-
Name
AMINOMALONONITRILE P-TOLUENESULFONATE
- EINECS 225-817-1
- CAS No. 5098-14-6
- Article Data3
- CAS DataBase
- Density g/cm3
- Solubility almost transparency
-
Melting Point
174 °C (dec.)(lit.)
- Formula C7H8O3S.C3H3N3
- Boiling Point 484°Cat760mmHg
- Molecular Weight 253.282
- Flash Point 246.5°C
- Transport Information
- Appearance
- Safety 4050747
- Risk Codes R20/21/22
-
Molecular Structure
-
Hazard Symbols

- Synonyms Aminomalononitrile p-toluenesulfonate
- PSA 136.35000
- LogP 2.38366
Synthetic route

| Conditions | Yield |
|---|---|
| With mesitylenesulfonylhydroxylamine 1.) THF, 0 deg C, 2.5 h; 2.) 0 deg C, 1 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With O-(diphenylphosphinyl)hydroxylamine; sodium hydride 1.) THF, room temp., 1 h, 2.) THF, -78 deg C, 2 h, room temp.; Yield given. Multistep reaction; |
-
-
36568-05-5
2-(hydroxyimino)malononitrile

-
-
109-77-3
malononitrile

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

| Conditions | Yield |
|---|---|
| With sulfuric acid; toluene-4-sulfonic acid; acetic acid; sodium nitrite; Pt/C In water; toluene |
-
-
67-56-1
methanol

-
-
89-98-5
2-chloro-benzaldehyde

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
126210-91-1
(E)-3-Amino-2-{[1-(2-chloro-phenyl)-meth-(E)-ylidene]-amino}-3-methoxy-acrylonitrile

| Conditions | Yield |
|---|---|
| With sodium acetate at 22 - 24℃; for 6h; | 99% |

-
-
67-56-1
methanol

-
-
555-16-8
4-nitrobenzaldehdye

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
126210-93-3
(E,E)-4-amino-3-cyano-4-methoxy-1-(4-nitrophenyl)-2-aza-1,3-butadiene

| Conditions | Yield |
|---|---|
| With sodium acetate at 22 - 24℃; for 13h; | 99% |

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
2131-55-7
4-Chlorophenyl isothiocyanate

-
-
134312-08-6
5-amino-2-<(4-chlorophenyl)amino>-4-cyanothiazole

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one at 22 - 24℃; for 20h; | 96% |
| With 1-methyl-pyrrolidin-2-one at 22 - 24℃; | 58% |
-
-
403-43-0
4-fluorobenzoyl chloride

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
958633-52-8
5-amino-2-(4-fluorophenyl)oxazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 120℃; Microwave irradiation; | 96% |
| In 1-methyl-pyrrolidin-2-one at 20℃; for 144h; | 53% |
-
-
532-54-7
oxo-phenyl-acetaldehyde oxime

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
50627-20-8
2-amino-3-cyano-5-phenylpyrazine-1-N-oxide

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 20℃; for 48h; Inert atmosphere; | 95% |
| With toluene-4-sulfonic acid In isopropyl alcohol for 5h; Ambient temperature; Yield given; |
-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
98-88-4
benzoyl chloride

-
-
5098-18-0
5-amino-2-phenyl[1,3]oxazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 120℃; Microwave irradiation; | 95% |
| In various solvent(s) at 23 - 25℃; | 93% |
| In 1-methyl-pyrrolidin-2-one at 20℃; for 12h; | 79% |
| In 1-methyl-pyrrolidin-2-one at 20℃; for 192h; | 57% |
| In 1-methyl-pyrrolidin-2-one at 20℃; |
-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
122-04-3
4-nitro-benzoyl chloride

-
-
53657-73-1
2-(4-nitrophenyl)-5-amino-4-cyano-1,3-oxazole

| Conditions | Yield |
|---|---|
| In various solvent(s) at 23 - 25℃; | 95% |
| In 1-methyl-pyrrolidin-2-one at 20℃; for 168h; | 90% |
-
-
6068-72-0
4-cyanobenzoyl chlorIde

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
906512-02-5
5-amino-2-(4-cyanophenyl)oxazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 120℃; Microwave irradiation; | 95% |
-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
100-07-2
4-methoxy-benzoyl chloride

-
-
53657-71-9
5-amino-2-(4-methoxyphenyl)[1,3]oxazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 120℃; Microwave irradiation; | 94% |
| In various solvent(s) at 23 - 25℃; | 51% |
| In 1-methyl-pyrrolidin-2-one at 20℃; for 12h; | 51% |
| In 1-methyl-pyrrolidin-2-one at 20℃; |
-
-
5271-67-0
2-Thiophenecarbonyl chloride

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
909077-40-3
5-amino-2-(thiophen-2-yl)oxazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 120℃; Microwave irradiation; | 94% |
| In 1-methyl-pyrrolidin-2-one at 20℃; for 144h; | 6% |
-
-
67867-33-8
(E)-2-(furan-2-yl)-2-oxoacetaldehyde oxime

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
120930-04-3
2-amino-3-cyano-5-(furan-2-yl)pyrazine 1-oxide

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 20℃; for 70h; | 92% |
| In isopropyl alcohol at 20℃; for 70h; | 92% |
| In isopropyl alcohol for 88h; Ambient temperature; | 68% |
-
-
67-56-1
methanol

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
6630-33-7
ortho-bromobenzaldehyde

-
-
126249-12-5
(E)-3-Amino-2-{[1-(2-bromo-phenyl)-meth-(E)-ylidene]-amino}-3-methoxy-acrylonitrile

| Conditions | Yield |
|---|---|
| With sodium acetate at 22 - 24℃; for 3h; | 92% |

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
85118-06-5
2,5-difluorobenzylamine

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
417942-76-8
5-amino-4-cyano-1-(2,5-difluorobenzyl)-2-hydroxyimidazole

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; 2,5-difluorobenzylamine With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at -78℃; Stage #2: 2-aminomalononitrile p-toluenesulfonate With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 24h; | 92% |

-
-
1871-76-7
diphenylacetic acid chloride

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
1465971-36-1
5-amino-2-benzhydryl-4-cyano-1,3-oxazole

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 20℃; | 92% |
-
-
62965-10-0
(S)-N-carbobenzoxy-tert-butylleucine

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

| Conditions | Yield |
|---|---|
| With pyridine; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride at 20℃; for 18h; | 92% |
-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
132740-43-3
4-fluorobenzylisocyanate

-
-
226908-37-8
5-amino-4-cyano-1-(4-fluorobenzyl)-2-hydroxyimidazole

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran; ethyl acetate | 89% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 24h; |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In pyridine | 86% |
-
-
622-78-6
Benzyl isothiocyanate

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
624737-93-5
5-amino-2-(benzylamino)-1,3-thiazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 40℃; for 72h; | 86% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20 - 40℃; for 72h; Inert atmosphere; | 68% |
-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
3173-56-6
benzyl isothiocyanate

-
-
226908-19-6
5-amino-1-benzyl-4-cyano-2-hydroxyimidazole

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 24h; | 85% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 24h; | |
| With sodium hydroxide; N-ethyl-N,N-diisopropylamine In tetrahydrofuran; ethyl acetate | 106% |
-
-
177348-43-5
4,5-dicyano-2-pentylimidazole

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
2854-16-2
1-Amino-2-methyl-propan-2-ol

-
-
1535168-75-2
5-amino-1-(2-hydroxy-2-methylpropyl)-2-pentyl-1H-imidazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2-aminomalononitrile p-toluenesulfonate With triethylamine In tetrahydrofuran at 25℃; for 0.5h; Stage #2: 4,5-dicyano-2-pentylimidazole In tetrahydrofuran for 3h; Reflux; Stage #3: 1-Amino-2-methyl-propan-2-ol In tetrahydrofuran at 25℃; for 15h; | 85% |

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
2284-20-0
4-Methoxyphenyl isothiocyanate

-
-
134312-06-4
5-amino-4-cyano-2-<(4-methoxyphenyl)amino>thiazole

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one at 22 - 24℃; for 20h; | 83% |
| With 1-methyl-pyrrolidin-2-one at 22 - 24℃; | 55% |
-
-
919-29-9
triethyl orthovalerate

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
2854-16-2
1-Amino-2-methyl-propan-2-ol

-
-
1375262-90-0
5-amino-2-butyl-1-(2-hydroxy-2-methylpropyl)-1H-imidazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: triethyl orthovalerate; 2-aminomalononitrile p-toluenesulfonate With triethylamine In tetrahydrofuran at 25℃; for 5h; Inert atmosphere; Reflux; Stage #2: 1-Amino-2-methyl-propan-2-ol With triethylamine In tetrahydrofuran at 25℃; for 15h; Inert atmosphere; | 83% |
| Stage #1: triethyl orthovalerate; 2-aminomalononitrile p-toluenesulfonate With triethylamine In tetrahydrofuran for 5h; Reflux; Stage #2: 1-Amino-2-methyl-propan-2-ol With triethylamine In tetrahydrofuran at 25℃; for 15h; | 83% |

-
-
3282-30-2
pivaloyl chloride

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
958633-58-4
2-(t-butyl)-4-cyano-5-(2,2-dimethylpropanoyl)amino-1,3-oxazole

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 20℃; for 72h; | 82% |
-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
2131-61-5
p-nitrophenyl isothiocyanate

-
-
134312-09-7
5-amino-4-cyano-1-<(4-nitrophenyl)amino>thiazole

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one at 22 - 24℃; | 81% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 25℃; for 4h; | 81% |

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 120℃; Microwave irradiation; | 81% |
-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
79-30-1
isobutyryl chloride

-
-
124927-63-5
2-isopropyl-5-amino-4-cyano-1,3-oxazole

| Conditions | Yield |
|---|---|
| In various solvent(s) at 23 - 25℃; | 80% |
-
-
67-56-1
methanol

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
552-89-6
2-nitro-benzaldehyde

-
-
126210-92-2
(E)-3-Amino-3-methoxy-2-{[1-(2-nitro-phenyl)-meth-(E)-ylidene]-amino}-acrylonitrile

| Conditions | Yield |
|---|---|
| With sodium acetate at 22 - 24℃; for 72h; | 80% |

-
-
2799-17-9
(S)-1-amino-2-propanol

-
-
13820-09-2
trimethyl orthovalerate

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

-
-
1535168-78-5
(S)-5-amino-2-butyl-1-(2-hydroxypropyl)-1H-imidazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2-aminomalononitrile p-toluenesulfonate With triethylamine In tetrahydrofuran at 25℃; for 0.5h; Stage #2: trimethyl orthovalerate In tetrahydrofuran for 3h; Reflux; Stage #3: (S)-1-amino-2-propanol In tetrahydrofuran at 25℃; for 15h; | 80% |

-
-
18542-42-2
(2-aminoethyl)methylsulfide

-
-
13820-09-2
trimethyl orthovalerate

-
-
5098-14-6
2-aminomalononitrile p-toluenesulfonate

| Conditions | Yield |
|---|---|
| Stage #1: 2-aminomalononitrile p-toluenesulfonate With triethylamine In tetrahydrofuran at 25℃; Stage #2: trimethyl orthovalerate In tetrahydrofuran for 3.5h; Reflux; Inert atmosphere; Stage #3: (2-aminoethyl)methylsulfide With triethylamine In tetrahydrofuran at 25℃; for 18h; | 78.5% |

2-Aminomalononitrile 4-methylbenzenesulphonate Chemical Properties
Molecular Structure of 2-Aminomalononitrile 4-methylbenzenesulphonate (CAS No.5098-14-6):
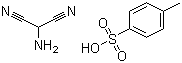
Molecular Formula: C10H11N3O3S
Molecular Weight: 253.2776
CAS No: 5098-14-6
H bond acceptors: 3
H bond donors: 1
Freely Rotating Bonds: 1
Polar Surface Area: 51.75 Å2
Flash Point: 246.5 °C
Enthalpy of Vaporization: 78.92 kJ/mol
Boiling Point: 484 °C at 760 mmHg
Vapour Pressure: 3.51E-10 mmHg at 25°C
InChI: InChI=1/C7H8O3S.C3H3N3/c1-6-2-4-7(5-3-6)11(8,9)10;4-1-3(6)2-5/h2-5H,1H3,(H,8,9,10);3H,6H2
InChIKey: MEUWQVWJLLBVQI-UHFFFAOYAV
Std. InChI: InChI=1S/C7H8O3S.C3H3N3/c1-6-2-4-7(5-3-6)11(8,9)10;4-1-3(6)2-5/h2-5H,1H3,(H,8,9,10);3H,6H2
Std. InChIKey: MEUWQVWJLLBVQI-UHFFFAOYSA-N
Product Categories: Aromatic Esters;C1 to C5;Cyanides/Nitriles;Nitrogen Compounds
IUPAC Name: 2-Aminopropanedinitrile; 4-methylbenzenesulfonic acid
2-Aminomalononitrile 4-methylbenzenesulphonate Safety Profile
Hazard Codes:  Xn
Xn
Risk Statements: 20/21/22
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
Safety Statements: 36
S36:Wear suitable protective clothing.
RIDADR: 3439
WGK Germany: 3
HazardClass: 6.1
PackingGroup: III
2-Aminomalononitrile 4-methylbenzenesulphonate Specification
2-Aminomalononitrile 4-methylbenzenesulphonate (CAS No.5098-14-6), its synonyms are Aminomalononitrile 4-methylbenzenesulfonate (1:1) ; Dicyanomethanaminium 4-methylbenzenesulfonate ; Propanedinitrile, 2-amino-, 4-methylbenzenesulfonate (1:1) .
Related Products
- 2-Aminomalononitrile 4-methylbenzenesulphonate
- 5098-16-8
- 50982-12-2
- 50982-13-3
- 50982-32-6
- 50-98-6
- 50986-18-0
- 50986-74-8
- 50990-93-7
- 50992-43-3
- 50995-48-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View