid at 90 - 120℃; Green chemistry; | 85.6% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
869-24-9
2-chloro-N,N-diethylethylamine hydrochloride

-
-
27591-97-5
2,7-bis(2-[diethylamino]ethoxy)-9-fluorenone

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; toluene for 8h; Reflux; | 85% |
| With potassium hydroxide In water; toluene for 24h; Reflux; | 72% |
| With potassium hydroxide In toluene | |
| With potassium hydroxide; trimethylbenzylammonium bromide In toluene | |
| With potassium hydroxide In water; toluene for 24h; Reflux; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone Heating; | 85% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 60℃; for 20h; Inert atmosphere; | 84% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
96-32-2
bromoacetic acid methyl ester

-
-
123305-05-5
2,7-bis(methoxycarbonylmethoxy)fluoren-9-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 0.5h; Heating; | 83% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
109-70-6
1,3-chlorobromopropane

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 24h; | 83% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
540-63-6
ethane-1,2-dithiol

-
-
129395-16-0
2,7-dihydroxyfluoren-9-(1,3-dithiolane)

| Conditions | Yield |
|---|---|
| With aluminium trichloride In 1,4-dioxane at 50℃; for 2.5h; | 82% |
-
-
18991-98-5
n-butyl bromoacetate

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
123278-11-5
(7-Butoxycarbonylmethoxy-9-oxo-9H-fluoren-2-yloxy)-acetic acid butyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 0.5h; Heating; | 80% |
-
-
5197-66-0
2-(2-(2-(2-chloroethoxy)ethoxy)ethoxy)ethanol

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
844638-09-1
2,7-bis(2-{2-[2-(2-hydroxyethoxy)ethoxy]ethoxy}ethoxy)-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Stage #1: 2,7-dihydroxy-9H-fluoren-9-one With potassium carbonate; sodium iodide In N,N-dimethyl-formamide at 80℃; for 1h; Stage #2: 2-(2-(2-(2-chloroethoxy)ethoxy)ethoxy)ethanol In N,N-dimethyl-formamide at 80℃; for 35h; | 80% |
| Stage #1: 2,7-dihydroxy-9H-fluoren-9-one With potassium carbonate; sodium iodide In N,N-dimethyl-formamide at 60℃; for 1h; Stage #2: 2-(2-(2-(2-chloroethoxy)ethoxy)ethoxy)ethanol In N,N-dimethyl-formamide at 80℃; for 35h; | 80% |
| With potassium carbonate; sodium iodide In N,N-dimethyl-formamide at 80℃; for 35h; | 80% |
-
-
5197-67-1
14-chloro-3,6,9,12-tetraoxatetradecanol

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
1062593-11-6
2,7-bis(15-hydroxy-1,4,7,10,13-pentaoxapentadecyl)-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sodium iodide In ethanol for 50h; Heating; | 80% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
100-63-0
phenylhydrazine

-
-
84487-74-1
2,7-Dihydroxy-9-fluorenone phenylhydrazone

| Conditions | Yield |
|---|---|
| In ethanol for 4h; | 79% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
119-26-6
(2,4-dinitro-phenyl)-hydrazine

| Conditions | Yield |
|---|---|
| With sulfuric acid In methanol at 50℃; for 0.5h; | 78% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
628-89-7
2-(2-Chloroethoxy)ethanol

-
-
299167-98-9
2,7-bis[2-(2-hydroxyethoxy)ethoxy]-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| Stage #1: 2,7-dihydroxy-9H-fluoren-9-one With potassium carbonate; sodium iodide In N,N-dimethyl-formamide at 80℃; for 1h; Stage #2: 2-(2-Chloroethoxy)ethanol In N,N-dimethyl-formamide at 80℃; for 35h; | 78% |
| Stage #1: 2,7-dihydroxy-9H-fluoren-9-one With potassium carbonate; sodium iodide In N,N-dimethyl-formamide at 60℃; for 1h; Stage #2: 2-(2-Chloroethoxy)ethanol In N,N-dimethyl-formamide at 80℃; for 35h; | 78% |
| With potassium carbonate In N,N-dimethyl-formamide at 80 - 85℃; for 30h; | 78% |
| With potassium carbonate; sodium iodide In N,N-dimethyl-formamide at 80℃; for 35h; | 78% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
107-07-3
2-chloro-ethanol

-
-
129375-33-3
2,7-bis(2-hydroxyethoxy)-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 30 - 70℃; for 4h; | 78% |
| With sodium hydroxide In dimethyl sulfoxide at 70℃; | 55% |
-
-
111-24-0
1,5-dibromo-pentane

-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 24h; | 78% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 12h; | 78% |
-
-
42523-29-5
2,7-dihydroxy-9H-fluoren-9-one

-
-
84487-72-9
3,6-Dibromo-2,7-dihydroxyfluoren-9-one

| Conditions | Yield |
|---|---|
| With 1,4-dioxane dibromide at 60 - 65℃; for 0.666667h; | 77% |
2,7-Dihydroxy-9-fluorenone Chemical Properties
Structure of 2,7-Dihydroxy-9-fluorenone (CAS NO.42523-29-5):
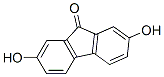
Empirical Formula: C13H8O3
Molecular Weight: 212.2008
Index of Refraction: 1.749
Molar Refractivity: 57.68 cm3
Molar Volume: 141.6 cm3
Polarizability: 22.86×10-24cm3
Surface Tension: 77.2 dyne/cm
Density: 1.497 g/cm3
Flash Point: 236.7 °C
Enthalpy of Vaporization: 72.93 kJ/mol
Boiling Point: 444.5 °C at 760 mmHg
Vapour Pressure: 1.63E-08 mmHg at 25°C
Product Categories: Fluorene Derivatives;Fluorenes, Flurenones
Synonyms of 2,7-Dihydroxy-9-fluorenone (CAS NO.42523-29-5): 9H-fluoren-9-one, 2,7-dihydroxy- ; 2,7-Dihydroxy-9-fluorenone .
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 4252-78-2
- 42530-35-8
- 4253-22-9
- 4253-24-1
- 4253-34-3
- 42534-61-2
- 425366-61-6
- 425370-68-9
- 425373-64-4
- 425379-12-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View