-
Name
2-Bromopentane
- EINECS 203-521-3
- CAS No. 107-81-3
- Article Data63
- CAS DataBase
- Density 1.211 g/cm3
- Solubility practically insoluble in water
- Melting Point -95 °C
- Formula C5H11Br
- Boiling Point 118.3 °C at 760 mmHg
- Molecular Weight 151.046
- Flash Point 20.6 °C
- Transport Information UN 2343 3/PG 2
- Appearance clear to yellow liquid
- Safety 16-26-36/37/39
- Risk Codes 11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xi
Xi
- Synonyms 2-Pentyl bromide;BRN 1718829;NSC7896;Pentane, 2-bromo-;
- PSA 0.00000
- LogP 2.56990
Synthetic route

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; bromine at 36℃; for 1h; | A 79% B 20% |
| With hydrogen bromide; dihydrogen peroxide In water at 20℃; for 6h; Irradiation; | |
| With manganese(IV) oxide; bromine at 36℃; for 1h; Inert atmosphere; Overall yield = 47 %; Overall yield = 0.7 g; |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; phosphorus tribromide at 80℃; for 0.666667h; | A 66% B 8% |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; phosphorus tribromide at 55℃; for 0.666667h; | A 52% B 13% |
| With hydrogen bromide; phosphorus tribromide at 55℃; | A 52% B 13% |

| Conditions | Yield |
|---|---|
| With hydrogen bromide | |
| With phosphorus tribromide | |
| With hydrogen bromide at -10℃; und nachfolgenden Erwaermen des Reaktionsgemisches auf 60grad; optically inactive 2-bromo-pentane; |

| Conditions | Yield |
|---|---|
| With hydrogen bromide | |
| With phosphorus tribromide |

| Conditions | Yield |
|---|---|
| With hydrogen bromide | |
| With phosphorus tribromide |

| Conditions | Yield |
|---|---|
| With water; hydrogen bromide at 16 - 20℃; | |
| With peroxidene; hydrogen bromide bzw.in Gegenwart von Inhibitoren wie Thiokresol,Diphenylamin oder Hydrochinon; | |
| With HFeBr4 In tetrachloromethane at 4℃; |

| Conditions | Yield |
|---|---|
| With hydrogen bromide at 25℃; optically inactive 2-bromo-pentane; |

| Conditions | Yield |
|---|---|
| With sodium bromide; diethylene glycol |

| Conditions | Yield |
|---|---|
| at 230℃; |

| Conditions | Yield |
|---|---|
| With hydrogen bromide |

| Conditions | Yield |
|---|---|
| With hydrogen bromide | |
| With hydrogen bromide optically inactive 2-bromo-pentane; | |
| With hydrogen bromide; acetic acid optically inactive 2-bromo-pentane; | |
| With hydrogen bromide; acetic acid |

| Conditions | Yield |
|---|---|
| With triethylamine at 80℃; for 3h; Yield given. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; nickel tetraaza macrocycle; water at 25℃; |


| Conditions | Yield |
|---|---|
| With Trichloroethylene; N-bromobis(trimethylsilyl)amine at 49.9℃; for 1.58333h; Product distribution; Mechanism; Irradiation; variation of temperature, time, reagents; | |
| With bromine; sodium t-butanolate In cyclohexane at 40℃; Product distribution; Further Variations:; Reagents; Temperatures; |

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
-
64-19-7
acetic acid

-
-
109-68-2
2-pentene

-
A
-
107-81-3
2-bromopentane

-
B
-
1809-10-5
3-bromopentane

| Conditions | Yield |
|---|---|
| mixtures of cis-and trans-form of pentene-(2); |


| Conditions | Yield |
|---|---|
| bei Ausschluss von Peroxyden; |

| Conditions | Yield |
|---|---|
| at 20℃; |

| Conditions | Yield |
|---|---|
| (+-)-pentanol-(2); |
-
-
1809-10-5
3-bromopentane

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
-
64-19-7
acetic acid

-
-
107-81-3
2-bromopentane

| Conditions | Yield |
|---|---|
| at 100℃; |


-
-
107-81-3
2-bromopentane

| Conditions | Yield |
|---|---|
| With hydrogen bromide dextrorotatory 2-bromo-pentane; |

| Conditions | Yield |
|---|---|
| With acetone |

-
-
107-81-3
2-bromopentane

| Conditions | Yield |
|---|---|
| With tetrachloromethane; bromine anfangs unter Kuehlung,zuletzt in der Siedehitze; |
-
-
512-85-6
ascaridole

-
-
627-20-3
(Z)-pent-2-ene

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
A
-
107-81-3
2-bromopentane

-
B
-
1809-10-5
3-bromopentane

| Conditions | Yield |
|---|---|
| at 0℃; |

-
-
646-04-8
(E)-pent-2-ene

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
A
-
107-81-3
2-bromopentane

-
B
-
1809-10-5
3-bromopentane

-
-
6032-29-7
(+/-)-2-pentanol

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
A
-
107-81-3
2-bromopentane

-
B
-
1809-10-5
3-bromopentane

| Conditions | Yield |
|---|---|
| Mengenverhaeltnis der Reaktionsprodukte; |

-
-
584-02-1
2-pentanol

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
A
-
107-81-3
2-bromopentane

-
B
-
1809-10-5
3-bromopentane

| Conditions | Yield |
|---|---|
| Mengenverhaeltnis der Reaktionsprodukte; |

-
-
107-81-3
2-bromopentane

-
-
10111-08-7
2-imidazolecarbaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 50℃; for 6h; | 99% |
-
-
107-81-3
2-bromopentane

-
-
534-18-9
sodium trithiocarbonate

-
-
89622-58-2
Trithiocarbonic acid bis-(1-methyl-butyl) ester

| Conditions | Yield |
|---|---|
| cetyltributylphosphonium bromide at 70℃; for 2h; | 94% |

| Conditions | Yield |
|---|---|
| With Li2[di-n-butylcyanocuprate] In tetrahydrofuran 1) 0 deg C, 2h, 2) RT, 1h; | 94% |
-
-
107-81-3
2-bromopentane

-
-
138851-61-3
N-(2-fluorophenyl)pyrimidin-2-amine

| Conditions | Yield |
|---|---|
| With (1,2-dimethoxyethane)dichloronickel(II); lithium tert-butoxide In 1,4-dioxane for 16h; Schlenk technique; Inert atmosphere; chemoselective reaction; | 94% |
| With N,N'-di-tert-butylethylenediamine; C40H32N12Ni2; lithium tert-butoxide In 1,4-dioxane at 120℃; for 16h; | 92% |

| Conditions | Yield |
|---|---|
| With iron(III) acetylacetonate; N,N,N,N,-tetramethylethylenediamine; hexamethylenetetramine In tetrahydrofuran at 0℃; for 1.33333h; | 93% |

| Conditions | Yield |
|---|---|
| With potassium hexafluorophosphate; 1-(1-methylbutyl)-3-methylimidazolium hexafluorophosphate at 80℃; for 15h; | 92% |

| Conditions | Yield |
|---|---|
| With sodium tetrafluoroborate; 3-(1-methylbutyl)-1-methylimidazolium tetrafluoroborate at 80℃; for 15h; | 91% |

| Conditions | Yield |
|---|---|
| With magnesium In diethyl ether at 0℃; Grignard reaction; | 90% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol Solvent; Reagent/catalyst; Reflux; | 88.1% |

| Conditions | Yield |
|---|---|
| With iron(III) acetylacetonate; N,N,N,N,-tetramethylethylenediamine; hexamethylenetetramine In tetrahydrofuran at 0℃; for 1.33333h; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: piperonal; N-trimethylsilyl N-methylcyclohexylamine With t-butyldimethylsiyl triflate In toluene for 0.05h; Inert atmosphere; Glovebox; Stage #2: 2-bromopentane With nickel(II) bromide dimethoxyethane; zinc In toluene at 50℃; for 24h; Inert atmosphere; Sealed tube; | 88% |


| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 20 - 25℃; for 11h; Molecular sieve; chemoselective reaction; | 87% |
-
-
107-81-3
2-bromopentane

-
-
66107-29-7
4-methoxyphenyl triflate

-
A
-
4125-32-0
4-(1-methyl-butyl)-anisole

-
B
-
20056-58-0
1-(4-Methoxyphenyl)pentane

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 2-(dibutylphosphanyl)-1-phenylpyrrole; magnesium; lithium chloride; zinc(II) chloride In tetrahydrofuran at 60℃; for 16h; Reagent/catalyst; Inert atmosphere; Glovebox; Sealed tube; | A n/a B 87% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 2h; Reflux; | 84.86% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 12h; Reflux; | 82% |
| With potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 80℃; for 6h; | 60% |
-
-
107-81-3
2-bromopentane

| Conditions | Yield |
|---|---|
| Stage #1: 2-(((2-hydroxyphenyl)amino)methylene)-5,5-dimethylcyclohexane-1,3-dione With sodium hydride In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: 2-bromopentane In tetrahydrofuran at 20℃; | 82% |
-
-
78646-39-6
2-(4-methoxyphenoxy)pyridine

-
-
107-81-3
2-bromopentane

| Conditions | Yield |
|---|---|
| With [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; adamantane-2-carboxylic acid; potassium carbonate In benzene at 120℃; for 24h; Inert atmosphere; Sealed tube; | 81% |
| With [RhCl2(p-cymene)]2; potassium carbonate; (adamant-1-yl)-acetic acid In benzene at 120℃; for 24h; Sealed tube; Inert atmosphere; | 62% |

| Conditions | Yield |
|---|---|
| With magnesium | 79% |
-
-
107-81-3
2-bromopentane

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide | 79% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In benzene Heating; | 76% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromopentane; Benzeneacetamide With copper(l) iodide; lithium tert-butoxide In N,N-dimethyl-formamide; acetonitrile at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: In N,N-dimethyl-formamide; acetonitrile at 20℃; for 24h; Inert atmosphere; Irradiation; | 76% |
-
-
107-81-3
2-bromopentane

| Conditions | Yield |
|---|---|
| Stage #1: 3',5'-O-bis(tert-butyldimethylsilyl)-N2-dimethylaminomethylene-2'-deoxyguanosine With caesium carbonate In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: 2-bromopentane In tetrahydrofuran at 70℃; | 75% |
-
-
107-81-3
2-bromopentane

-
-
33757-49-2
4-methyl-N-(quinolin-8-yl)benzamide

-
-
1609183-60-9
4-methyl-2-(pentan-2-yl)-N-(quinolin-8-yl)benzamide

| Conditions | Yield |
|---|---|
| With (1,2-dimethoxyethane)dichloronickel(II); Bis<2-(N,N-dimethylamino)aethyl>aether; lithium tert-butoxide In toluene at 160℃; for 20h; Inert atmosphere; | 75% |
-
-
107-81-3
2-bromopentane

-
-
1325815-45-9
1-methyl-N-(quinolin-8-yl)-1H-indole-2-carboxamide

-
-
1609183-63-2
1-methyl-2-(pentan-2-yl)-N-(quinolin-8-yl)-1H-indole-3-carboxamide

| Conditions | Yield |
|---|---|
| With (1,2-dimethoxyethane)dichloronickel(II); Bis<2-(N,N-dimethylamino)aethyl>aether; lithium tert-butoxide In toluene at 160℃; for 20h; Inert atmosphere; | 73% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); 1,3,4,6,7,8-hexahydro-2H-pyrimido[1,2-a]pyrimidine In dimethyl sulfoxide under 760.051 Torr; for 60h; Schlenk technique; Irradiation; Sealed tube; | 71% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 70℃; for 24h; | 71% |
-
-
107-81-3
2-bromopentane

-
-
27262-40-4
(S)-2',6'-pipecoloxylidide

| Conditions | Yield |
|---|---|
| Stage #1: (S)-2',6'-pipecoloxylidide With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 0.333333h; Stage #2: 2-bromopentane In N,N-dimethyl-formamide at 80℃; for 14h; | 70% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 24h; | 68% |

| Conditions | Yield |
|---|---|
| With [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; potassium carbonate; Trimethylacetic acid In 1,4-dioxane at 120℃; for 24h; Sealed tube; Inert atmosphere; | 68% |
2-Bromopentane Chemical Properties
Product Name: 2-Bromopentane (CAS NO.107-81-3)
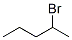
Molecular Formula: C5H11Br
Molecular Weight: 151.04 g/mol
Boiling point: 118.3 °C at 760 mmHg
Storage Temperature: Flammables area
Flash Point: 20.6 °C
Density: 1.211 g/cm3
Acidity: Stable. Highly flammable. Incompatible with strong bases, strong oxidizing agents.
Water Solubility: practically insoluble
Index of Refraction: 1.44
Molar Refractivity: 32.9 cm3
Molar Volume: 124.6 cm3
Surface Tension: 25.6 dyne/cm
Enthalpy of Vaporization: 34.18 kJ/mol
Vapour Pressure: 20 mmHg at 25°C
XLogP3-AA: 2.7
H-Bond Donor: 0
H-Bond Acceptor: 0
Structure Descriptors of 2-Bromopentane (CAS NO.107-81-3):
IUPAC Name: 2-bromopentane
Canonical SMILES: CCCC(C)Br
InChI: InChI=1S/C5H11Br/c1-3-4-5(2)6/h5H,3-4H2,1-2H3
InChIKey: LGAJYTCRJPCZRJ-UHFFFAOYSA-N
2-Bromopentane Uses
2-Bromopentane (CAS NO.107-81-3) is mainly used as an intermediate for drug synthesis.
2-Bromopentane Toxicity Data With Reference
| 1. | ihl-rat TCLo:90 mg/m3/4H/17W-I | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 25 (5)(1981),51. | ||
| 2. | ihl-mus LC50:33 g/m3 | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 25 (5)(1981),51. | ||
| 3. | ihl-mus TCLo:90 mg/m3/4H/17W-I | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 25 (5)(1981),51. | ||
| 4. | ipr-mus LD50:150 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) AD691-490 . |
2-Bromopentane Consensus Reports
Reported in EPA TSCA Inventory.
2-Bromopentane Safety Profile
Safety Information of 2-Bromopentane (CAS NO.107-81-3):
Hazard Codes: F ,Xi
,Xi
Risk Statements: 11-36/37/38
R11:Highly flammable.
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 16-26-36/37/39
S16:Keep away from sources of ignition.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
RIDADR: UN 2343 3/PG 2
WGK Germany: 3
RTECS: RZ9800000
HazardClass: 3
PackingGroup: II
2-Bromopentane Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid
2-Bromopentane Specification
2-Bromopentane , its CAS NO. is 107-81-3, the synonyms are 2-Pentyl bromide ; Pentane, 2-bromo- .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View